Penetrating keratoplasty (PK) is a procedure in which full-thickness host corneal tissue is replaced with donor corneal tissue.
Objectives of PK maybe
- Optical-to establish a clear visual axis or to minimize refractive error in severely distorted corneas
- Tectonic-to provide structural integrity
- Therapeutic-to eliminate infection
- Occasionally for cosmoses
Complications of PK may be divided as intraoperative, early postoperative, and late postoperative.
Intra-operative complications:
Intra-operative complications may occur at each step of the surgery. These complications can be avoided with meticulous pre-operative planning and careful attention at each step.
1. Anaesthesia:
In the case of the retrobulbar or peri-bulbar block (1), globe perforation may occur. A meticulous pre-operative assessment could avoid this complication as eyes with a posterior staphyloma possess a higher risk of perforation.
A retrobulbar block may cause scleral perforation, orbital hemorrhage, optic nerve trauma, or orbital apex injury (2). Careful blocks, no massage, and preferable general anesthesia help in preventing such complications.
Retrobulbar hemorrhage may occur due to local anesthesia which can be medically managed with the hyperosmotic agent like mannitol 20%, administered as a rapid intravenous infusion of 1.5-2 g/kg over 30 min or IV acetazolamide 500 mg to decrease the pressure and/or IM/IV hydrocortisone 100 mg to decrease inflammation, edema and provide some neuroprotection to the optic nerve by stabilizing cell membranes. In patients with vision loss, proptosis, extraocular movement deficiencies, or any sign of elevated IOP, surgical intervention to evacuate the hemorrhage may be required.
An inadvertent intra-dural injection may cause CNS side effects like seizures, brainstem anesthesia or respiratory arrest.
The inadvertent intravenous injection may cause cardiac arrhythmias.
Peri-bulbar anesthesia results in less morbidity as compared to retrobulbar.
2. Fixation ring:
A fixation ring may be used to stabilize the globe in pediatric patients (due to their elastic sclera) or aphakic eyes. The fixation ring facilitates intra-operative manipulation and suturing and prevents scleral collapse.
Scleral perforation with a retinal hole may occur in rare cases while fixing the ring. Cryotherapy should be done at this site.
In case of ciliary body damage, bleeding may be seen at the angle. This is self-limiting and resolves in few minutes.
A fixation ring may also induce unwanted astigmatism (3).
3. Donor cornea damage:
It is preferable to prepare the donor cornea prior to the preparation of the recipient bed as the surgery may be aborted in case of complications in donor cornea preparation. The correct size of trephines must be used for the donor and the host cornea. The donor cornea trephine is 0.25-0.5mm more than the host cornea trephine and care must be taken to prevent interchange (4,5). In case this occurs, and the host has been trephined, the small button must be carefully sutured in place and leaks must be prevented. A hyperopic shift occurs due to smaller donor cornea which is compensated for by increasing the intraocular lens power by 2 or 3 D. The trabecular meshwork may be deformed due to tighter sutures and this may increase the risk of postoperative glaucoma (6). The graft can be replaced by an appropriate-sized graft at a later date in such cases.
Trephination should be done in one pressing stroke with a sharp trephine and the endothelial side facing up. This minimizes damage to the donor cornea. A dull trephine can cause a Descemet’s membrane detachment (DMD).
The prepared donor cornea should be neatly stored on a Teflon block with viscoelastic to prevent damage to it.
4. Host cornea dissection:
A paracentesis must be made and viscoelastic should be injected into the anterior chamber (AC) to prevent inadvertent iris and lens damage while trephining or using the scissors. Partial trephination also prevents such damage. In phakic patients, the pupil must be constricted preoperatively to prevent lens damage. It may be prudent to perform preoperative biometry in a patient who is planned for a phakic penetrating keratoplasty to account for anterior lens capsule damage that may require lens removal and intraocular lens implantation.
The host cornea must be trephined in the center and equidistant from the limbus on all sides. Care must be taken to prevent slippage of the trephine. Eccentric trephination can increase postoperative astigmatism. The trephine must also be aligned perpendicular to the corneal plane to prevent oblique entry into the anterior chamber. An oblique entry causes poor wound apposition, wound leak, and astigmatism (7,8).
