Introduction
Retinopathy of Prematurity (ROP) is a potentially blinding retinal vasoproliferative disorder affecting premature and low birth weight infants. With improving survival of preterm infants with lower birth weights and gestational ages, ROP is reaching epidemic proportions in many middle- income countries.1
Although a majority of infants would develop a mild self-limiting disease, some can progress to tractional retinal detachment and become permanently and irreversibly blind. Timely detection and treatment along with good supportive neonatal care offer the best chance of successful outcomes in these infants. Key pearls of ROP management are described as under:
Pearl 1: Which infants should be examined for ROP?
The guidelines for screening infants for ROP vary between different countries and geographical regions based on individual incidence rates. For India, the current guidelines recommend that all infants with a birth weight of 2000 grams or less and /or gestational age less than 34 weeks should be screened. Infants with gestational age between 34 to 36 weeks having risk factors such as those requiring prolonged oxygen support, adverse cardio-respiratory profile, poor postnatal weight gain, exchange transfusion and those with an unstable clinical course as determined by the neonatologist or paediatrician should also be screened for ROP.2
Pearl 2: When should the first ROP examination be done?
The first screening should not be delayed beyond the first 4 weeks of life (before day 30 of life) for all eligible preterm infants. Infants who are less than 28 weeks can be screened earlier within the first 2-3 weeks of life. The first examination can be done bedside in the neonatal nursey as well as within the incubator through its sloping walls.3 Subsequent follow ups can be conducted in the ophthalmology out patient service. A concerted collaborative effort starting from gynaecologists who deliver a preterm infant and administer anti-natal steroids followed by neonatologist/ pediatrician and nurses who manage the neonatal complications. Ophthalmologist as well as parents of the child are required to ensure timely screening. Communication to parents regarding the necessity of getting a dilated eye examination within the first 30 days of life lies with the paediatricians/nurses and this should be the rule rather than an exception. Delayed or absent screening is a frequent cause of late presentation and can lead to adverse outcomes following treatment despite the best of efforts.4 The examination should be done after pupillary dilatation with cyclopentolate 0.5% or a combination of 2.5% phenylephrine and 0.8% tropicamide using an indirect ophthalmoscope and a 20 or a 28 D lens. An infant wire vectis or a Schepens infant scleral depressor and an alphonso type of eye speculum can be used where peripheral visualization is difficult. Wide field retinal imaging cameras such as the RETCAM, PANOCAM, Phoenix ICON and lower cost alternatives such as the indigenous 3Nethra Neo camera5 can be used to objectively document, classify and follow up the disease. These cameras are also excellent teaching tools and also play important role in telemedicine for ROP.
Pearl 3: How to classify the disease.
The International Classification of ROP (ICROP) which was first published in 1984 and modified later in the year 2005 should be used by clinicians and researchers as a standard method to document and report the findings in ROP.6
The classification describes ROP based on its location in zones, based on severity in stages and extent as clock hours of retinal surface involved. These are summarized as follows.
Location
The retina is divided into three zones centred on the optic disc. The more posterior the location of the junction of the vascularized and non-vascularized retina the more severe is the likelihood of the disease. Zone I (Innermost) is a circle whose radius extends from the centre of the optic disc to twice the distance from the centre of the optic disc to the centre of the macula. Zone II extends centrifugally from the edge of zone I to the nasal ora-serrata and Zone III is the residual crescent of retina temporal to zone II.
Extent: Extent of retinal involvement is defined as the number of clock hours involved from 1-12. Each clock hour represents 30-degree sectors.
