Introduction
Polypoidal Choroidal Vasculopathy (PCV), first described by Yannuzzi et al1 in 1982, is a distinct clinical entity characterized by persistent recurrent serous leakage and hemorrhage in the macula, and is seen in the elderly population. The disorder was poorly understood earlier and had been described by various authors as recurrent pigment epithelial detachment (PED) and posterior uveal bleeding syndrome.2.5 The primary abnormality, as described by Yannuzzi et al,1, 2, 6involves the choroidal circulation, and the characteristic lesion is an inner choroidal vascular network of vessels ending in an aneurysmal bulge visible clinically as a reddish-orange spheroidal polyp like structure. More recently with improvement in choroidal imaging using Enhanced Depth Imaging and Swept-Source optical coherence tomography (EDI-OCT and SS-OCT), PCV is regarded as a part of the Pachychoroid spectrum of diseases and a variant of Type I choroidal neovascularization.7, 8, 9
Epidemiology
The age of presentation of PCV is earlier than that for Age-related macular degeneration (AMD).5PCV has been documented to occur most frequently between 60 to 72years, although there have been case reports ranging from 20 years to 80 years.10In Indian population, the mean age noted was 61.06years (41-80).11PCV is seen more commonly in pigmented races i.e. Asians and African Americans compared to Caucasians.10 The prevalence of PCV amongst patients diagnosed with neovascular age-related macular degeneration is as high as 24.5% to 54.7% in Chinese12 and Japanese13 population respectively, 49% in the Taiwanese14 and 24.6% in the Korean15 populations compared to 4% to 9.8%in Caucasians.16, 17, 18Interestingly, PCV is seen more commonly in males in Asian population, having a M: F ratio of 1.4:1 in the Indian population, while in Caucasians, it is more common in females (75%).10, 11PCV is a more commonly unilateral disease with bilaterality of around 21 to 55% in Caucasians, less than 20% in Asians and around 17.8% in Indian population.10
Genetics and Biomarkers
The genetic association analysis have shown two major age-related macular degeneration (AMD) susceptibility loci, namely complement factor H (CFH) gene on chromosome 1q32 and 2 tightly linked genes age-related maculopathy susceptibility 2 (ARMS2) and high-temperature requirement factor A1 (HTRA1) on chromosome 10q26, to influence the risk of PCV.19 And of these, the single-nucleotide polymorphism (SNP) in the ARMS2 /HTRA1 locus, rs10490924 (A69S) and rs11200638, are significantly associated with PCV.The variation in rs10490924 shows a significant difference between PCV and wet AMD (n=5, OR=0.66, p<0.00001).20The risk genotypes of rs10490924 have been noted to be associated with larger lesion size, greater chance of vitreous hemorrhage, and poor therapeutic response in PCV.20
Histopathology and Immunohistochemistry
