Introduction:
Thyroid eye disease (TED) or thyroid-associated orbitopathy (TAO) also called Basedow-Grave’s disease. It is an autoimmune inflammatory disorder characterized by enlargement of the orbital fat and extraocular muscles.1 It is a consequence of thyroid gland dysfunction, where antibodies are targeted against thyrotropin receptors (TSHR) known as thyroid-stimulating globulin(TSI).1 It is independent of the serum level of the circulating hormone levels. 2
TED is characterized by an active or inflammatory stage and an inactive or fibrotic stage. The active or the inflammatory stage is characterized by inflammation, adipogenesis, and accumulation of extracellular fluid, glycosaminoglycans which in turn causes enlargement of the extraocular muscles and expansion of the orbital fat. Once the inflammation settles, there is predominant scarring of the orbital tissues which marks the onset of the fibrotic stage.
The natural course of the disease is quite variable. The majority of TED patients show a mild, self-limiting course, while 3–7% of TED patients exhibit a severe sight-threatening disease secondary to compressive optic neuropathy or corneal exposure. 2,3 A spectrum of patients fall between these two extremes and present with lid signs, proptosis motility restriction, diplopia with or without strabismus which has an adverse effect on their quality of life, both functionally and aesthetically.
Epidemiology:
About 90% of TED cases are associated with hyperthyroidism, 5% with hypothyroidism (such as Hashimoto’s thyroiditis) and about 5% are euthyroid.2
Age Between 3rd – 5th decade with greater severity in older patients.4
Sex
- Female preponderance, with M: F - 1:4.
- Men tend to have late-onset but a more severe disease as compared to females.4
Race Caucasians have greater severity of disease as compared to Asians. 4
|
NON-MODIFIABLE RISK FACTORS |
|
|
MODIFIABLE RISK FACTORS |
Smoking1,6 (Stimulates hyaluronic acid production and an increase in adipogenesis)
|
|
Selenium deficiency
|
|
|
High serum cholesterol |
|
|
Life stressors |
Pathophysiology:
Orbital fibroblasts are the main effector cells. In TED patients, orbital fibroblasts can differentiate into mature adipocytes or myofibroblasts.5
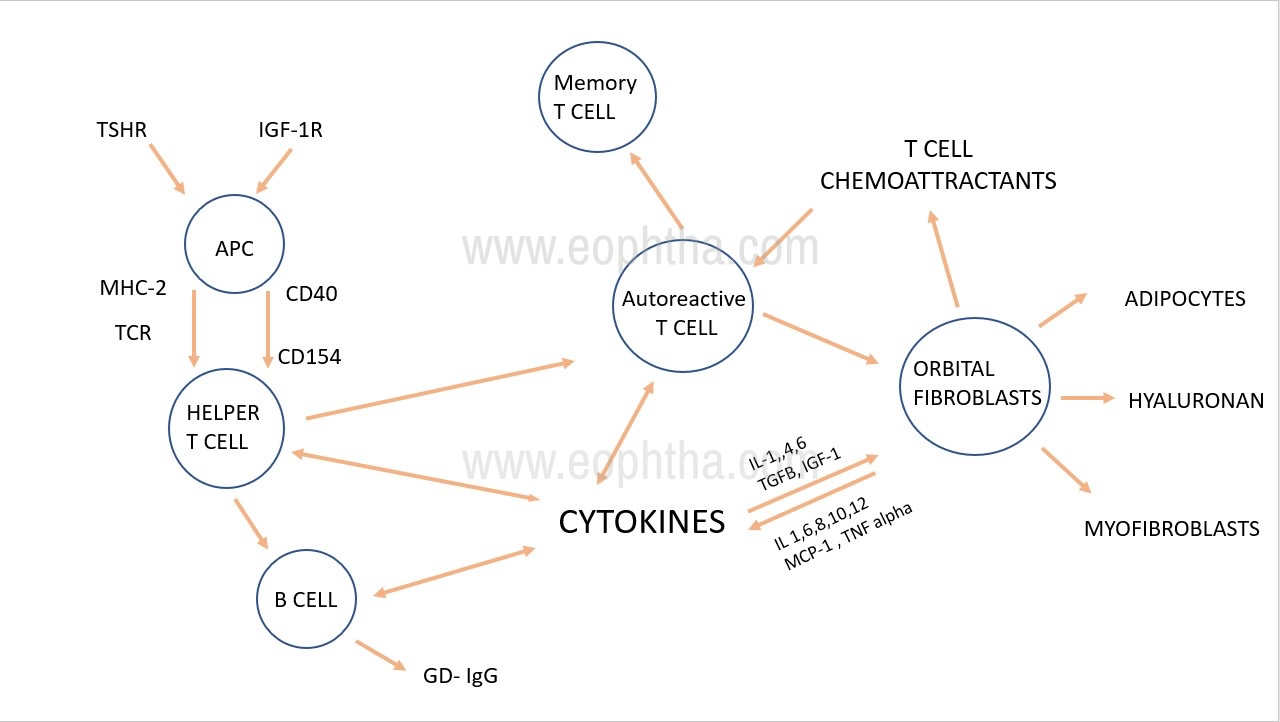
Figure 1: Pathogenesis of Thyroid Eye DiseaseAPC=Antigen-presenting cells, GDIgGs = Graves Disease Immunoglobulins, OF=orbital fibroblasts, TNF= Tumor necrosis factor, IL=interleukin
Both Thyroid-stimulating hormone receptor (TSHR) and Insulin-like growth factor-1R (IGF-1R) are antigens expressed by orbital fibroblasts. which activate the PI3K/AKT pathway to upregulate peroxisome proliferator-activated receptor-γ (PPAR-γ) expression, differentiation, and proliferation of adipocytes and enhance adipogenesis.(Figure no. 1)
