Diabetic retinopathy (DR) is the leading cause of visual impairment worldwide. It almost accounts for 2.6% of treatable blindness globally (1). It is estimated that the overall prevalence of diabetic retinopathy is 34.6 % worldwide, while the prevalence of proliferative diabetic retinopathy (PDR) is 6.96% and that of vision-threatening DR (VTDR) is computed to be 10.2% (2).
Currently, the prevalence of PDR in India is estimated to be 17% which is significantly higher compared to patients from other locations of Asia (3). Early detection and treatment is the only key to avoiding complications and risks of vision loss (4). But the major obstacle ahead of us in India is the failure to diagnose DR in early stages; as it is observed that only 18–35% of diabetics undergo ophthalmologic evaluation regularly (5).
Pathogenesis
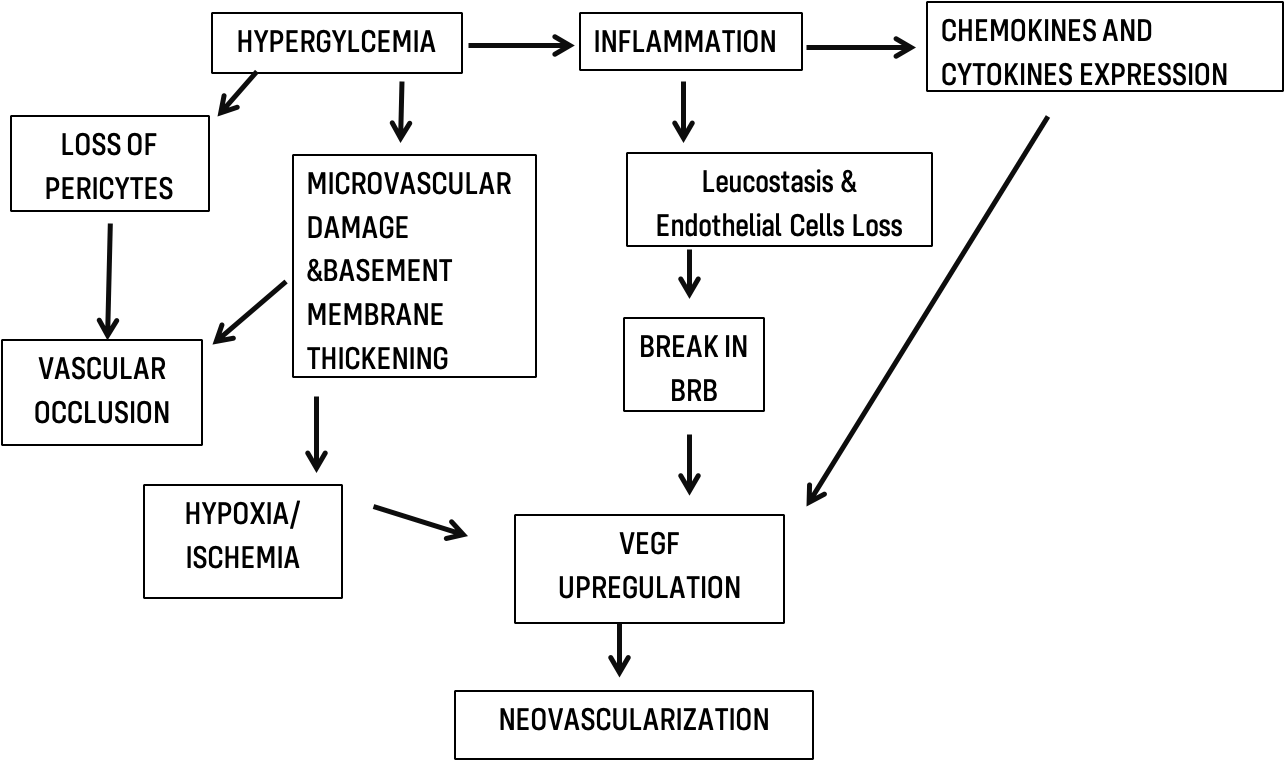
Diabetic retinopathy is recognized as a neuro-vascular complication of diabetes. The triad of hyperglycemia, inflammation, and hypoxia along with retinal neurodegeneration is responsible for the development of DR (6).
Hyperglycemia and Hypoxia
The major culprit in the development of DR is constant hyperglycemia that causes retinal microvascular damage in the form of dilatation of blood vessels and loss of pericytes, which leads to the formation of microaneurysms(6). Hence, microaneurysms are the earliest signs of DR. Another response of retinal blood vessels to hyperglycemia is the thickening of the basement membrane(7), which contributes to the impairment of the blood-retinal barrier. Loss of pericytes (8) along with thickened basement membrane, in turn, result in capillary occlusion and ischemia, leading to the upregulation of vascular endothelial growth factor (VEGF), an integral factor involved in the development of PDR in the form of the start of neovascularization.