On histopathological evaluation, PCV demonstrates hyalinization of choroidal vessels, replacement of smooth muscle component with pseudocollagenous tissue along with massive exudation of fibrin and blood. Immunohistochemical studies have revealed CD68 positive cells along the hyalinised vessels, CD34 staining revealed discontinuity of endothelium and absence of smooth muscle actin positive cells.21The vascular endothelial cells in PCV are negative for vascular endothelial growth factor (VEGF), which may explain the reason for poor response to anti-VEGF therapy.22 It has also been noted that vortex veins are engorged in cases with PCV.15Hence the terminal polyps may represent the aneurysms that result due to chronic choroidal venous hypertension.23
Clinical Features
The classical clinical findings of PCV is the presence of reddish-orange subretinal nodules (fig.1).19, 24They can be small, medium or large in size. . Apart from polyps, the clinical features more commonly are seen include varying degrees of serous or serosanguinous pigment epithelial detachments (PED’s), subretinal hemorrhage, lipid deposition as well as neurosensory retinal detachment in the peripapillary or macular region.19
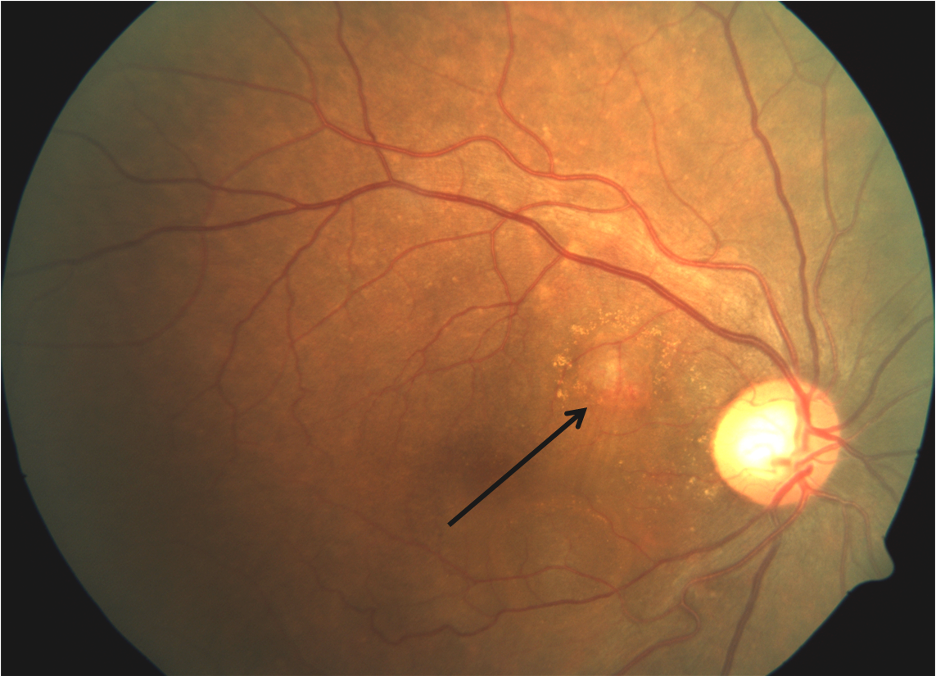
Figure 1: Colour fundus photograph of a patient with PCV demonstrating classic reddish-orange subretinal nodule (Black arrow)
Clinically, PCV can be classified as follows (fig.2):
- Exudative:It is characterized by the presence of lipid exudation along with serous macular detachment, intra-retinal fluid and PED (fig2A).
- Haemorrhagic:It is characterized by the presence of Sub-retinal or Sub-RPE hemorrhage which may be associated with hemorrhagic PED (fig.2B)
- Mixed: Presence of features of both exudative and hemorrhagic variety (fig.2C)
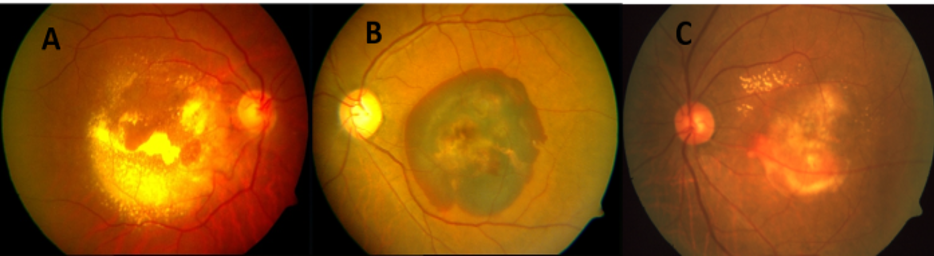
Figure 2: Colour fundus photograph showing clinical types of PCV: A) Exudative; B) Haemorrhagic; C) Mixed
Imaging
Indocyanine green angiography (ICGA):
Rationale in PCV
Polypoidal choroidal vasculopathy (PCV) by virtue of primarily being an abnormality of choroidal circulation is imaged and characterized better by ICG Angiography than Fluorescein Angiography (FFA).25 While the retinal pigment epithelium (RPE) and choroidal absorb 59% to 75% of the blue-green light in FFA, only 21% to 38% of the near-infrared light employed in ICGA is absorbed.26, 27 Additionally, ICG being largely protein-bound (98%), tends to remain within the lumen of the fenestrated choroidal vasculature. This permits greater delineation of the choroidal anatomy unlike fluorescein, which extravasates into the interstitium to obfuscate the choroidal details. Hence ICGA is considered to be the current gold standard for the detection and evaluation of PCV.
When is ICG Angiography indicated?
It should be performed when routine ophthalmologic examination indicates a serosanguinous maculopathy with one of the following features: Clinically visible orange-reddish nodules, spontaneous sub-retinal hemorrhages, notched or hemorrhagic pigment epithelial detachment and a lack of response to anti-VEGF therapy.
What are the classic features in ICG Angiography?
The characteristic features in ICG angiography is early nodular hyperfluorescence arising from the choroidal circulation noted within the first six minutes of the dye injection (Fig.3).28, 29Theymay be solitary or multiple and arranged in strings or cluster.