After partial trephination of the host cornea, the dissection is completed using Castroviejo’s scissors. Some surgeons prefer a beveled cut with a posterior wound ledge facilitating a watertight closure while others cut the host cornea perfectly perpendicular to the corneal surface to minimize astigmatism. It must be confirmed that the scissors have entered the AC and full-thickness dissection is complete as the Descemet’s membrane (DM) may be left behind and the scissors may remain above it (9). This may be done by inspecting the iris with forceps or sponge. A retained DM may cause pseudo-AC and endothelial damage post-operatively. Nd: YAG laser may be used to create a central opening in the retained membrane as it may opacify with time (10).
5. Iris trauma:
Injury to the iris in form of iridodialysis, holes in the iris, or iatrogenic aniridia can occur due to incomplete, eccentric, or over aggressive trephination, which is more common in thin or perforated corneas. Making a paracentesis and injecting viscoelastic into the AC prior to trephination may protect the intraocular structures. In perforated corneas, sealing the wound with cyanoacrylate glue prior to trephination and formation of the AC with viscoelastic helps to reduce the risk of iris trauma.
6.Bleeding:
Iris bleeding can be controlled with viscoelastic, fine-tipped underwater diathermy, topical application of 1:10000 epinephrine or 1:100 thrombin on a soaked cellulose sponge, and preoperative brimonidine drop.
7. Intra-ocular lens (IOL) explantation:
If a closed loop anterior chamber IOL is present, it may be replaced with a sulcus or scleral fixated posterior chamber IOL or an iris claw lens or a flexible open loop anterior chamber IOL (15-18).
8. Positive vitreous pressure:
Auto-expulsion of the crystalline lens can occur in the case of positive vitreous pressure or when trephination is done aggressively. Due to the removal of the cornea, there is a loss of barrier to vitreous expansion. This may cause shallowing of AC and protrusion of the lens, leading to increased risk of complications.
Positive vitreous pressure can be reduced by mechanical, medical, and surgical methods. Pre-operative assessment and administration of injection mannitol, constriction of the pupil with pilocarpine, mechanical pressure on the globe (to be avoided in case of perforation) either manually (19 20) or with a Honan balloon (21) or with a super-pinky can help reduce intra-operative positive vitreous pressure. An additional measure is to position the patient in reverse Trendelenburg, decreasing the orbital venous pressure.
Positive vitreous pressure might be encountered intra-operatively due to the pressure transmitted by the lid speculum. Lifting or repositioning the speculum or providing anterior traction on the globe with the fixating suture may help to avoid such inadvertent complications. The role of limited vitrectomy or vitreous tap is controversial.
9. Supra-choroidal hemorrhage:
Supra-choroidal or expulsive hemorrhage can occur if vitreous up-thrust is thereafter removing the recipient cornea. It is the most dreadful intra-operative complication. Incidence is 0.45% to 1.08%. The most common site of bleeding is the long or short posterior ciliary artery. It occurs due to sudden globe decompression in the ‘open sky’ phase of the procedure. The intra-ocular contents extrude due to loss of resistance (22, 23).
Risk factors predisposing to supra-choroidal hemorrhage are (24):
- A long period of hypotony
- Prior supra-choroidal hemorrhage
- Raised episcleral venous pressure
- Previous vitrectomy
- Anticoagulant therapy
- Anesthetics
- Old age
- Glaucoma
- Tachycardia, Systemic hypertension, Arteriosclerosis
Signs of impending supra-choroidal hemorrhage are the appearance of a dark choroidal detachment, forward movement of intra-ocular content, and complaints of acute severe ocular pain.
Management is with mechanical tamponade and wound closure with the graft or temporary keratoprosthesis. The role of sclerotomies to drain the blood is controversial. Some surgeons believe that it decreases forward pressure on the vitreous whereas others believe that the accumulated blood serves as tamponade which is lost due to drainage leading to worsening of the hemorrhage.
10. Suture:
Suturing may be done with interrupted sutures or a continuous suture. The continuous suture has fear of breakage for which a double-armed suture might be taken. Alternatively, a new suture may be spliced to the broken suture and the suturing may be completed. The sutures must be taken at about 90% of the depth of the cornea. This prevents the suture from forming a direct tract for the spread of infection into the AC and reduces the chances of endophthalmitis.