Severity: The clinical appearance of the stages of ROP is related to the morphology at the junction of the vascular-avascular retina. More than one stage may be present in the same eye, in which case the more severe stage is named while classifying the disease. (Figure 1)
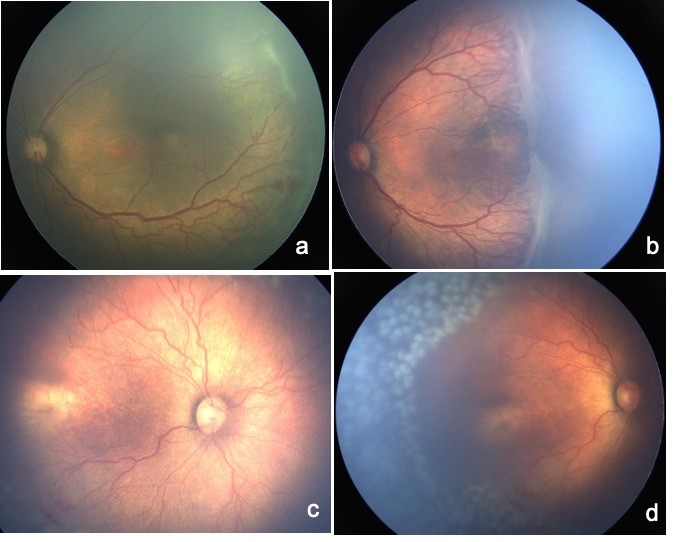
Stage 1, Demarcation line: A thin flat, white “line-like “structure at vascular-avascular junction labeled as a demarcation line.
Stage 2, Ridge: Presence of a prominent demarcation line demonstrating width and height termed as the “ridge” (Figure 1a). Small isolated tufts of new vessels may accompany the posterior aspect of the ridge as “popcorn lesions.”
Stage 3, Extraretinal fibrovascular proliferation: A ridge with extraretinal fibrovascular proliferation that extends above the plane of the retina. (Figure 1b)
Stage 4, Subtotal retinal detachment: Retinal detachment in ROP is most commonly tractional and sometimes exudative. Depending upon the involvement of the fovea it is classified as 4A (not involving the macula) and 4B (retinal detachment involving the macula).
Stage 5, Total retinal detachment: Retinal detachments in ROP are funnel-shaped, mostly tractional, funnel-shaped and further classified based on the funnel configuration into open-open, narrow-open, open-narrow, and narrow-narrow.
Pearl 4: Recognizing Aggressive Posterior ROP (APROP).
This term was introduced by the International Classification of ROP in 2005.
Prior to this, various terms such as “Rush Disease” or “Fulminate ROP” were used to describe this morphology. APROP is a rapidly progressive retinopathy characterized by increased arteriolar tortuosity and venous dilatation in all four quadrants of Zone I and posterior Zone II. The vascular features in the posterior retina are more prominent with lack of a peripheral retinopathy or “Stage.” These eyes can progress rapidly to Stage 5. Absence of a defined vascular-avascular junction, flat neovascularization, vascular loops and prominent arteriovenous shunts are frequently seen. (Figure 1c). A subset of APROP may have very severe arrest of vascularization to the extent that the fovea may also not be vascularized. These are described as posterior zone I 7 or zone ½ ROP. Such eyes may not develop a prominent plus disease but have arterio-venous shunts and arteriolar tortuosity with a prominent tunica vasculosa lentis which should alert the clinician for treatment.
Pearl 5: Recognizing Preplus and Plus disease.
These terms are used to describe the appearance of the posterior retinal blood vessels to identify eyes with high risk of progression to treatable stage of ROP. Plus disease refers to increased venous dilatation and arteriolar tortuosity, increasing iris engorgement, pupillary rigidity and vitreous haze. Standard clinical photographs have been used to compare and define the disease in the revised ICROP classification.6 At least two quadrant involvement of the signs are required to define plus disease. Presence of Plus disease warrants treatment. Preplus is defined as a state of active ROP where the vascular features are insufficient to label it as plus disease but are more marked than normal. Presence of preplus disease warrants closer follow up for progression to plus and treatment stage.
Pearl 6: Risk factors for Retinopathy of Prematurity.
ROP is a multifactorial disease 8 and the most consistent risk factor is low gestational age and low birth weight. In addition other risk factors include unblended and unmonitored supplemental oxygen, sepsis, respiratory distress, double volume exchange transfusion, anaemia, thrombocytopenia, and poor postnatal weight gain.
Pearl 7: Indications for Treatment
Treatment indications have evolved from treatment at threshold stage defined by CRYO-ROP study to treatment at earlier than threshold stages in the current times. Current indications for treatment were defined in the Early Treatment for Retinopathy of Prematurity (ETROP) study.9 Type 1 ROP (zone I ROP at any stage with plus disease, zone I ROP stage 3 without plus disease, and zone II stage 2 or 3 with plus disease) are indications for treatment. In addition any APROP should be treated promptly.