Additional ICGA features include:
- Hypofluorescent halo around the hyperfluorescent nodule
- Abnormal vascular channels terminating in the polyps (Fig.3). In many cases, a feeder vessel can be appreciated which fills the entire abnormal vascular network, which is called the branching vascular network (BVN) (Fig.4)
- Pulsation filling of the polyps (Video ICGA)
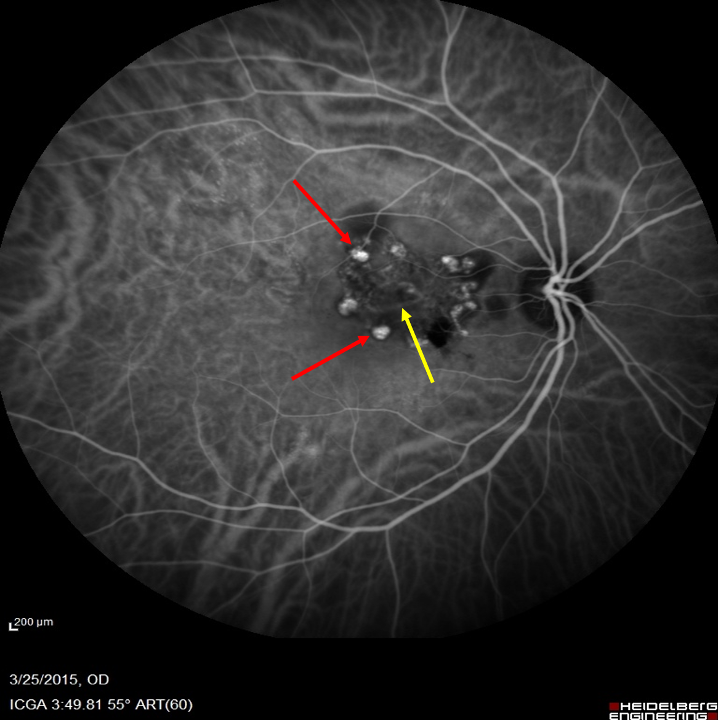
Figure 3: ICGA of a patient demonstrating a network of fine vascular channels (Yellow arrow) terminating in the nodular hyperfluorescent polyps (Red arrow)
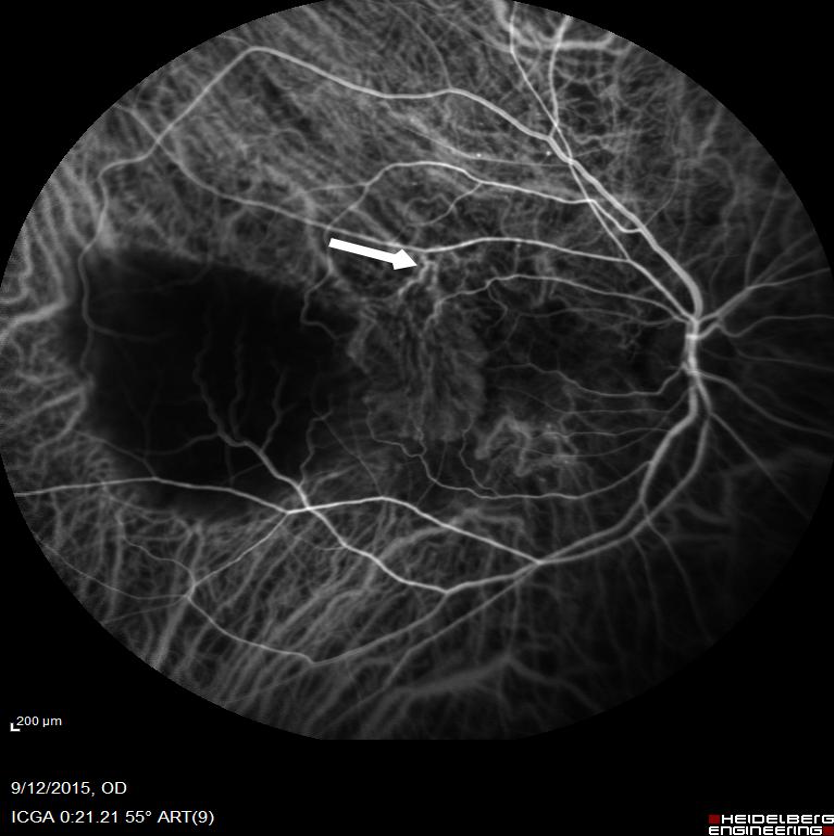
Figure 4: ICGA of a patient demonstrating the feeder vessel (White arrow) filling the network of fine vascular channels, also known as branching vascular network (BVN)
Based on ICGA polyps can be classified (Fig.5) as:
- Subfoveal polyps: Below the fovea (Fig.5A)
- Juxtafoveal Polyps: Located within 1-199 microns from the centre of the fovea (Fig.5B)
- Extrafoveal polys: Polyps located 200 microns or more from the centre of the fovea (Fig.5C)
- Peri-papillary polyps: Located with one disc diameter from the margins of the disc (Fig. 5D)
- Peripheral polyps: Located outside the arcade (Fig. 5E)
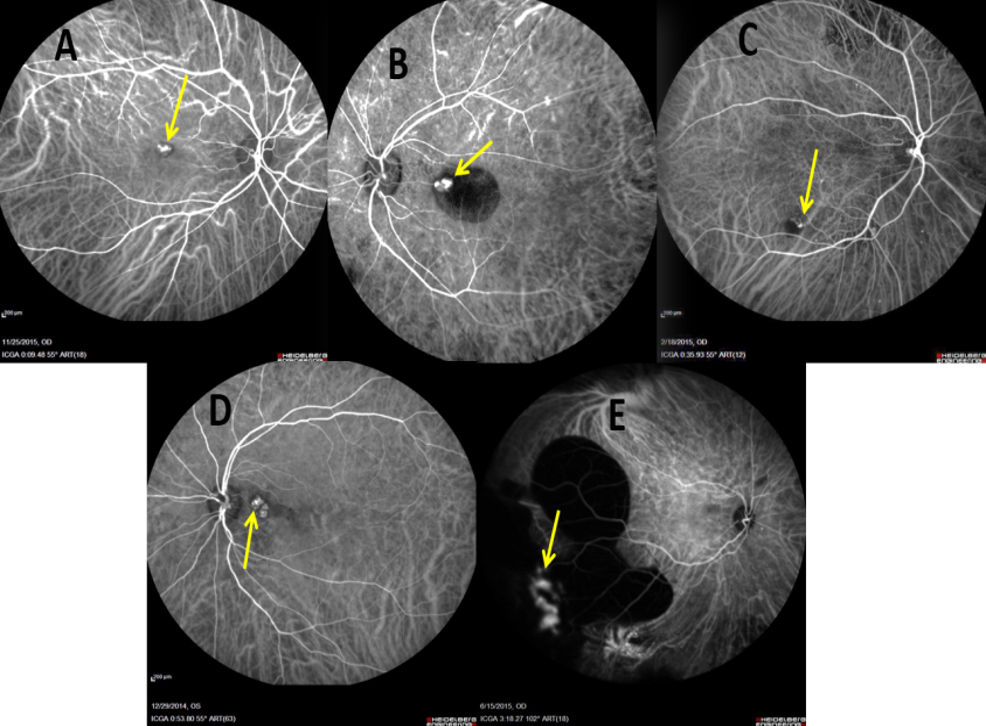
Figure 5: ICGA images showing various topographic locations of polyps (Yellow arrow): A) Subfoveal; B) Juxtafoveal; 3) Extrafoveal; 4) Peri-papillary; 5) Peripheral
Fundus Fluorescein angiography (FFA):