Risk factors 1:
Clinical Features:
TED can be divided based on the stage of the disease into active and inactive or quiescent disease.(Figure no. 2)
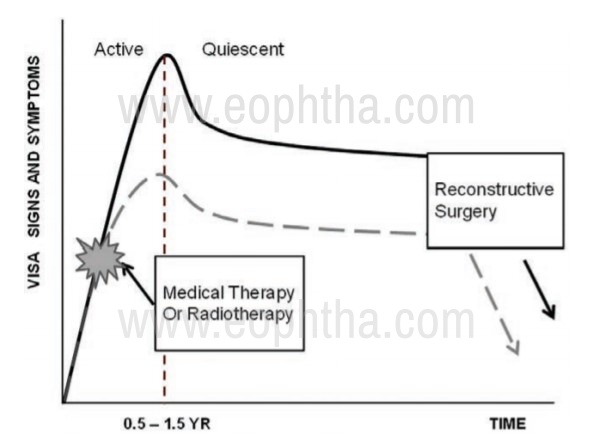
Figure 2: Rundle's curve: Describes the disease course.
|
ACTIVE DISEASE Lasts for about 6 to 24 months Proptosis Episcleral injection (at the insertion of muscles) Conjunctival injection and chemosis Diplopia Sight threatening complication secondary to corneal ulceration or compressive optic neuropathy |
QUIESCENT DISEASE Progressive fibrosis Stable proptosis Eyelid retraction Persistent restrictive strabismus No signs of inflammation Corrective Surgical management to be planned |
Symptoms
- The prominence of the eye
- Grittiness, redness, photophobia, tearing and puffy lids
- Retrobulbar dull aching pain and pain on eye movements
- Diurnal variation (worse in the morning)
- Staring look, difficulty in closing eyes and ocular surface symptoms
- Double vision, discomfort in certain positions of gaze
- Diminution of vision
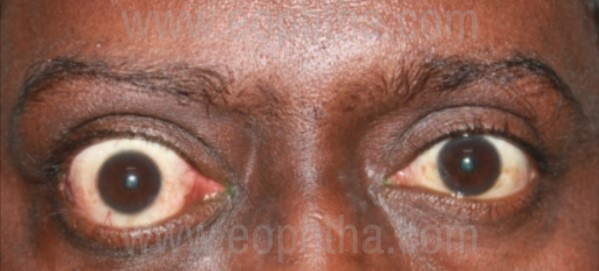
Figure 3: Clinical external picture depicting right eye axial proptosis. Note the tell-tale signs of TED. (Right upper lid and lower retraction, superior and inferior scleral show and upper lid lateral flare)
|
Soft Tissue Inflammation |
|
|
Signs
|
Cause
|
|
Eyelid |
|
|
Signs
|
Cause
|
|
Ocular Surface Discomfort |
|
|
Signs
|
Cause
|
|
Proptosis (Exophthalmos) |
|
|
FEATURES
|
CAUSE
|
|
Ocular Motility Restrictions |
|
|
FEATURES
|
CAUSE
|
|
Diplopia |
|
|
FEATURES Gorman grading for the severity of diplopia:
|
CAUSE Proptosis Mechanical Restrictive strabismus |
|
Intraocular Pressure8 |
|
|
FEATURES Categorized into
Differential IOP – Increase IOP in up gaze (> 6mm Hg- significant) Management –Treatment of Grave’s orbitopathy. Topical Antiglaucoma medication. |
CAUSE
|
|
Compressive Optic Neuropathy |
|
|
SIGNS
|
CAUSE
|
|
Signs |
|
|
Eyelid signs |
|
|
Upper lid signs |
|
|
Lower lid signs |
|
|
Extraocular movements signs |
|
|
Facial signs |
|
|
Pupillary and conjunctival signs |
|
Classification
A. No Specs/ Werners Classification 9
|
Grades |
|
0 No signs or symptoms |
|
1 Only signs, no symptoms (signs limited to upper lid retraction and stare, with or without lid lag and proptosis) |
|
2 Soft tissue involvement (symptoms and signs) |
|
3 Proptosis |
|
4 Extraocular muscle involvement |
|
5 Corneal involvement |
|
6 Sight loss (optic nerve involvement) |
B. CAS (Clinical Activity Score)9
|
Initial Visit (1 point each) |
||
|
||
|
Follow-up Visit (1 point each) |
||
|
Criteria 1-7
CAS ≥ 4 → "Active" |
C. EUGOGO Severity Scale (European Group On Graves Orbitopathy) based on clinical features7,9
|