Type 2 ROP (Zone I, Stage 1 or 2 ROP without plus, Zone II Stage1 to 3 ROP without plus, peripheral avascular retina without any stage of ROP) requires follow-up till spontaneous regression or progression to type I ROP which then will qualify for treatment.
Pearl 8: Treatment of ROP
Cryotherapy was the first established therapy for ROP. Fifteen-year follow-up of this study showed a reduction of more than 40% in unfavorable structural outcomes and reduction of 30% in unfavourable visual acuity outcomes.9 Cryotherapy has however been replaced by laser photocoagulation as the standard of care for most forms of ROP in current times. Diode laser photocoagulation shown to be as effective as cryoablation was adopted as the preferred method of ablation, with lower rates of lid edema, conjunctival hyperemia, chemosis, and myopia compared to cryopexy.11 In recent times a double frequency Nd-Yag laser (532nm) which is widely available in most vitreoretinal setups has been shown to be as effective as diode laser and can be used across the spectrum of ROP even in the presence of tunica vasculosa lentis.12 Laser burns should be applied to the entire avascular retina up to the orra serrata anterior to the ridge/stage. Laser spots are applied in a near confluent manner via a laser indirect ophthalmoscope under monitoring by a pediatrician or anaesthetist. Advantages of performing laser photocoagulation include requirement of a single session in most cases, possible under topical anaesthesia, finite follow up period after disease regression, possible protection from late retinal detachments and not having long term systemic/ developmental effects.
The most recent addition to the treatment armamentarium has been Anti-VEGF therapy. The BEAT-ROP trial established the efficacy of bevacizumab 0.625 mg in treatment of stage 3+ ROP in zone1 by showing a reduction in the recurrence rates (22% in laser vs 4%) in the bevacizumab group at 54 weeks post menstrual age. At 2.5 years the Anti-VEGF treated eyes in the BEAT-ROP study showed lower degrees of myopia compared to the laser treated eyes.13
The efficacy of ranibizumab 0.2 mg in very low birth weight infants with ROP in zone I and II was reported to be 80% and 88% respectively compared to 66% with laser photocoagulation alone by the RAINBOW trail.14
Current indications for anti-VEGF therapy include zone 1 disease, infant unfit for laser therapy, severe TVL with non-dilating pupil, and as an adjunct to surgery to reduce vascularity of the proliferation.
Potential advantages of Anti-VEGF therapy include ease of administration, shorter procedure, rapid resolution of acute phase ROP changes such as plus and TVL, potential to growth of retinal vessels, potential to reduce unfavourable outcomes in posterior disease and reduced risk of myopia. The major concern is the risk of recurrences and persistent avascular retina in the periphery. Due to these reasons anti-VEGF treated eyes needs frequent and long term follow up much longer than laser treated eyes.
Surgery is advocated for progressive disease with stage 4 or 5 retinal detachments. Earliest techniques for repair of retinal detachment in ROP included scleral buckling. Advances in vitreoretinal instrumentation, have subsequently translated to similar progress in the realm of ROP surgery. With the availability of smaller gauge instrumentation (23,25,27 gauge), shorter instruments, valved cannulas, improved illumination and cutter technology vitrectomy has become safe and efficacious for ROP. Guiding principles for ROP surgery are to enter at 1-1.5 mm from the limbus in infants less than 6-month-old through the pars plicata, relieving ridge to ridge, ridge to periphery, ridge to lens and ridge to disc traction
Pearl 9: How often to follow up and when to stop screening.
The principle of regular follow up is to detect treatable ROP in time so that treatment can be offered. In general the more posterior the vascular-avascular junction or immature retina is located the closer the follow up should be. A follow up of 1 week or less is advised for immature retina, stage 1 or 2 ROP in Zone I, stage 3 ROP in zone II, and presence or suspected presence of APROP. 1-2 weekly follow up is advised for regressing ROP in zone 1, immature retina in posterior zone II or stage 2 ROP in Zone II. 2 weekly follow up is acceptable for immature retina, stage 1 ROP, or regressing ROP in Zone II. 2–3 weekly follow up is recommended for stage 1 ROP in Zone II and stage 1 or 2 ROP, regressing ROP in zone III.