Burial of the knots is essential to prevent foreign body sensations. The knots may also act as a nidus for infection if not buried. Tight sutures cause the Donut effect (central flattening with surrounding steepening) and stress lines. Loose sutures cause wound gape and central steepening with surrounding flattening.
Watertight closure must be ensured. Additional interrupted sutures may be taken to prevent wound leak.
Postoperative complications:
The incidence of postoperative complications is
- Post-op 1 month: 50%
- Post-op 3 months: 30%
- Post-op 1 year: 10%
- >1year post op: 10%
A careful post-operative examination is essential for the prevention and early detection of complications. On the first post-operative day, a history of any complaints must be taken. Visual acuity must be determined and slit lamp examination must be done. The patient should be followed up frequently until complete corneal epithelization occurs. Once this occurs, the patient must be seen every 1 or 2 weeks for the first month and then every 2–4 weeks for the next 3 months. The patient should be asked to report immediately in case of
- Increased pain
- Increased redness
- Decreased vision
The newly implanted graft is anesthetic hence patching may be done till epithelization occurs in 1-3 days usually. The eye is protected with eyewear during the day and an eye shield at night for 2-3 months.
Early Postoperative complications (within 2-3 weeks):
Wound leak:
It can be avoided by advanced surgical techniques and meticulous wound apposition at the end of the surgery. Prompt re-suturing should be attempted if non-surgical methods like patching or bandage contact lens fail to seal the leakage.
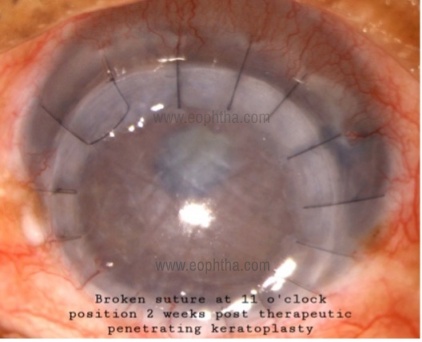
Suture problems:
Patients with broken sutures or exposed knots in the early postoperative period before the wound healing should undergo immediate suture repair as there is a risk of the development of graft over-ride or loss of the anterior chamber. Suture infiltrates should be carefully distinguished as infective (solitary, on graft or host side with epithelial defect and/or AC reaction) or inflammatory (multiple, usually on host side with no epithelial defect or AC reaction) so as to manage them accordingly with appropriate anti-microbials or corticosteroids respectively.
Epithelial defect:
Damage to donor epithelium can occur before harvesting, during storage, or intra-operatively. Stable epithelium over the transplanted cornea is very crucial for the successful outcome of the surgery. Delayed or incomplete epithelialization of the transplanted donor cornea can lead to potentially disastrous results. Epithelial defects may lead to infections, so it is always prompted to treat them aggressively. A stable epithelial surface is the first step of a successful transplant. The average time for complete epithelialization is 4-6 days. A persistent epithelial defect is one without improvement or healing for more than 10-14 days. Incomplete epithelialization may lead to Bowman membrane damage and stromal melting over time that leads to graft haze and scarring. In addition, it may also cause infections. It is important to prevent risk factors to reduce the incidence of this complication.
During surgery, all the care must be taken to avoid epithelial damage. Lid problems, ocular surface problems should be taken care of to avoid delayed epithelial healing. Post-operatively preservative-free lubricant drops and/or ointments or autologous serum should be prescribed. Any topical epithelial toxic medication should be avoided or replaced by less toxic medication. Use of least number of drugs in minimum dosing should be advocated. Pressure patching can be done for 24-48 hours. Sutures with exposed ends should be removed or replaced. Topical steroids should be used in minimum dosing as it might retard epithelial healing. A soft bandage contact lens can be placed or a tarsorrhaphy can be performed in resistant non-healing defects. In the case of melts, a conjunctival flap may be useful. Persistent non-healing defects may also be due to herpes infections in which case appropriate anti-virals must be used.
Shallow/flat AC:
This is usually avoidable with a watertight wound but if present, should be managed as soon as possible to avoid synechiae formation which may result in irreversible endothelial cell loss and consequently early graft failure. AC can be reformed with air, viscoelastic, or balanced salt solution.
Hyphema:
It may be due to trauma to iris vessels or bleeding from the angle. This could be avoided with a tamponade using viscoelastic or sponges soaked with epinephrine 1:1000. Persistent hyphema should be immediately evacuated as it increases IOP.