Management category |
Criteria |
|
Mild |
Eyelid retraction Mild proptosis Minimal muscle involvement |
|
Moderate to severe |
Greater proptosis (>25mm) Inflammation Significant motility restriction |
|
Sight threatening |
Dysthyroid optic neuropathy Corneal ulceration |
D. VISA’ Classification / ITEDS (International Thyroid Eye Disease Study) 9– grades disease activity and severity based on the subjective and objective evaluation of Vision, Inflammation, Strabismus and Appearance9. More than 4 signifies activity. VISA form
CAS and VISA inflammatory score depicts the disease activity while NOSPECS, EUGOGO, VSA score of VISA define the severity of the disease.9,10
Management:
TED is a self-limiting disease, with patients moving from the active to quiescent phase within 1-3 years with a 5-10% risk of recurrence.
|
Systemic investigations |
Ocular investigation |
|
Thyroid function test (Free T3, T4, TSH) |
Vision, color vision |
|
Antibody to thyroid peroxidase (TPO) and thyroglobulin |
Fundus evaluation |
|
Rule out other associated auto-immune conditions: Myasthenia Gravis and Pernicious Anemia |
Diplopia charting |
|
Ultrasound neck (To looks for thyroid gland) |
Visual fields perimetry, VEP fundus photo (DON) |
Orbital imaging
- Ultrasound orbit – (seldomly used) can be used for distinguishing active enlargement of extraocular muscles with lower internal reflectivity from the quiescent stage showing high reflectivity.
- Computed Tomography (CT) orbit
- Useful to understand the orbital bony architecture, floor, and adjacent sinuses and enables proper surgical management.11
- Magnetic resonance imaging (MRI)
- Preferred for soft tissue delineation.
- Gives a measure of fat hypertrophy and extraocular muscle enlargement.
- Detects apical crowding and early optic nerve compression.
- Radiological activity – inflammatory oedema and activity of extraocular muscles appear hyperintense on T2 weighted images while in fibrotic stage, they appear hypointense on T2W
- Aids in monitoring response to treatment
- Barrett’s index – Using a mid-orbit scan, vertical muscle index was calculated using the superior rectus muscle–levator muscle complex (A) and inferior rectus muscles(B) along with the perpendicular orbital height (C).
- The vertical muscular index = ([A+B/C] ×100)
- The horizontal muscular index is expressed as a percentage of the sum of the horizontal recti muscles and orbital width were measured to determine the horizontal muscle index ([D+E/F]). The larger of the two was taken as muscle index with 67% or more indicating optic nerve apical compression11.
- Coco-cola bottle sign12 – The bellies of the extraocular muscles are enlarged sparing the tendons which give an appearance similar to the Coco- colaTM bottle.