FFA does not have any characteristic feature in PCV and the features mimic than of occult choroidal neovascular membrane.30 Areas of hyperfluorescence and leakage can be observed corresponding to the abnormal vascular network. However the polyps and polypoidal network cannot be identified in FFA. Therefore FFA does not have any diagnostic features in PCV.
Spectral-Domain Optical coherence tomography (SD-OCT) imaging in PCV (Fig.6, 7)
There are characteristic SD-OCT features of PCV.31, 32, 33
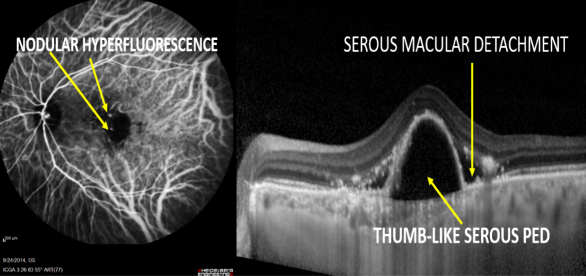
- Thumb like a polyp (TLP), the thumb like elevation of pigment epithelium denotes the polypoidal network underneath the pigment epithelium identified by ICG Angiography. Exudates from the polypoidal network further elevate the retinal pigment epithelium
- Tomographic notch in the PED: It signifies the polypoidal lesion seen in ICG angiography at the location of the notch in the PED
- A hyper-reflective ring surrounding an area of hyporeflectivity located in the TLP probably representing the polypoidal lumen
- Double-layered sign (DLS): Consisting of two hyper-reflective layers. The inner layer is the hyperreflective irregularly elevated RPE and the outer layer is the inner layer of the Bruch’s membrane. The two layers are separated by a gap and the extent of the DLS in SD-OCT can be assessed by vertical and horizontal scans. The DLS is a very characteristic feature of PCV and seen in most of the cases. It represents the extent of the Branching vascular network.