Inflammation
Inflammation plays an essential role in the pathogenesis of DR. Leukostasis is also recognized as a key process in the early stages of DR. With an increased duration of diabetes, Leukostasis contributes to endothelial cell loss and eventually the breakdown of the blood-retinal barrier (BRB) and forms a leukocyte-endothelial adhesion complex. Chemokines& inflammatory cytokines which normally regulate the attraction and activation of leukocytes, also play a crucial role. Chemokines such as monocytes(9), chemotactic protein-1, macrophage inflammatory protein-1alpha(MIP-1) (10) & inflammatory cytokines such as TNF-α, IL-6, IL-8, and IL-1β are significantly up-regulated in diabetics, which in turn up-regulates VEGF production in the vitreous humor, marking the start of angiogenesis(11).
Retinal Neurodegeneration
Retinal neurodegeneration is an early event during the progression of DR. Retinal ganglion cells and amacrine cells are the first neurons to succumb to diabetes-induced apoptosis, followed by the photoreceptors (12). The structural consequence of this apoptotic death is the reduced thickness of the nerve fiber layer, which can be detected by optical coherence tomography (OCT) (13).
There is growing evidence that retinal neurodegeneration can be an independent entity in the development of DR and loss of ganglion cells along with thinning of retinal nerve fiber layer is observed way before microvascular alterations (14-15).
Therefore, hyperglycemia, inflammation, and hypoxia along with neurodegeneration play a collaborative role in the development of PDR. A better understanding of pathophysiology will facilitate an efficient and specific treatment and hold better prospects in the future. The cascade of events is shown in the chart below:
Risk factors
These can be identified beforehand to predict the severity of retinopathy, anticipate the findings and stage of retinopathy (2, 16).
Risk factors can be classified into (17-19):
|
Unmodifiable |
Modifiable |
|
Genetics |
Obesity |
|
Ethnicity |
Hypertension |
|
Family History |
Poor glycemic control |
|
Age |
Dyslipidemia |
|
Longer duration of disease |
Smoking |
|
Anemia |
Clinical features of PDR
Presence of new vessels is the hallmark of PDR
- Neovascularization of disc.
- Neovascularization elsewhere.
- Vitreous hemorrhage
- Sub-hyaloid hemorrhage (occasionally).
- Tractional retinal detachment, which is classified as follows:
- TRD involving the macula
- Extra-macular TRD.
- Neo-vascularization of iris, angle or neovascular glaucoma (NVG) (20).
High-risk PDR is defined as any one of the following:
1. NVD ≥ 1/3 disc area
2.Any NVD with vitreous hemorrhage
3. NVE ≥ ½ disc area with vitreous hemorrhage
High-risk PDR is also defined as three or more of the following high-risk characteristics:
1.Presence of vitreous hemorrhage or pre-retinal hemorrhage
2.Presence of any active neovascularization
3. Location of neovascularization on or within one disc diameter of the optic disc
4. NVD > 1/3 disc area or NVE > ½ disc area (21).
Investigations & Examination
Prior to investigations a thorough clinical examination is essential.
This includes: Routine vision assessment (UCVA, BCVA)
IOP measurement,
Anterior segment examination.
Posterior segment evaluation including:
1. Ophthalmoscopy. Direct or indirect ophthalmoscopy remains the mainstay for the diagnosis of diabetic retinopathy.
2. Fundus photography. It is an important tool for documentation and follow up. The traditional Early Treatment Diabetic Retinopathy Study (22), ETDRS fields have been the standard for determining severity of diabetic retinopathy for ages, which evaluates the lesion of retina, using 7 stereoscopic photographs per eye (23). The newer modalities of imaging are Widefield and Ultra-wide field which represent a major advancement in imaging. Extensive PDR lesions, captured by UWF imaging outside of ETDRS fields, carry a great prognostic value (24).