- Fat effacement – is a measure of apical crowding measured on coronal images. The amount of effacement of perineural fat planes by enlarged extraocular muscles in percentage is graded into 0 -none, 1- (up to 25%), 2-(25-50%), 3-(more than 50%)10. (Figure no.4)
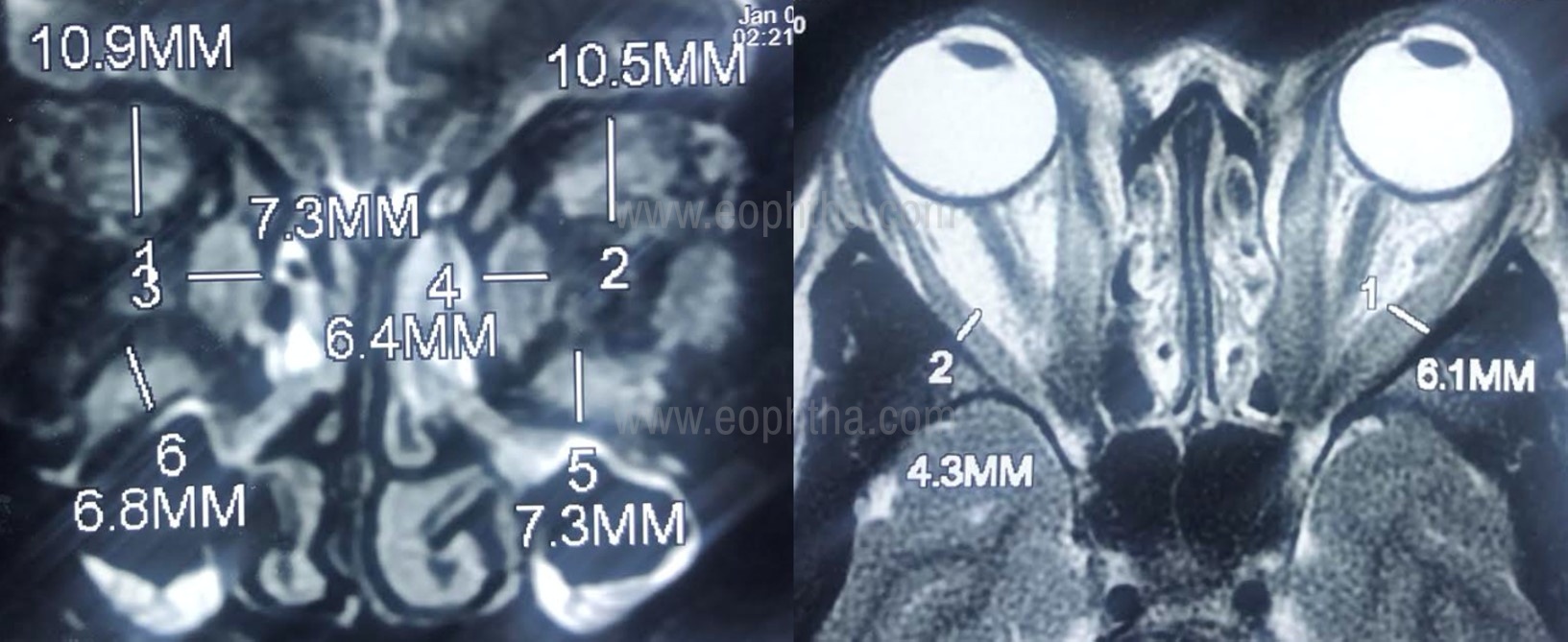
Figure 4: MRI orbits T2- weighted image (A: Coronal, B: Axial cut) showing enlargement of the extraocular muscle right orbit with intramuscular T2 hyperintense signal suggestive of radiological activity. Extraocular muscle thickness > 4 mm is considered significant.Note the classical Coco-cola bottle (B) sign depicting the tendon sparing enlargement of the muscles on axial cut.
Treatment depends on
- The stage of the disease
- Disease activity and severity based on clinical and radiological findings.