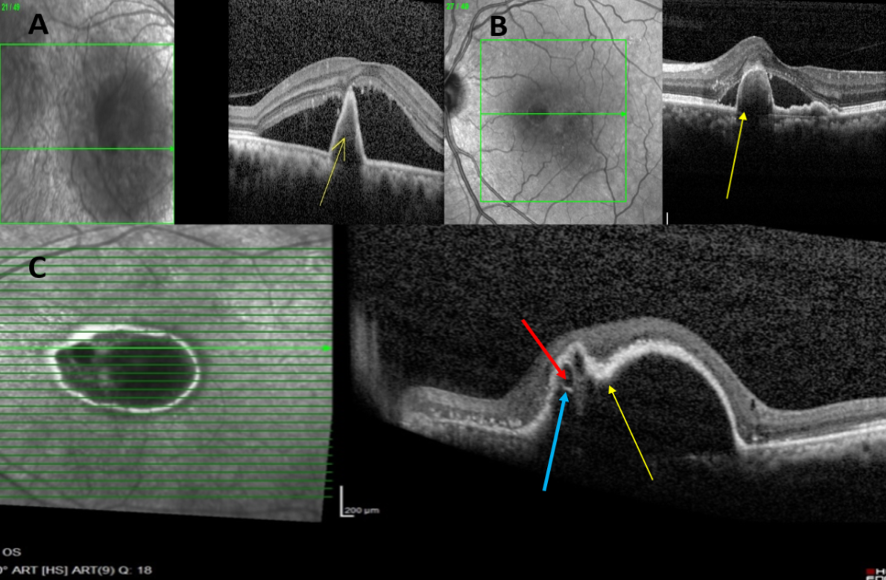
Figure 6: Spectral-domain optical coherence tomography (SD-OCT) images showing characteristic features of PCV: A) Sharp-peaked PED (Yellow arrow); B) Thumb-like PED, also called thumb-like polyp (TLP; Yellow arrow); C) Notch present at the nasal portion of PED (Notched PED; Yellow arrow), along with hyporeflective lumen (Red arrow) surrounded by a hyperreflective ring (Blue arrow) attached to undersurface of RPE
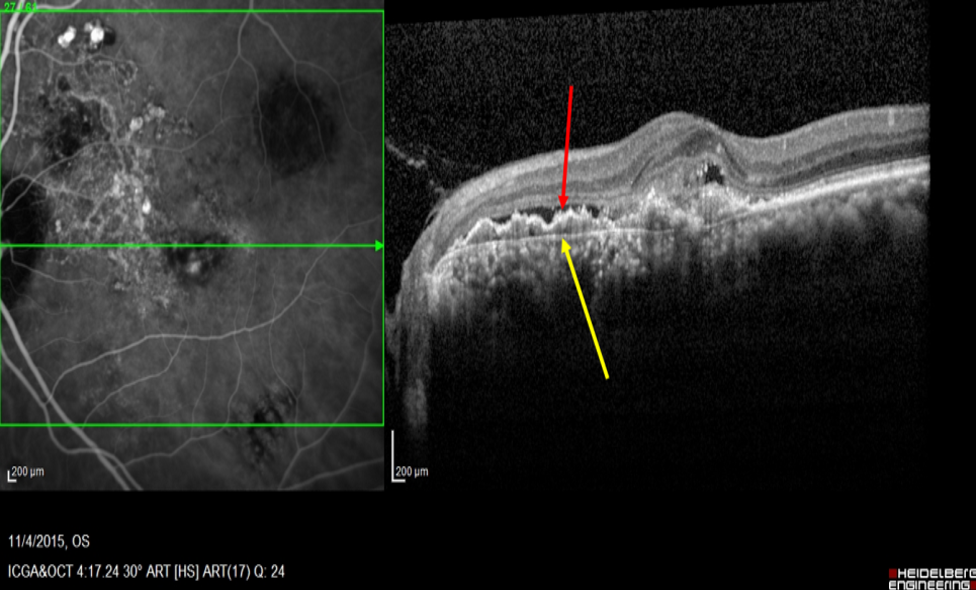
Figure 7: SD-OCT showing the presence of Double-layer sign (DLS), represented by two hyperreflective lines, inner signifying shallow RPE elevation (Red arrow) and outer signifying Bruch’s membrane (Yellow arrow) indicative of abnormal vascular network (AVN)
Natural Course
Patients with PCV can be asymptomatic if there is no leakage from the polypoidal lesions. Patients with symptomatic PCV can present both acutely or with progressive visual loss. Acute visual loss in PCV is usually secondary to spontaneous rupture of the polypoidal lesions resulting in a submacular hemorrhage .5, 10 Patient may also present with the progressive visual loss with metamorphopsia and gradual progressive decrease in vision due to the accumulation of subretinal fluid and exudates. The natural course of PCV is variable and it has been estimated that 50% of patients will have a favorable course in which spontaneous regression of polyps can occur without treatment.34, 35However, in the remaining 50% repeated bleeding and exudation will result in RPE and photoreceptor degeneration, scarring, and irreversible visual loss.36 The presence of a large cluster of polyps might be a risk factor for poor visual prognosis due to the high risk of bleeding. Kwok et-al in a retrospective study, with a mean, follow up of 28.2 months without treatment noticed that 76.9% of cases had a final vision of 20/200 or worse.37
Therefore the natural course of the disease is not favorable and active intervention is indicated in patients with symptomatic PCV.