Raised intraocular pressure (IOP):
Control of post-operative raised IOP is vital as elevated pressure may result in endothelial cell damage (25).
Other causes of elevated IOP in the early postoperative period include retained viscoelastic, intraocular inflammation, trabeculitis, anterior synechiae causing angle closure and pupillary block.
Anterior uveitis:
It depends on the degree of iris manipulation and the condition of the eye before surgery. This could be minimized by thorough removal of inflammatory material from the AC if any and gentle manipulation of the iris during surgery. The aggressive control of postoperative inflammation is also essential to prevent PAS formation. Usually, the uveitis is resolved with the use of cycloplegic and corticosteroid drugs, but the latter should be used with caution in infective cases.
Pupillary block:
Due to the pupillary block, aqueous is obstructed in its movement from posterior to the anterior chamber, the iris bulges anteriorly resulting in shallowing of the AC and the angle is then obstructed. It results in raised IOP. It is usually seen if the peripheral iridectomy is not performed during the surgery. So, the iridectomy is almost always performed in the case of therapeutic keratoplasty to avoid the pupillary block. In the postoperative period, vigorous dilation of the pupil may relieve the block.
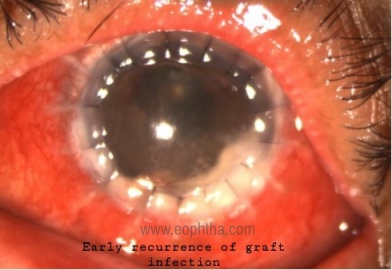
Graft infection and Endophthalmitis:
Graft infection and endophthalmitis can occur both immediately after and late in the course after corneal transplantation. The incidence of endophthalmitis is between 0.1% and 2%. The prognosis for endophthalmitis after corneal transplantation has been very poor. Infections in the immediate post-keratoplasty period are mostly due to contaminated donor button, intraoperative contamination, or recurrence of host infection. With improved eye banking techniques, the risk of donor transmission of infectious disease has been decreased.
Risk factors for endophthalmitis:
- Keratitis sicca
- Graft infection/failure
- Blepharitis
- Previous herpetic keratitis
- Acne rosacea
- Ocular adnexa and lid abnormalities, Trichiasis
- Cicatricial conjunctival disease (ocular pemphigoid, alkali burns, and Stevens-Johnson syndrome)
- Trachoma
Intraoperative factors:
- Contaminated instruments or solutions
- Extensive tissue manipulation
- Contaminated donor tissue or storage media
Postoperative factors:
- Epithelial defects or severe punctate keratopathy
- Wound dehiscence
- Exposed or loose sutures
- Corticosteroid use
- Soft contact lens use
Cultures of the donor rim at the time of keratoplasty may help guide the therapeutic regimen in the immediate post-operative period. The strict sterile operative technique decreases the chances of perioperative infection. The recurrence of host infections is dependent on the location of active infection (highest risk with the involvement of the limbus), postoperative medications (corticosteroids), and adjunctive surgical techniques used (such as cryotherapy). The recurrence of herpes simplex keratitis can be influenced by antiviral prophylaxis.
Primary graft failure:
It is irreversible graft edema in the immediate postoperative period, which may be due to inadequate endothelial cell function. This is a rare complication. It is unresponsive to steroids and hypertonic agents.
Causes:
- Unhealthy donor endothelium,
- Inadequate tissue preservation or
- Surgical trauma
Prevention:
- Donor endothelial count >2000cells/mm²
- Storage time <7 days
- Preoperative IOP lowering
- Intra-operative viscoelastic while suturing
- Proper handling
Treatment is to observe for 4-6 weeks and re-grafting if it persists beyond that.
It is the irreversible edema of the corneal graft in the immediate post-operative period. It is due to inadequate endothelial cell function, inadequate tissue preservation, or surgical trauma to the donor endothelium. Long-term graft survival can be optimized by selecting the donor cornea having at least 2000 endothelial cells/mm2 with a storage time less than 7 days and avoiding iatrogenic trauma to the endothelium during surgery.
Late Postoperative complications
Wound Dehiscence:
Causes for wound dehiscence may be:
- Thin, necrotic recipient bed
- Trauma
- Uncontrolled glaucoma (IOP >40mmHg)
Treatment is by surgical repair.