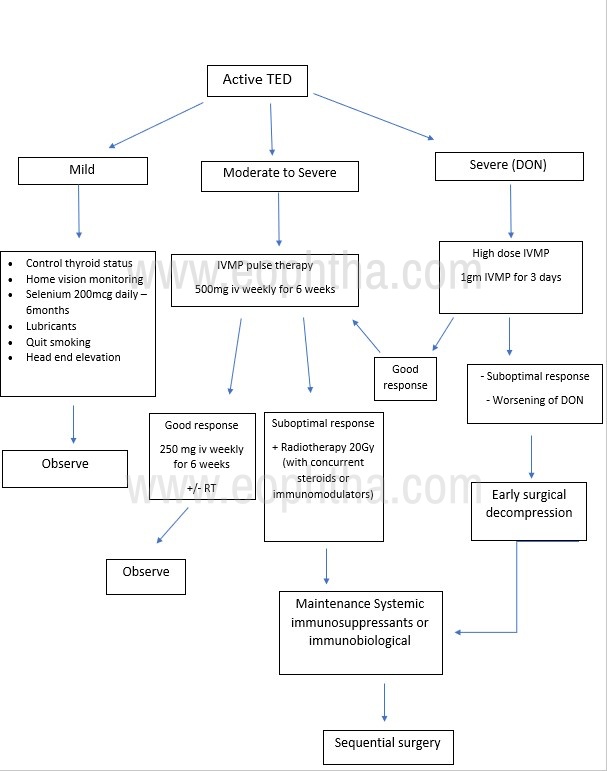
Based on various classifications available, the therapeutic protocol was charted out for mild, moderate-severe, and sight-threatening disease.
The primary goal in the active phase:
- preserve visual function
- prevent complications and sequelae
|
Disease severity |
Management |
|
Mild |
Topical lubricants Home vision monitoring Behavioral modifications
Diplopia management - with prisms/ occlusion till disease stabilization Oral antioxidants – selenium 100mcg twice a day for 6 months7 |
|
Moderate - Severe |
Medical decompression Intravenous methylprednisolone (IVMP) pulse: Initial 500 mg/week × 6 weeks Followed by 250 mg/week × 6 weeks (IVMP -8 g of maximum cumulative dosage)13 |
|
Sight threatening [DON (Dysthyroid optic neuropathy)] |
IVMP 1gm daily for 3 days followed by pulse IVMP |
| Non- responders / Steroid dependent |
Immunomodulator drugs Orbital radiotherapy (20Gy in 10 divided fractions) Surgical decompression |
Medical Management (Progressive Disease)
Corticosteroids
- The mainstay of treatment in active disease.
- Intravenous corticosteroids
- Mechanism of action – steroids act on fibroblasts, B and T cells and reduce the release of pro-inflammatory cytokines, reduced prostaglandin release and fibroblast activity.10
- First-line therapy for compressive optic neuropathy
- (Refer Table for dosage).
- Earlier oral corticosteroids were used but are now less popular due to reduced efficacy as compared to intravenous steroids and increased systemic adverse effects.
Immunosuppressive therapy
Drugs:
- Azathioprine (50mg twice a day for 1 week up to three times a day)
- Methotrexate (525mg once per week)
- Cyclophosphamide i. v. (500-100mg every 4-8 weeks) or oral 12mg/kg every day
- Cyclosporine (50mg twice a day, increase up to 200mg twice a day)
- Regular monitoring of hematological profile, kidney and liver functions.4
Newer drugs in the management of TED10
|
Drug |
MOA |
|
Teprotumumab |
Inhibits insulin-like growth factor 1 receptor (IGF1R) signalling |
|
Rituximab |
a monoclonal antibody that targets CD20 |
|
Tocilizumab |
humanized monoclonal antibody against the IL-6 receptor |
|
Belimumab (ongoing trials) |
A monoclonal antibody directed towards the B cell-activating factor |
Orbital Irradiation
Indications: Active disease - clinically or radiologically.
Treatment regimen: 2000 cGy or 20 Gy over 10 sessions in divided fractions, during a 2-week time course (standard regime)
Can be administered with concurrent oral or intravenous steroids
- Contraindications –
- Younger patients (<35 yrs)
- Patients with microvascular retinopathy (diabetic or hypertensive retinopathy)
- Adverse ophthalmic effects
- Dry eye, cataract, worsening of retinopathy, optic neuropathy, long term secondary tumours.