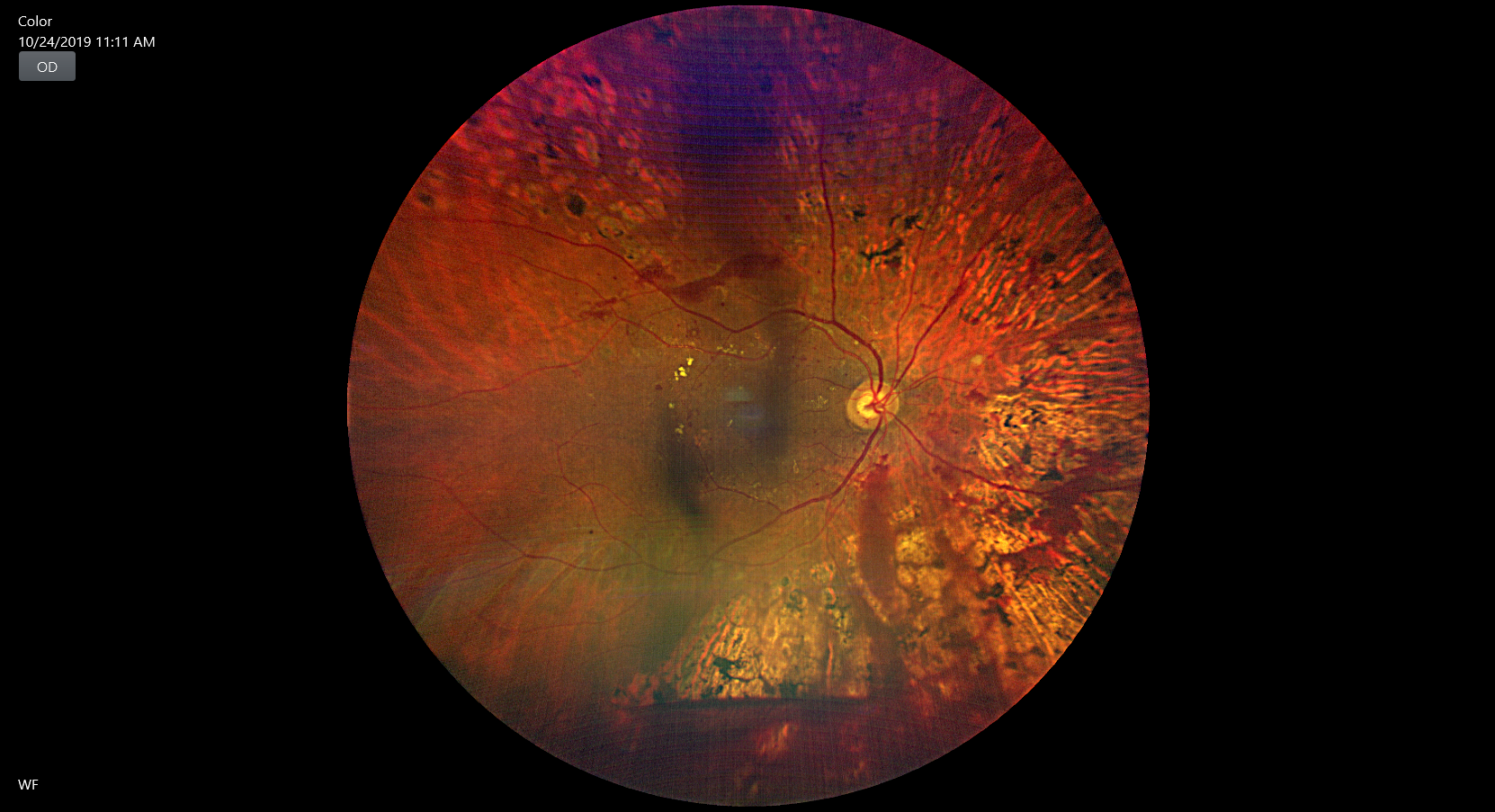
3. Fundus fluorescein angiography (FFA). FFA has been the gold standard investigation for retinal diseases. The dye has a unique property to absorb blue light (peak- 465to 490nm) and produce fluorescence at yellow-green wavelengths (520-530nm) (25).
With FFA, blood circulation of retinal vessels can be accurately understood by observing their state of fluorescence, thus discovering earlier pathologies not identified by ophthalmoscopy or slit-lamp biomicroscopy. Hyperfluorescence occurs because of vascular leakage or neovascularization & hypo or blocked fluorescence due to preretinal hemorrhage or vitreous hemorrhage.
It also detects an increase in the foveal avascular zone (FAZ) or subtle maculopathy. But the downside of it is its invasive nature and potentially serious side effects. Thus, not advisable in patients on dialysis, with kidney disease or other comorbidities (26).