Filamentary keratitis:
It occurs in 25% of patients post PK. Degenerated epithelial cells and mucus (Rose Bengal stain) are seen. They adhere to the corneal surface and move with blinking. The patient presents with pain and foreign body sensation.
Treatment is with lubricants, bandage contact lens or N-acetylcysteine 10-20% drops. Mechanical removal of filaments may be done.
Kaye dots:
Discrete white dots are seen in donor epithelium. These are epithelial cells in stages of degeneration and do not stain with fluorescein. Patient is asymptomatic. They appear 6.5 weeks post operatively and disappear around 30 days after suture removal.
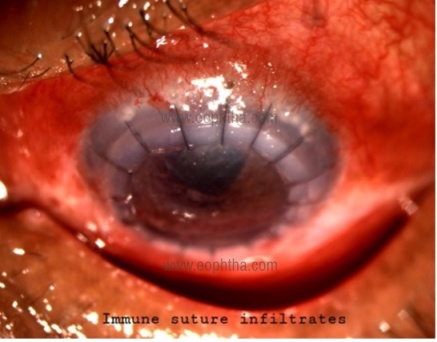
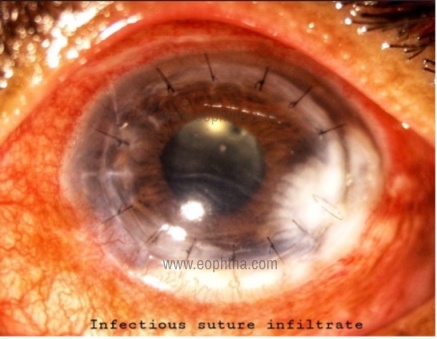
Immune vs Infectious Infiltrates:
|
Immune suture infiltrates |
Infectious suture infiltrates |
|
Multiple |
Solitary |
|
Host cornea |
Host and donor cornea |
|
No epithelial defect |
Epithelial defect |
|
No AC reaction |
AC reaction |
|
Rx with steroids |
Rx with anti-microbials |
Anterior synechiae:
Anterior synechiae may develop at the peripheral cornea, in the corneal wound itself, or may extend from one to the other. Anterior synechiae may form later as the result of a wound leak with a flat chamber, a persistent shallow chamber, or from trauma with partial or complete iris prolapse. The pupil should be aggressively dilated with a phenylephrine hydrochloride cyclopentolate series and, if this is not effective, constricted with pilocarpine. If unsuccessful and if the IOP is controlled, it is best left alone.
Graft rejection:
|
Acute rejection |
Chronic Rejection |
|
Few weeks post-transplantation |
Months to years post-transplantation |
|
Cell-mediated |
Cell-mediated + humoral mediated |
|
Macrophages and lymphocytes |
Cells and newly formed antibodies |
Risk factors:
- Young age (<40yrs)
- >2 quadrants of stromal vascularization
- Vascularization to graft margin
- Large size graft
- Prior graft failure (8% in 1st graft to 40% in 2nd or more graft)
- Prior glaucoma surgery
- Anterior synechiae
- Preexisting inflammation
Symptoms are blurring of vision, redness, photophobia. Graft rejections are detected 70% by patient symptoms and 30% by clinical examination hence a thorough history taking and clinical examination is essential.