Management of PCV
All eyes with Active symptomatic PCV should be treated. The choice of treatment depends on the location of the polypoidal network, extent of the network, and associated clinical features.38The treatment should be targeted to include the polyps and the surrounding abnormal vascular network.
Treatment of sub-foveal and Juxtafoveal polyps:
Active Sub-foveal and juxtafoveal PCV should be treated with full fluence photodynamic therapy (PDT) and three loading dose of Anti VEGF injection based on Level 1 evidence form the EVEREST study.39
The EVEREST study was the first randomized controlled trial comparing Standard fluence PDT with or without Ranibizumab 0.5mg and Ranibizumab monotherapy in Polypoidal Choroidal Vasculopathy. The primary endpoint was the proportion of patients with complete regression of polyps at 6 months as determined by ICG. This study established the efficacy of PDT in the closure of the polyps. The study shared that both combination therapy and PDT monotherapy resulted in a significantly higher proportion of PCV eyes having complete regression of polyps on ICG Angiography than Ranibizumab alone at 6 months (77.8% Vs 71.4% Vs 28% respectively)
Before performing PDT, ICG Angiography should be performed to evaluate the location and size of the PCV lesion. The PDT laser spot size is generally determined by the Greatest Linear Dimension (GLD) of the lesion including both the polyp and the interconnecting BVN based on ICG Angiography.40 Veteporfin is infused intravenously at a dose of 6mg/sqm of body surface area over a period of 10 minutes followed by application of 689 nm laser 15 minutes after initiation of infusion with a light dose of 50J/sq cm of 83 seconds.41 Patients should be warned to avoid excessive sunlight exposure for 48-72 hours after PDT. It is hypothesized that application of the non-thermal PDT laser on the targeted area results in transient local choroidal vascular occlusion which reduces perfusion to PCV lesions and subsequently leads to thrombosis of supplying choroidal vessels and finally results in regression of PCV lesions.42
After completion of PDT, Intravitreal Anti VEGF injection can be given within the first 24 hours. Adjunct treatment with Anti VEGF agent suppresses the immediate upregulation of VEGF levels following PDT.43, 44 The ideal recommendation based on the EVEREST study and the Evidence-based guidelines on the management of PCV recommends a loading dose of three Anti VEGF Intravitreal injections on a monthly basis following PDT.38, 39 Combination therapy helps early resolution of the sub-retinal and serosanguinous fluid and thereby help in the better visual outcome.
Case 1: A 63-year-old male presented with blurring of vision in OS for 1 month with the presence of subfoveal PED. His BCVA was 6/6 in OD and 6/60 in OS. SD-OCT confirmed the presence of subfoveal thumb-like serous PED and serous macular detachment (SMD) and ICGA demonstrated nodular hyperfluorescence correspondingly. A diagnosis of polypoidal choroidal vasculopathy (PCV) was made.
After 3 months of treatment with full-fluence photodynamic therapy (PDT) and three doses of intravitreal ranibizumab, there was complete resolution of nodular hyperfluorescence on ICGA in addition to the resolution of PED and SMD on SD-OCT.
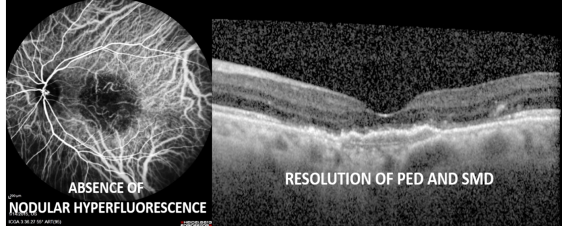
Follow up recommendations
Best-corrected visual acuity, SD-OCT, FFA and ICG are repeated three months following combination therapy.38If there is complete regression of polyp in ICG angiography, patients are followed up at three monthly intervals with best-corrected visual acuity, SD-OCT and Biomicroscopic examination of the macula. If there is any suspicion of recurrence of polyps found on clinical examination or SD-OCT, ICG and FFA can be repeated.
Treatment of Extrafoveal Polyps
Direct thermal laser photocoagulation with argon green, double frequency ND-YAG or diode laser may be utilized for the treatment of Extrafoveal PCV. 42 Laser is directly applied to the polyp and surrounding BVN identified by ICG Angiography.43, 44 Thermal laser is routinely done with 532 green laser with a spot size of 100-200 µm and duration of 200 msec so as to result in whitening of the retina in a confluent manner..11
One major concern with the use of thermal laser is chorioretinal scarring.45, 46 Therefore in lesions that are 200-500 microns from the centre of the fovea and large in size, the use of thermal laser can result in collateral damage.45, 46For these patients, it is preferable to consider PDT with three loading doses of anti-VEGF agents.