Orbital Decompression
Indications
Active disease
- Sight threatening (DON), severe disease
- No response to medical management
- Severe progressive active disease with gross proptosis and exposure keratopathy
Quiescent phase
- Cosmetic
Approach
- External- transconjunctival
- Endoscopic
|
Surgical Decompression |
Reduction in Proptosis (mm) |
|
Fat Removal Orbital decompression (FROD) |
2-3mm |
|
Bone removal Orbital decompression (BROD)
|
2-3 mm 4-5 mm 6-7 mm |
|
FROD + 3 wall decompression + lateral rim removal Balanced decompression – Medial + lateral wall (cosmetic) |
10-12 mm14 |
|
Bony Decompression |
Bones removed |
|
Lateral wall decompression |
Zygomatic bone – lateral to inferior orbital fissure Greater wing of the sphenoid bone Frontal bone – Lacrimal gland fossa |
|
Medial wall decompression |
Segments of the ethmoid bone Orbital contents extend into the ethmoid sinus |
|
Orbital floor decompression |
Posteromedial part of the floor - maxillary bone Expansion of orbital content into the maxillary sinus |
Care should be taken to avoid damage to the neurovascular bundle, the inferior oblique muscle along the floor, and more importantly the inferomedial orbital strut to prevent hypoglobus.
Complications:
|
Intraoperative |
Postoperative |
|
Bleeding |
Early – Infection Bleeding Retrobulbar haemorrhage Diminution of vision Diplopia Scarring |
|
Lacrimal/supraorbital/infraorbital nerve injury |
|
|
Hypoglobus Globe rupture Retinal detachment Optic nerve Injury |
|
|
Inferior oblique muscle injury |
Late – Asymmetry/ Supra, infraorbital nerve paresthesia |
Management of Sequelae/Fibrotic Phase
Any further intervention (sequential surgery) for cosmesis, diplopia, or strabismus is done after 6 months of stable disease with normal thyroid function tests. (Figure no. 5)
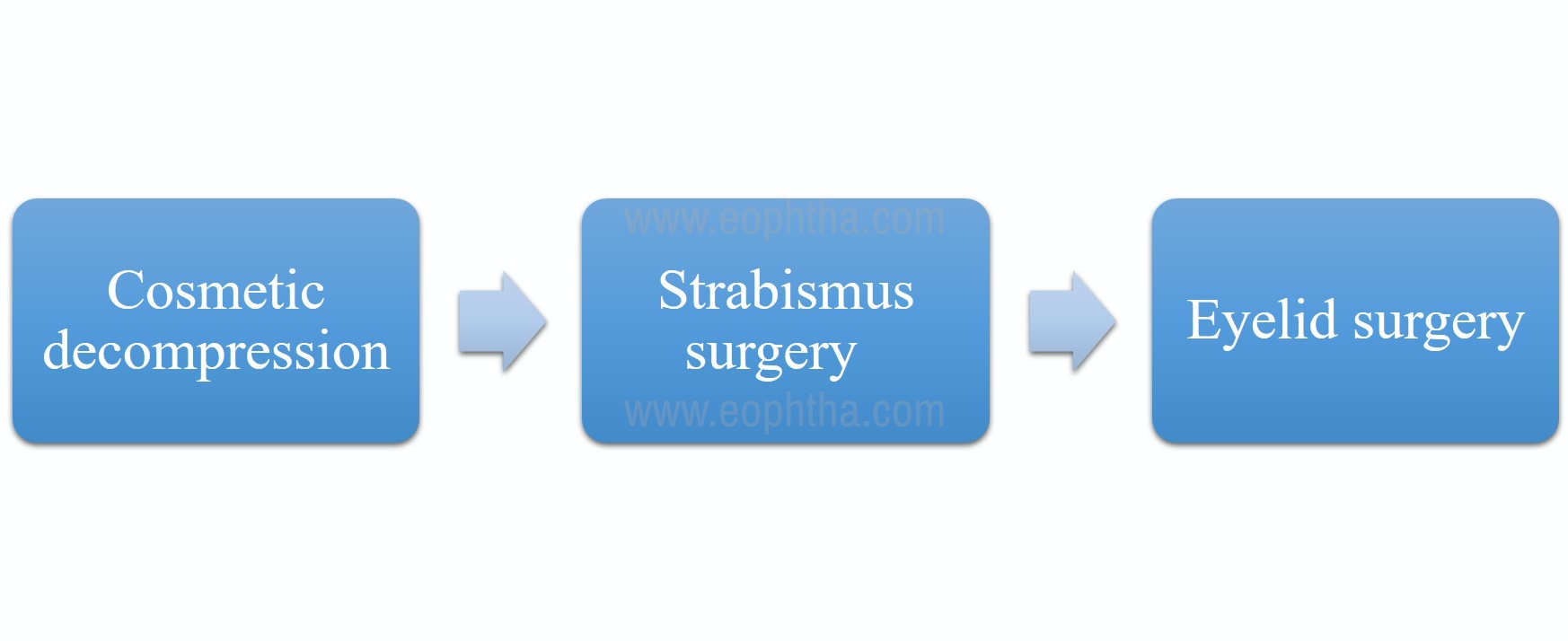
Figure 5: Chart depicting the preferred sequential order of surgical intervention in an inactive thyroid eye disease
|
Lid retraction |
|
|
Treatment |
|
|
Surgical correction of lid retraction |
|
|
Upper lid retraction |
|
|
Lower lid retraction |
|
- Blepharoplasty for orbital fat prolapse.