|
Rejection |
Treatment |
|
Epithelial rejection (10%)
|
Topical steroids |
|
Subepithelial infiltrates (10-15%) Grayish white infiltrates 0.2-0.5mm in donor cornea |
Topical steroids |
|
Stromal rejection (1–24%) Stromal haze spreading from periphery to center Associated with endothelial rejection |
Topical steroids Laser photocoagulation of stromal vessels+ Avastin |
|
Endothelial rejection (8-37%)
|
Topical steroids + Subconjunctival steroid/ oral prednisone/ i.v. methyl prednisolone/ dexamethasone ointment at night Cycloplegics Intracameral/ intravitreal triamcinolone (resistant cases) |
Epithelial downgrowth and Fibrous ingrowth:
|
Epithelial downgrowth |
Fibrous ingrowth |
|
Corneal/ Conjunctival epithelial cells |
Subepithelial connective tissue, stromal fibroblasts |
|
Scalloped, smooth, thickened edge with refractile membrane Avascular |
Translucent membrane with frayed irregular edge May be vascular |
|
Argon laser photocoagulation of iris Specular microscopy Confocal microscopy |
Confocal microscopy |
|
Rapidly progressive |
Slowly progressive/ Stable |
|
Complete removal of membrane Glaucoma control |
Anti-inflammatory Glaucoma control |
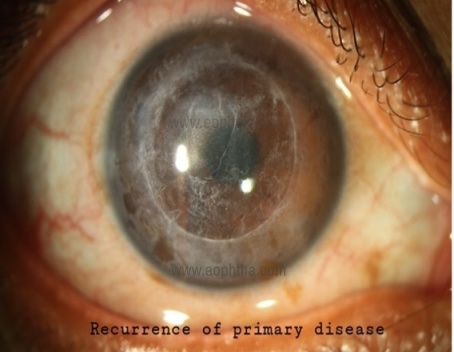
Recurrence of the underlying disease:
This occurs due to the migration of recipient keratocytes into graft stroma. Treatment depends on the nature of the disease and may require medical management, regrafting or phototherapeutic keratectomy.
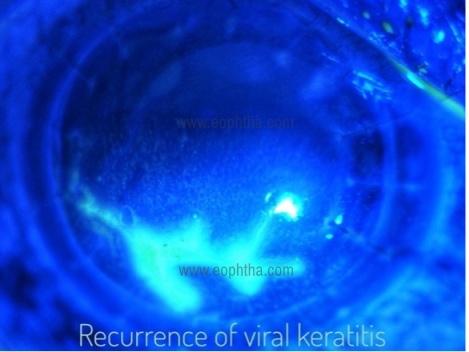
Infectious keratitis:
The incidence of graft infections post penetrating keratoplasty is 11.9% in developing countries and 1.7-7.4% in developed countries. Most of these occur in the 1st year post-surgery. Higher incidence in developing countries may be related to indications, non-compliance of post-operative management schedule for socioeconomic reasons, and quality of healthcare.
Causes of infectious keratitis:
- Loose or broken suture
- Contaminated donor tissue
- Epithelial defects
- Contact lenses
- Trichiasis
- Dry eye
- Lid abnormalities
Indication of keratoplasty:
- Certain indications such as bullous keratopathy have higher chances of post-operative infection than conditions like corneal dystrophies
- Contamination of donor tissue:This may be pre-existing as in septicemia or may occur during storage and processing.
- Pre-existing infections:Ocular surface infections such as dacryocystitis, blepharitis, and meibomitis may predispose to infectious keratitis.
- Recurrence of host infection:It is usually seen due to incomplete excision of the infected host tissue. In case of transplantation post herpes simplex infection, recurrences occur in 23% of cases.
- Dry eye and ocular surface disorders:Altered tear film and dellen increase adherence of microbes to the transplanted cornea.
- Persistent epithelial defect:The epithelium serves as a mechanical barrier to prevent the entry of microbes into the cornea. The presence of a persistent epithelial defect compromises this barrier.
- Sutures:Loose or broken sutures act as a nidus for the deposition of mucin and microbes. This is the most important predisposing factor for infectious keratitis (14-60%) cases.
Retrocorneal membrane formation:
Retrocorneal membranes may include:
- Epithelial downgrowth- the epithelial invasion into the anterior chamber. It is important to remove the invading epithelium.
- Fibrous ingrowth- fibrous proliferation and invasion of the tissues surrounding the surgical site into the anterior chamber. It is managed medically by controlling inflammation or by viscodissection of membrane out of the visual axis.
- Inflammatory membranes- responds well to anti-inflammatory treatment.
- Retained host’s Descemet’s membrane- due to incomplete removal, particularly in oedematous host cornea. Surgical excision or Nd-YAG laser opening in the membrane is recommended if the potential best corrected visual acuity is limited. It can happen if the membrane begins to opacify, the graft viability is compromised by contact with the retained tissueor there is increased endothelial cell loss.
- Descemet’s detachment of the graft- may require air descemetopexy.