Optical coherence tomography (OCT): Introduction of OCT in 1991 revolutionized visualization of the vitreoretinal interface. Its noninvasive nature with high reproducibility,the cross-section of the retina could be visualized at a glance and features that normally co-exist with PDR such as intraretinal cystic changes subretinal fluid, epiretinal membrane (ERM), vitreomacular traction or macular edema which could be missed on clinical examination if subtle
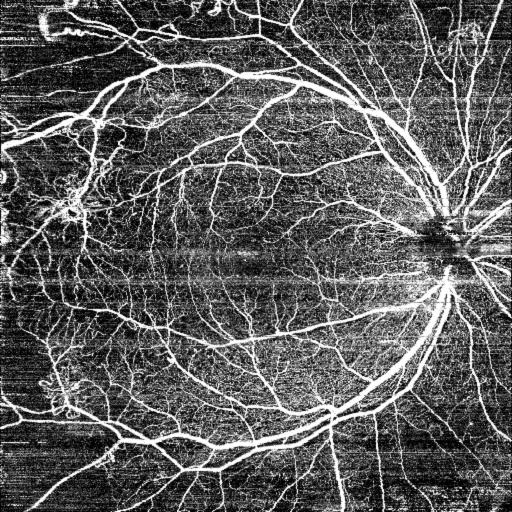
Optical coherence tomography angiography: OCTA is a promising noninvasive imaging modality. It allows visualization of the FAZ with high resolution, permitting detection of ischemic alterations, and layer-by-layer analysis of the different retinal vascular arcades. OCTA also provides a scan of the mid-periphery of the retina (figure 2), highlighting significant vascular alterations, such as neovascularization or ischemic areas (26-29).
USG B Scan: It is a valuable tool in the evaluation of eyes with vitreoretinal disorders with opaque media. As it is a dynamic scan, that evaluates the degree of mobility of the echoes corresponding to the vitreous membranes or the retina. Echoes can be classified as linear echoes representing vitreous membranes or retinal detachment and dot echoes corresponding to vitreous hemorrhage (30).
Management
A multimodal approach including lifestyle modifications, medical and surgical interventions can together prevent loss of vision due to PDR.
Some of the modalities are:
Lifestyle and dietary modifications
Nutrition plays an important role in controlling the progression of diabetes. Optimizing glycemic and lipid control is one of the first-line interventions in diabetics yet a much-neglected aspect. A balanced diet rich in omega-3, PUFAs, polyphenols, flavonoids, alkaloids, and anthocyanins is recognized as a healthy diet, and shown to have protective effects against DR and slows the progression to PDR (31-32).
Laser Photocoagulation.
Laser photocoagulation has been a mainstay for the treatment of PDR, because of long term suppressive effect on retinal neovascularization. Diabetic Retinopathy Study (DRS) Research Group showed that Pan Retinal Photocoagulation (PRP) could reduce severe vision loss in PDR by more than 50% during a two year period (21).
Indications of PRP mainly are (33):
High-risk PDR
NVD
NVI or NVA
Pre proliferative retinopathy in the other eye.
In our set up, PRP (fig-2) is performed via a slit lamp laser delivery system with approx 1200-1500, 200-micronburns, separated by the width of a burn starting with the inferior quadrant first. Normally three to four such sessions are done depending upon the severity of PDR.
Some of the complications of PRP are: Pain, Vitreous hemorrhage, decreased visual field, accidental foveal burn, macular edema, raised IOP (34). Recent developments in laser such as sub-threshold, semi‐automated patterned scanning laser, navigated laser with rapid application of multiple laser spots with shorter pulse duration have lesser complications as compared to the conventional laser delivery system. (35).
Anti-VEGF drugs
Though PRP has been the mainstay for treatment of PDR for a long time now, its destructive nature and visual field constriction associated; has led to the exploration of alternative strategies. The inhibition of VEGF has emerged as a promising treatment modality in the treatment of PDR.
DRCR.net Protocol S study evaluated efficacy and safety of ranibizumab versus PRP for 2 years and concluded that though the primary outcome of visual acuity was similar in both groups, the VEGF arm had better preserved peripheral vision (36).
It also has been reported that VEGF-treated eyes show the reduced retinal thickness and lesser chances of needing vitrectomy in the future (37-38).
Aflibercept, also known as the VEGF trap, when compared with PRP proved to be more effective, showing a higher proportion of total regression of new vessels than with PRP (39).
In another significant study, PROTEUS Study (40), which compared the efficacy of ranibizumab plus PRP versus PRP alone in patients with high-risk PDR over a 12-month period, concluded that combination treatment delivered better visual acuity and anatomical outcome. As anti-VEGF provides additional benefit when macular edema co-exists with PDR.
We feel that, in the Indian scenario, customization of treatment approaches by offering either PRP alone or in combination with Anti -VEGF therapy based on patient compliance and socioeconomic considerations is necessary.
In addition, Anti-VEGFs, are also being used preoperatively to reduce intraoperative and postoperative bleeding, surgical time, and chances of resurgery in complex cases such as macula-involving TRD with active NV or vitreous hemorrhage with extensive rubeosis (41-42).