Screening for ROP that did not reach treatment stage is discontinued if Zone III vascularization occurs without previous zone I or II disease, infant attains a postmenstrual age of 50 weeks or regression of previous ROP occurs without abnormal vascular tissue that could reactivate. Eyes which have undergone treatment need to be followed up longer for long term sequelae and complications. The parents should be counselled for late sequelae and refractive errors.
Pearl 10: Long term follow up
The need for long term follow up of infants cannot be stressed enough and this should be clearly communicated to parents. These infants may develop recurrences which are especially common after anti-VEGF therapy, refractive errors especially myopia, anisometropia, amblyopia, strabismus, nystagmus, cataract, glaucoma, vitreous haemorrhage and late retinal detachments which require treatment. In addition, structural sequelae such as disc drag, macular drag, narrow temporal vascular arcades, vitreous membranes, falciform retinal folds may occur.
In conclusion successful management of infants with ROP should remain focused on early detection of at-risk eyes, appropriate prompt treatment to prevent progression and subsequent long term follow up. Continued sustained efforts to improve the neonatal care are required to reduce the number of infants developing ROP in the first place.
References
- Gilbert C. Retinopathy of prematurity: a global perspective of the epidemics, population of babies at risk and implications for control. Early Hum Dev. 2008;84: 77–82.
- Guidelines for universal eye screening in newborns including retinopathy of prematurity. http://www.nhm.gov.in/images/pdf/programmes/RBSK/ Resource Documents/Revised_ROP_Guidelines-Web_Optimized.pdf. Last accessed May 31, 2021.
- Dogra MR, Vinekar A, Viswanathan K, et al. Laser treatment for retinopathy of prematurity through the incubator wall. Ophthalmic Surg Lasers Imaging. 2008;39:350-352.
- Sanghi G, Dogra MR, Katoch D, Gupta A. Demographic profile of infants with stage 5 retinopathy of prematurity in North India: implications for screening. Ophthalmic Epidemiol 2011;18:72-74.
- Vinekar A, Rao SV, Murthy S, Jayadev C, Dogra MR, Verma A, Shetty B. A Novel low cost, wide-field infant retinal camera “Neo”: technical and safety approach for use on premature infants. Transl Vis Sci Technol. 2019, 8;8(2):2.
- International Committee for the Classification of Retinopathy of Prematurity. The International Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol 2005;123:991-999.
- Katoch D, Dogra MR, Aggarwal K, Sanghi G, Samanta R, Handa S, et al. Posterior zone I retinopathy of prematurity: spectrum of disease and outcome after laser treatment. Can J Ophthalmol 2019;54: 87-93.
- Dutta S, Narang S, Narang A, Dogra M, Gupta A. Risk factors of threshold retinopathy of prematurity. Indian Pediatr 2004; 41:665-671.
- Good WV,Early Treatment for Retinopathy of Prematurity Cooperative Group. Final results of the Early Treatment for Retinopathy of Prematurity (ETROP) randomized trial.Trans Am Ophthalmol Soc. 2004;102:233-248.
- Palmer EA, Hardy RJ, Dobson V, et al.15-year outcomes following threshold retinopathy of prematurity: final results from the multicenter trial of cryotherapy for retinopathy of prematurity.Arch Ophthalmol 2005;123: 311-318.
- Connolly BP, McNamara JA, Sharma S, et al.A comparison of laser photocoagulation with trans-scleral cryotherapy in the treatment of threshold retinopathy of prematurity.Ophthalmology. 1998;105:1628–1631.
- Sanghi G, Dogra MR, Vinekar A, Gupta A. Frequency-doubled Nd: YAG (532 nm green) versus diode laser (810 nm) in treatment of retinopathy of prematurity. Br J Ophthalmol. 2010;94:1264–1265.
- Geloneck MM, Chuang AZ, Clark WL, Hunt MG, Norman AA, Packwood EA, Tawansy KA, Mintz-Hittner HA; BEAT-ROP Cooperative Group. Refractive outcomes following bevacizumab monotherapy compared with conventional laser treatment: a randomized clinical trial. JAMA Ophthalmol. 2014;132:1327-33.
- Stahl A, Lepore D, Fielder A, Fleck B, Reynolds JD, Chiang MF, et al. Ranibizumab versus laser therapy for the treatment of very low birthweight infants with retinopathy of prematurity (RAINBOW): an open-label randomised controlled trial. Lancet. 2019;394:1551-1559.