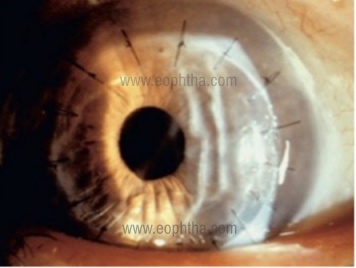
Glaucoma:
It is one of the most common causes of irreversible visual loss and the second leading cause for graft failure. Post-keratoplasty glaucoma is defined as an elevated IOP greater than 21 mmHg, with or without associated visual field loss or optic nerve head changes.
Causes:
- Pre-existing- Primary or secondary
- Secondary angle-closure
- Trabecular meshwork collapse (aphakia)
- Post-operative- Pupillary block, iritis, hemorrhage, lens/OVD induced, steroid response, malignant.
Cataract:
Post-operative steroid use and intraoperative iris manipulation may lead to cataract formation after penetrating keratoplasty. Cataract extraction with intraocular lens implantation gives better visual rehabilitation in such cases with proper surgical technique and IOL power calculation.
Infectious crystalline keratopathy:
It is a rare infection of the cornea that occurs due to long-term steroid therapy or an immunocompromised state. There is presence of grey-white branching stromal opacities in the cornea. Although rare it is most commonly seen after penetrating keratoplasty. The organisms secrete a biofilm in response to the nutrient deficient environment where it is growing, and this leads to a scant inflammatory response. It has an indolent clinical course. The epithelium is typically intact and there is the stromal deposition of crystalline or needle-like white-grey infiltrates. It is difficult to treat and treatment involves tapering of steroids to alleviate the immunocompromised state. Topical linezolid has shown variable results in recent studies.
Astigmatism:
Average postoperative induced astigmatism is 4-6 D. The 6 o’clock interrupted suture is most crucial for postoperative astigmatism. Post-operative astigmatism is assessed by corneal topography. Suture removal is done at 3-6 months post-operative if astigmatism >3 D.
|
Host factors |
Donor factors |
Surgical factors |
|
>8.75mm: less astigmatism <7mm: more astigmatism
|
|
- Residual astigmatism may be treated with spectacles/ RGP lenses/ Scleral lenses. Refractive surgery post penetrating keratoplasty is also another option for the treatment of post-keratoplasty astigmatism. 8-20% of astigmatism is usually not neutralized with glasses/ contact lenses.
- Relaxing incisions alone may be given. Arcuate keratotomy ≥ 1 arc-shaped relaxing incisions in stroma or at graft host junction. Stabilization occurs in 3-6 weeks.
- Relaxing incisions + compression sutures can be done.
- Wedge resections may be considered for Astigmatism >8-10D.
- Laser correction is done by PRK with MMC or LASIK. PRK with MMC preferred over LASIK to avoid suction ring and flap-related complications.
Urrets Zavalia syndrome:
Permanent fixed dilated pupil is seen in this condition. Multiple posterior synechiae and iris atrophy occurs.
This may be caused by iris ischemia. Treatment is to reduce intraocular pressure and treat the glare associated with it.
Graft failure:
Causes:
- Graft rejection
- Chronic inflammation
- Endothelial decompensation
- Recurrence of infection
It requires regraft keratoplasty- penetrating or lamellar.
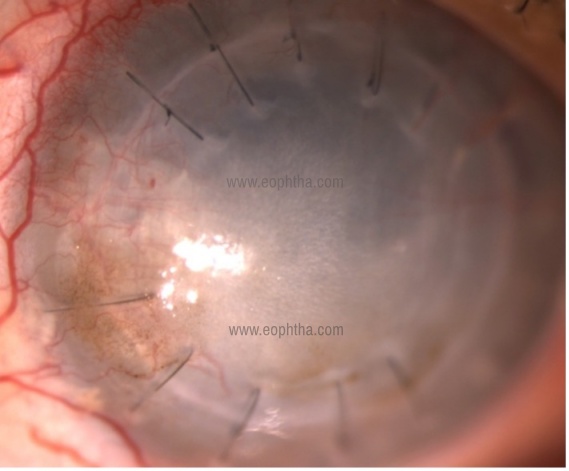
Phthisis bulbi:
Severe inflammation can cause alteration and disorganization of intraocular structures and atrophy.