Vitrectomy
Pars plana vitrectomy as a surgical treatment for PDR was first described more than 40 years ago. PPV offers relief from vitreoretinal traction and stabilizes the vasoproliferative process. The Diabetic Retinopathy Vitrectomy Study (DRVS) showed the superiority of timely surgical intervention over observation in preserving the vision (43).
Indications for PPV are:
- Non-Resolving vitreous hemorrhage,
- Retinal detachment involving or threatening the macula.
- Combined RD (TRD with RRD).
- Pre-macular traction with macular edema.
- Epiretinal membrane with macular edema.
Key surgical steps in vitrectomy are:
|
Core vitrectomy after sclerotomy sites |
|
Opening of the posterior hyaloid and removal of hemorrhage |
|
Membrane peeling |
|
Control of bleeding |
|
Endolaser or Cryotherapy |
|
Intraoculartamponade, if needed |
After a good vitrectomy, many factors define the visual gain prognostication of the surgery (44-45).
|
FAVORABLE FACTORS |
UNFAVORABLE FACTORS |
|
The brief duration of detachment |
Initial poor VA |
|
Presence of previous PRP |
Macular ischemia |
|
Absence of vitreous hemorrhage |
DME |
|
ERM |
|
|
NVG or NVI |
These unfavorable factors can manifest as poor visual or anatomical outcomes and many times needing revision surgery for non-resolving vitreous hemorrhage, re-detachment or development of ERM.
According to the DRIVE UK Study, patients requiring revision vitrectomy, 47.2% were because of re-detachment, 38.9% for non-resolving vitreous hemorrhage, and 13.9% for epiretinal membrane. Further analysis showed that 18.60% of eyes with TRD as the primary presentation had revision surgery as compared to 36.8% who presented with a combination of TRD and VH as the primary presentation (46).
The technique and instrumentation of vitrectomy have refined over the years, the size of the vitrectomy port has reduced from 20 gauge to 25 MIVS, now to 27 gauge. Smaller gauge instruments with higher cutting rates facilitate better dissection and shaving of fibrovascular membranes, while significantly reducing the operating time, postoperative recovery period with added patient comfort (47-48). Along with reduced incidences of postoperative hypotony, scleral wound leakage, and endophthalmitis (49). With the development and use of smaller gauge instrumentation for vitrectomy, a trend shift towards earlier surgical intervention is observed that will be helpful in decreasing the therapeutic and monetary burden of monthly intravitreal injections (50).
But, in spite of all these advancements in the treatment, prevalence, and vision loss due to PDR is still not under control. Hence newer advancements in aspects of investigations, medical and operative techniques are the need of the hour for patient-specific and tailor-made treatment approaches.
Newer developments:
- Since the introduction of OCT, it has been groundbreaking in the visualization of the vitreoretinal interface. Cross-section of the retina can be visualized at a glance. To integrate this technology into the standard operating microscope is phenomenal. Microscope-integrated iOCT enables easy visualization of peripheral retinal lesions or breaks that could be missed preoperatively and enables identification of tissue planes beneath fibrovascular membranes especially in cases of TRD with VH. The iOCT system provides real-time feedback to the surgeon during the procedure which helps in the timely alteration of the surgical steps for a better surgical outcome (51-53).
- Use of artificial intelligence in medicine in an evolving technology which can be promising as a mass screening tool. DR is an ever-increasing problem, and the number of diabetics in the coming decades is expected to increase exponentially. As the patients seeking ophthalmologist opinions for DR are still very few. Early screening and timely treatment can reduce sight-threatening retinopathy and vision loss. Any tool which can aid in quick screening and minimize the requirement of trained human resources would be a boon for patients and ophthalmologists (54-55).
In conclusion, PDR management has to have a multi-disciplinary approach including ophthalmologists, diabetologists, and pathologists and a motivated patient ready to abide by the instructions to achieve a favorable outcome as a whole.
REFERENCE:
- Bourne RR, Stevens GA, White RA, Smith JL, Flaxman SR, Price H, Jonas JB, Keeffe J, Leasher J, Naidoo K, Pesudovs K. Causes of vision loss worldwide, 1990–2010: a systematic analysis. The lancet global health. 2013 Dec 1;1(6):e339-49.
- Yau JW, Rogers SL, Kawasaki R, Lamoureux EL, Kowalski JW, Bek T, Chen SJ, Dekker JM, Fletcher A, Grauslund J, Haffner S. Global prevalence and major risk factors of diabetic retinopathy. Diabetes care. 2012 Mar 1;35(3):556-64.
- Kaveeshwar SA, Cornwall J. The current state of diabetes mellitus in India. The Australasian medical journal. 2014;7(1):45.