Thermal laser may not be possible when there is a significant amount of exudates and or hemorrhage. In such situations Intravitreal Anti-VEGF therapy can be given initially to reduce the exudates and hemorrhage. ICG Angiography can be repeated three months following laser treatment.
The outcome of thermal laser is variable. Kwok et al reported that 56% of treated eyes with thermal laser had stable visual acuity as compared to 31% of the non-lasered group.37Lee et al found at 12 months after thermal laser, 75% of eyes had stable or improved vision and 64.3% of the eyes showed clinical or angiographic resolution of maculopathy.45Despite receiving thermal laser 14.3% of the eyes still had persistent leakage causing neurosensory detachment and 10.7% of treated eyes developed sub-foveal recurrence of polyps.
Case 2: A 63-year-old female presented with the blurring of vision in OD for 3 months. Her BCVA was 6/60 in OD and 6/6 in OS. ICGA demonstrated extrafoveal nodular hyperfluorescence, confirming the presence of Extrafoveal PCV. She was managed with thermal laser with three loading doses of intravitreal ranibizumab. Subsequently, there was complete resolution of polyps on repeat ICGA.
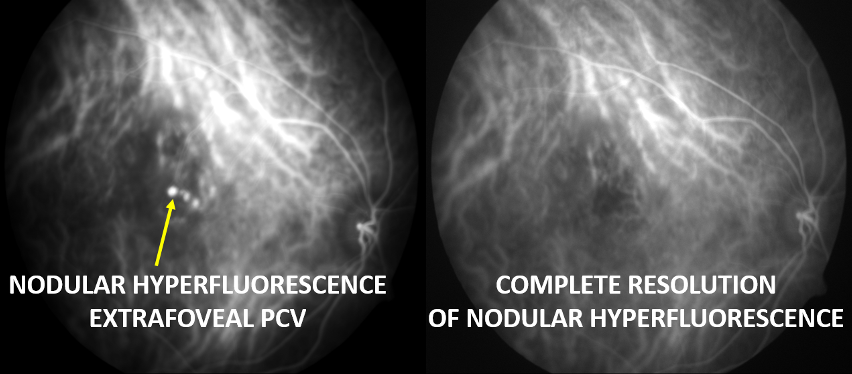
Anti-VEGF monotherapy for PCV
Various studies have found that Anti VEGF monotherapy may reduce sub-retinal fluid and cause stabilization of vision in eyes with PCV.49-51 However choroidal vascular changes on ICG Angiography generally persist despite Anti VEGF treatment. Kokame et al, reported that monthly Intravitreal injection of Ranibizumab resulted in visual stabilization in all twelve PCV patients at 6 months with macular edema and sub-retinal fluid improved in 80% and 60% of eyes respectively.48 Reduction in polyp on ICG Angiography was observed in only 33% of patients.48 Hikichi et-al reported the results of2 years of treatment with as-needed Ranibizumab agents for PCV in 75 PCV patients.50 All patients received three monthly loading doses of Intravitreal Ranibizumab injection followed by as-needed reinjection. Mean logMAR BCVA improved from 0.59 at baseline to 0.37 and 0.41 logMAR at 12 months and 24 months respectively. Compete for regression of polyps, however was seen in only 25% of the eyes at 24 months. Moreover, despite the promising visual outcomes, BVN persisted in all the patients and tended to increase in size.
Case 3: A 60-year-old female presented with the blurring of vision in OS for 3 months. Her BCVA was 6/6 in OD and CF 3m in OS. She had hemorrhagic PED at the posterior pole, which was confirmed on SD-OCT which showed thumb-like PED with dense hyperreflectivity with backshadowing suggestive of hemorrhagic PED. ICGA demonstrated with nodular hyperfluorescence, abnormal vascular network (AVN), and a large area of blocked hypofluorescence due to hemorrhage. Polypoidal choroidal vasculopathy (PCV) was confirmed.
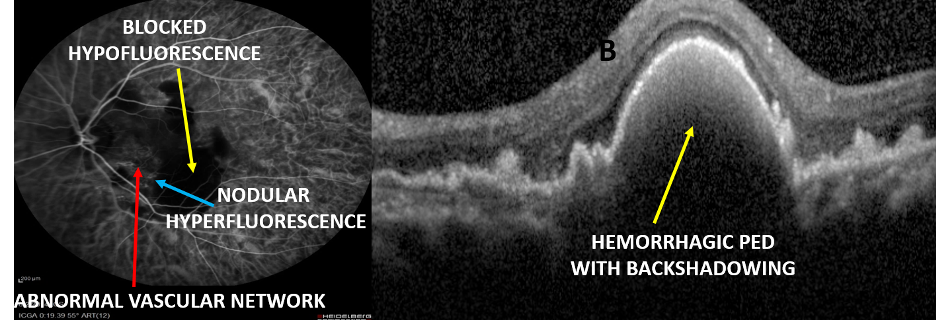
Since the patient had significant hemorrhage at the posterior pole, she was treated with four doses of monthly intravitreal ranibizumab. Subsequent ICGA showed residual polyps with AVN while the blocked fluorescence resolved completely and corresponding SD-OCT demonstrated resolution of PED but persistence of double-layer sign (DLS).