References:
1. Muraine M, et al. Peribulbar anaesthedia during keratoplasty: a prospective study of 100 cases. Br J Ophthalmol 1999; 83: 104-109 2. Edge R, Navon S. Scleral perforation during retrobulbar anaesthesia: risk factors and outcome in 50,000 consecutive injections. J Cataract and refract surg 1999; 25(9): 1237-1244 3. Rudd JC, et al. Introduction of corneal astigmatism through placement of a scleral fixation ring in eye bank eyes. Cornea 2001; 20(8): 864-865 4. Heidemann DG, et al. Oversized donor grafts in penetrating keratoplasty. A randomized trial. Arch Ophthalmol 1985; 103(12): 1807-1811 5. Olsen RJ. Variation in corneal graft size related to trephine technique. Arch Ophthalmol 1979; 97(7): 1323-1325 6. Olson RJ, Kaufman HE. A mathematical description of causative factors and prevention of elevated intraocular pressure after keratoplasty. Invest Ophthalmol Vis Sci 1977; 16(12): 1085-1092 7. Chern KC, et al. Small diameter, round, eccentric penetrating keratoplasties and corneal topographic correlation. Ophthalmology 1997; 104(4): 643-647 8. Riedel T, et al. Visual acuity and astigmatism after eccentric penetrating keratoplasty- a retrospective study on 117 patients. Klin Monatsbl Augenheilkd 2002; 219(1-2): 40-45 9. Lifshitz T, Oshry T, Rosenthal G. Retrocorneal membrane after penetrating keratoplasty. Ophthalmic Surg Lasers 2001; 32(2): 159-161 10. Masket S, Tennen DG. Neodymium: YAG laser optical opening for retained Descemet’s membrane after penetrating keratoplasty. J Cataract Refract Surg 1996; 22(1): 139-141 11. Volker-Dieben HJ, Regensburg H, Kruit PJ. A double blind, randomized study of Healon GV compared with Healon in penetrating keratoplasty. Cornea 1994; 13(5): 414–417.Chapter 51: Intraoperative complications of penetrating keratoplasty 457 12. Gruber PF, Schipper I, Kern R. Use of Healon for corneal trephination in penetrating keratoplasty. Ophthalmic Surg 1984; 15(9): 773. 13. Hirst LW, De Juan E Jr. Sodium hyaluronate and tissue adhesive in treating corneal perforations. Ophthalmology 1982; 89(11): 1250–1253. 14. Maguen E, Nesburn AB, Macy JI. Combined use of sodium hyaluronate and tissue adhesive in penetrating keratoplasty of corneal perforation. Ophthalmic Surg 1984; 15(1): 55–57. 15. Kornmehl EW, et al. Penetrating keratoplasty for pseudophakic bullous keratopathy associated with closed loop anterior chamber intraocular lenses. Ophthalmology 1990; 97(4): 407-412 16. Lois N, et al. Long-term graft survival in patients with flexible open-loop anterior chamber intraocular lenses. Cornea 1997; 16(4): 387-392 17. Rijneveld WJ, et al. Iris claw lens: anterior and posterior iris surface fixation in the absence of capsular support during penetrating keratoplasty. J Refract Corneal Surg 1994; 10(1): 14-19 18. Kirsch RE, Steinman W. Digital pressure, an important safeguard in cataract surgery. Arch Ophthalmol 1955; 54(5): 697-703 19. Kirsch RE. Further studies on the use of digital pressure in cataract surgery. Arch Ophthalmol 1957; 58(5): 641-646 20. Quist LH, Stapleton SS, McPherson SD Jr. Preoperative use of the Honan intraocular pressure reducer. Am J Ophthalmol 1983; 95(4): 536-538 21. Purcell JJ Jr, et al. Expulsive haemorrhage in penetrating keratoplasty. Ophthalmology 1982; 89(1): 41-43 22. Taylor DM. Expulsive haemorrhage, Am J Ophthalmol 1974; 78(6): 961-966 23. Manschot WA. The pathology of expulsive haemorrhage. Am J Ophthalmol 1955; 40(1): 1524 24. Rotkis WM, Chandler JW, Forstot SL. Filamentary keratitis following penetrating keratoplasty. Ophthalmology 1982; 89: 946–949 25. Guss RB, Koenig S, Dela Pena W, et al. Endophthalmitis after penetrating keratoplasty. Am J Ophthalmol 1983; 95: 651–658.

1.jpg)