- Natarajan S. The sweeter side of retina. Indian journal of ophthalmology. 2014 Aug;62(8):839.
- Yang QH, Zhang Y, Zhang XM, Li XR. Prevalence of diabetic retinopathy, proliferative diabetic retinopathy and non-proliferative diabetic retinopathy in Asian T2DM patients: a systematic review and Meta-analysis. International journal of ophthalmology. 2019;12(2):302.
- Wang W, Lo AC. Diabetic retinopathy: pathophysiology and treatments. International journal of molecular sciences. 2018 Jun;19(6):1816.
- Roy S, Ha J, Trudeau K, Beglova E. Vascular basement membrane thickening in diabetic retinopathy. Current eye research. 2010 Dec 1;35(12):1045-56.
- Ejaz S, Chekarova I, Ejaz A, Sohail A, Lim CW. Importance of pericytes and mechanisms of pericyte loss during diabetic retinopathy. Diabetes, Obesity and Metabolism. 2008 Jan;10(1):53-63.
- Schröder S, Palinski W, Schmid-Schönbein GW. Activated monocytes and granulocytes, capillary nonperfusion, and neovascularization in diabetic retinopathy. The American journal of pathology. 1991 Jul;139(1):81.
- Suzuki Y, Nakazawa M, Suzuki K, Yamazaki H, Miyagawa Y. Expression profiles of cytokines and chemokines in vitreous fluid in diabetic retinopathy and central retinal vein occlusion. Japanese journal of ophthalmology. 2011 May 1;55(3):256-63.
- Boss JD, Singh PK, Pandya HK, Tosi J, Kim C, Tewari A, Juzych MS, Abrams GW, Kumar A. Assessment of neurotrophins and inflammatory mediators in vitreous of patients with diabetic retinopathy. Investigative ophthalmology & visual science. 2017 Oct 1;58(12):5594-603.
- Bringmann A, Wiedemann P. Müller glial cells in retinal disease. Ophthalmologica. 2012;227(1):1-9.
- Simó R, Stitt AW, Gardner TW. Neurodegeneration in diabetic retinopathy: does it really matter?. Diabetologia. 2018 Sep 1;61(9):1902-12.
- Sohn EH, van Dijk HW, Jiao C, Kok PH, Jeong W, Demirkaya N, Garmager A, Wit F, Kucukevcilioglu M, van Velthoven ME, DeVries JH. Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proceedings of the National Academy of Sciences. 2016 May 10;113(19):E2655-64.
- Van Dijk HW, Verbraak FD, Kok PH, Stehouwer M, Garvin MK, Sonka M, DeVries JH, Schlingemann RO, Abramoff MD. Early neurodegeneration in the retina of type 2 diabetic patients.Investigative ophthalmology & visual science. 2012 May 1;53(6):2715-9.
- Song P, Yu J, Chan KY, Theodoratou E, Rudan I. Prevalence, risk factors and burden of diabetic retinopathy in China: a systematic review and meta-analysis. Journal of global health. 2018 Jun;8(1).
- Davis MD, Fisher MR, Gangnon RE, Barton F, Aiello LM, Chew EY, Ferris FL, Knatterud GL. Risk factors for high-risk proliferative diabetic retinopathy and severe visual loss: Early Treatment Diabetic Retinopathy Study Report# 18. Investigative ophthalmology & visual science. 1998 Feb 1;39(2):233-52.
- Chew EY, Klein ML, Ferris FL, Remaley NA, Murphy RP, Chantry K, Hoogwerf BJ, Miller D. Association of elevated serum lipid levels with retinal hard exudate in diabetic retinopathy: Early Treatment Diabetic Retinopathy Study (ETDRS) Report 22. Archives of ophthalmology. 1996 Sep 1;114(9):1079-84.
- Klein R, Klein BE, Moss SE, Davis MD, DeMets DL. The Wisconsin Epidemiologic Study of Diabetic Retinopathy: II. Prevalence and risk of diabetic retinopathy when age at diagnosis is less than 30 years. Archives of ophthalmology. 1984 Apr 1;102(4):520-6.
- Bowling B. Kanski's clinical ophthalmology: a systematic approach. Saunders Ltd; 2015.
- Diabetic Retinopathy Study Research Group. Photocoagulation treatment of proliferative diabetic retinopathy: clinical application of Diabetic Retinopathy Study (DRS) findings, DRS Report Number 8. Ophthalmology. 1981 Jul 1;88(7):583-600.
- Early Treatment Diabetic Retinopathy Study Research Group. Early Treatment Diabetic Retinopathy Study design and baseline patient characteristics: ETDRS report number 7. Ophthalmology. 1991 May 1;98(5):741-56.