- Correction of associated lacrimal gland prolapse and ptosis.
References
1. Li Z, Cestari DM, Fortin E. Thyroid eye disease: what is new to know? Curr Opin Ophthalmol. 2018 Nov;29(6):528–34. 2. Bartley GB. The epidemiologic characteristics and clinical course of ophthalmopathy associated with autoimmune thyroid disease in Olmsted County, Minnesota. Trans Am Ophthalmol Soc. 1994; 92:477–588. 3. Lazarus JH. Epidemiology of Graves’ orbitopathy (GO) and relationship with thyroid disease. Best Pract Res Clin Endocrinol Metab. 2012 Jun;26(3):273–9. 4. Liaboe CA, Clark TJ, Shriver EM, Carter KD.Thyroid Eye Disease: An Introductory Tutorial and Overview of Disease.EyeRounds.org. posted November 18, 2016; Available from http://www.EyeRounds.org/tutorials/thyroid-eye-disease/ 5. Shan SJC, Douglas RS. The pathophysiology of thyroid eye disease. J Neuroophthalmol. 2014 Jun;34(2):177–85. 6. Krassas GE, Wiersinga W. Smoking and autoimmune thyroid disease: the plot thickens. Eur J Endocrinol. 2006 Jun;154(6):777–80. 7. Barrio-Barrio J, Sabater AL, Bonet-Farriol E, Velázquez-Villoria Á, Galofré JC. Graves’ Ophthalmopathy: VISA versus EUGOGO Classification, Assessment, and Management. Journal of Ophthalmology. 2015:1–16. 8. Haefliger I, von Arx G, Pimentel A-R. Pathophysiology of Intraocular Pressure Increase and Glaucoma Prevalence in Thyroid Eye Disease: A Mini-Review. Klin Monatsbl Augenheilkd. 2010 Apr;227(04):292–3. 9. Dolman PJ. Grading Severity and Activity in Thyroid Eye Disease: Ophthalmic Plastic and Reconstructive Surgery. 2018 Jul; 34-40. 10. Taylor PN, Zhang L, Lee RWJ, Muller I, Ezra DG, Dayan CM, et al. New insights into the pathogenesis and nonsurgical management of Graves orbitopathy. Nat Rev Endocrinol. 2020 Feb;16(2):104–16. 11. Gonçalves A, Gebrim E, Monteiro M. Imaging studies for diagnosing Graves’ orbitopathy and dysthyroid optic neuropathy. Clinics. 2012 Nov 7;67(11):1327–34. 12. Kizilca Ö, Öztek A, Kesimal U, Şenol U. Signs in Neuroradiology: A Pictorial Review. Korean J Radiol. 2017;18(6):992. 13. Kahaly GJ, Pitz S, Hommel G, Dittmar M. Randomized, single blind trial of intravenous versus oral steroid monotherapy in Graves’ orbitopathy. J Clin Endocrinol Metab. 2005 Sep;90(9):5234–40. 14. Ediriwickrema LS, Korn BS, Kikkawa DO. Orbital Decompression for Thyroid-Related Orbitopathy During the Quiescent Phase: Ophthalmic Plastic and Reconstructive Surgery. 2018 May;1. 15. Grisolia ABD, Couso RC, Matayoshi S, Douglas RS, Briceño CA. Non-surgical treatment for eyelid retraction in thyroid eye disease (TED). Br J Ophthalmol. 2017 Aug 9. 16. Ribeiro SFT, Shekhovtsova M, Duarte AF, Velasco Cruz AA. Graves Lower Eyelid Retraction: Ophthalmic Plastic and Reconstructive Surgery. 2016;32(3):161–9.