- Aiello LP, Odia I, Glassman AR, Melia M, Jampol LM, Bressler NM, Kiss S, Silva PS, Wykoff CC, Sun JK. Comparison of Early Treatment Diabetic Retinopathy Study standard 7-field imaging with ultrawide-field imaging for determining severity of diabetic retinopathy. JAMA ophthalmology. 2019 Jan 1;137(1):65-73.
- Liu TA, Arevalo JF. Wide-field imaging in proliferative diabetic retinopathy. International Journal of Retina and Vitreous. 2019 Dec 1;5(1):20.
- Kwan AS, Barry C, McAllister IL, Constable I. Fluorescein angiography and adverse drug reactions revisited: the Lions Eye experience. Clinical & experimental ophthalmology. 2006 Jan;34(1):33-8.
- La Mantia A, Kurt RA, Mejor S, Egan CA, Tufail A, Keane PA, Sim DA. Comparing fundus fluorescein angiography and swept-source optical coherence tomography angiography in the evaluation of diabetic macular perfusion. Retina. 2019 May 1;39(5):926-37.
- Akiyama H, Li D, Shimoda Y, Matsumoto H, Kishi S. Observation of neovascularization of the disc associated with proliferative diabetic retinopathy using OCT angiography. Japanese journal of ophthalmology. 2018 May 1;62(3):286-91.
- Stanga PE, Papayannis A, Tsamis E, Stringa F, Cole T, D'Souza Y, Jalil A. New findings in diabetic maculopathy and proliferative disease by swept-source optical coherence tomography angiography. InOCT Angiography in Retinal and Macular Diseases 2016 (Vol. 56, pp. 113-121). Karger Publishers.
- Hwang TS, Jia Y, Gao SS, Bailey ST, Lauer AK, Flaxel CJ, Wilson DJ, Huang D. Optical coherence tomography angiography features of diabetic retinopathy. Retina (Philadelphia, Pa.). 2015 Nov;35(11):2371.
- Jalkh AE, Avila MP, El-Markabi H, Trempe CL, Schepens CL. Immersion A-and B-scan ultrasonography: its use in preoperative evaluation of diabetic vitreous hemorrhage. Archives of Ophthalmology. 1984 May 1;102(5):686-90.
- Robles-Rivera, R.R., Castellanos-González, J.A., Olvera-Montaño, C., Flores-Martin, R.A., López-Contreras, A.K., Arevalo-Simental, D.E., Cardona-Muñoz, E.G., Roman-Pintos, L.M. and Rodríguez-Carrizalez, A.D., 2020. Adjuvant Therapies in Diabetic Retinopathy as an Early Approach to Delay Its Progression: The Importance of Oxidative Stress and Inflammation.Oxidative Medicine and Cellular Longevity,2020.
- Zafra‐Stone S, Yasmin T, Bagchi M, Chatterjee A, Vinson JA, Bagchi D. Berry anthocyanins as novel antioxidants in human health and disease prevention. Molecular nutrition & food research. 2007 Jun;51(6):675-83.
- Diabetic Retinopathy Study Research Group. Indications for photocoagulation treatment of diabetic retinopathy: Diabetic Retinopathy Study Report no. 14. International Ophthalmology Clinics. 1987 Dec 1;27(4):239-53.
- Reddy SV, Husain D. Panretinal photocoagulation: a review of complications. InSeminars in ophthalmology 018 Jan 2 (Vol. 33, No. 1, pp. 83-88).Taylor & Francis.
- Jhingan M, Goud A, Peguda HK, Khodani M, Luttrull JK, Chhablani J. Subthreshold microsecond laser for proliferative diabetic retinopathy: a randomized pilot study. Clinical ophthalmology (Auckland, NZ). 2018;12:141.
- Diabetic Retinopathy Study Research Group. Photocoagulation treatment of proliferative diabetic retinopathy: clinical application of Diabetic Retinopathy Study (DRS) findings, DRS Report Number 8. Ophthalmology. 1981 Jul 1;88(7):583-600.
- Li X, Zarbin MA, Bhagat N. Anti-vascular endothelial growth factor injections: the new standard of care in proliferative diabetic retinopathy?.InManagement of Diabetic Retinopathy 2017 (Vol. 60, pp. 131-142).Karger Publishers.
- Gross JG, Glassman AR, Liu D, Sun JK, Antoszyk AN, Baker CW, Bressler NM, Elman MJ, Ferris FL, Gardner TW, Jampol LM. Five-year outcomes of panretinal photocoagulation intravitrealranibizumab for proliferative diabetic retinopathy: a randomized clinical trial. JAMA ophthalmology. 2018 Oct 1;136(10):1138-48.
- Sivaprasad S, Hykin P, Prevost AT, Vasconcelos J, Riddell A, Ramu J, Murphy C, Kelly J, Edwards RT, Yeo ST, Bainbridge J. Intravitreal aflibercept compared with panretinal photocoagulation for proliferative diabetic retinopathy: the CLARITY non-inferiority RCT. Efficacy and Mechanism Evaluation. 2018;5(5):1-12.
- Figueira J, Fletcher E, Massin P, Silva R, Bandello F, Midena E, Varano M, Sivaprasad S, Eleftheriadis H, Menon G, Amaro M. Ranibizumab plus panretinal photocoagulation versus panretinal photocoagulation alone for high-risk proliferative diabetic retinopathy (PROTEUS study). Ophthalmology. 2018 May 1;125(5):691-700.
- Hu X, Pan Q, Zheng J, Song Z, Zhang Z. Reoperation following vitrectomy for diabetic vitreous hemorrhage with versus without preoperative intravitreal bevacizumab. BMC ophthalmology. 2019 Dec;19(1):1-0.
- Castillo J, Aleman I, Rush SW, Rush RB. Preoperative bevacizumab administration in proliferative diabetic retinopathy patients undergoing vitrectomy: a randomized and controlled trial comparing interval variation. American journal of ophthalmology. 2017 Nov 1;183:1-0.
- DRVS Research Group. Two-year course of visual acuity in severe proliferative diabetic retinopathy with conventional management: Diabetic Retinopathy Vitrectomy Study (DRVS) report# 1. Ophthalmology. 1985 Apr 1;92(4):492-502.
- Yorston D, Wickham L, Benson S, Bunce C, Sheard R, Charteris D. Predictive clinical features and outcomes of vitrectomy for proliferative diabetic retinopathy. British journal of ophthalmology. 2008 Mar 1;92(3):365-8.
- Yorston D, Wickham L, Benson S, Bunce C, Sheard R, Charteris D. Predictive clinical features and outcomes of vitrectomy for proliferative diabetic retinopathy. British journal of ophthalmology. 2008 Mar 1;92(3):365-8.
- Gupta B, Sivaprasad S, Wong R, Laidlaw A, Jackson TL, McHugh D, Williamson TH. Visual and anatomical outcomes following vitrectomy for complications of diabetic retinopathy: the DRIVE UK study. Eye. 2012 Apr;26(4):510-6.
- Nagpal M, Wartikar S, Nagpal K. Comparison of clinical outcomes and wound dynamics of sclerotomy ports of 20, 25, and 23 gauge vitrectomy. Retina. 2009 Feb 1;29(2):225-31.
- Kumar A, Duraipandi K, Gogia V, Sehra SV, Gupta S, Midha N. Comparative evaluation of 23-and 25-gauge microincision vitrectomy surgery in management of diabetic macular traction retinal detachment. European journal of ophthalmology. 2014 Jan;24(1):107-13.
- Pollack JS, Sabherwal N. Small gauge vitrectomy: operative techniques. Current opinion in ophthalmology. 2019 May 1;30(3):159-64.
- Mansour SE, Browning DJ, Wong K, Flynn Jr HW, Bhavsar AR. The Evolving Treatment of Diabetic Retinopathy. Clinical Ophthalmology (Auckland, NZ). 2020;14:653.
- Ehlers JP, Modi YS, Pecen PE, Goshe J, Dupps WJ, Rachitskaya A, Sharma S, Yuan A, Singh R, Kaiser PK, Reese JL. The DISCOVER study 3-year results: feasibility and usefulness of microscope-integrated intraoperative OCT during ophthalmic surgery. Ophthalmology. 2018 Jul 1;125(7):1014-27.
- Ehlers JP, Dupps WJ, Kaiser PK, Goshe J, Singh RP, Petkovsek D, Srivastava SK. The prospective intraoperative and perioperative ophthalmic imaging with optical coherence tomography (PIONEER) study: 2-year results. American journal of ophthalmology. 2014 Nov 1;158(5):999-1007.
- Khan M, Srivastava SK, Reese JL, Shwani Z, Ehlers JP. Intraoperative OCT-assisted surgery for proliferative diabetic retinopathy in the DISCOVER study. Ophthalmology Retina. 2018 May 1;2(5):411-7.
- Padhy SK, Takkar B, Chawla R, Kumar A. Artificial intelligence in diabetic retinopathy: A natural step to the future. Indian journal of ophthalmology. 2019 Jul;67(7):1004.
- Stewart MW. Treatment of diabetic retinopathy: recent advances and unresolved challenges. World journal of diabetes. 2016 Aug 25;7(16):333.