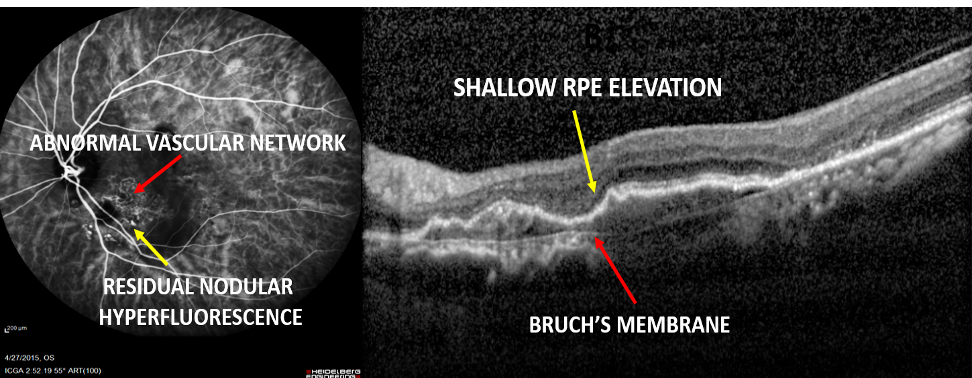
Summarising, monotherapy with Ranibizumab or Bevacizumab is effective in the resolution of sub-retinal fluid, haemorrhage, and improvement of visual acuity. However, regression of polyps is only seen in one-third of the treated eyes. Moreover, there are a small sub-group of eyes with PCV, which are non-responds to Anti VEGF therapy.
Chikako Hara et al reported the one year result of Intravitreal Afliberceptin PCV.51 At 3 months polypoidal changes in ICG angiography completed resolved in 60% of the eyes. Various other short-term studies have shown similar results with Aflibercept in PCV.52, 53 Aflibercept, therefore, appears to be more promising than other Anti-VEGF agents in the treatment of PCV. Further long-term studies are required to establish the role of Aflibercept in PCV.
Nonetheless, if initially the extent of lesion is not clearly defined on ICG due to the presence of blocked fluorescence secondary to hemorrhage, it is advisable to initiate anti-VEGF monotherapy alone. Once the hemorrhage clears, ICG + DFA should be performed followed by ICG guided PDT in combination with anti-VEGF agents.
Conclusion
More than three decades have passed since the original description of PCV in 1982 and it is now a well-recognized and common disease entity. Indocyanine green angiography is the gold standard for the diagnosis of PCV. SD-OCT imaging has characteristic features with high sensitivity and specificity to suspect PCV. Current treatment modalities for PCV include combination therapy using Verteporfin PDT with Anti-VEGF agent, Anti-VEGF monotherapy, and thermal laser photocoagulation. The choice of specific treatment modality depends upon the location and amount of polypoidal lesions, the extent of the abnormal vascular network, and concurrent amount of hemorrhage and exudation.
References
- Yannuzzi LA, Sorenson J, Spaide RF, Lipson B. Idiopathic polypoidal choroidal vasculopathy (IPCV). Retina 1990;10:1-8.
- Yannuzzi LA, Ciardella A, Spaide RF, et al. The expanding clinical spectrum of idiopathic polypoidal choroidalvasculopathy. Arch Ophtahlmol. 1997;115(4):478-85.
- Stern RM, Zakov ZN, Zegarra H, et al. Multiple recurrent serosangineous retinal pigment epithelial detachment inblack woman. Am J Ophthalmol. 1985;100(4):560-9.
- Kleiner RC, Brucker AJ, Johnston RL. The posterior uvealbleeding syndrome. Retina. 1990;10(1):9—17.
- Sho K, Takahashi K, Yamada H, Wada M, Nagai Y, Otsuji T, et al. Polypoidal choroidal vasculopathy: incidence, demographic features, and clinical characteristics. Arch Ophthalmol 2003;121(10):1392-6.
- Spaide RF, Yannuzzi LA, Slakter JS, et al. Indocyaninegreen videoangiography of idiopathic polypoidal choroidalvasculopathy. Retina. 1995;15(2):100—10.
- Freund KB, Zweifel SA, Engelbert M. Do we need a newclassification for choroidal neovascularization in age-related macular degeneration? Retina 2010;30:1333–1349.
- Ueta T, Obata R, Inoue Y, et al. Background comparison oftypical age-related macular degeneration and polypoidal choroidalvasculopathy in Japanese patients. Ophthalmology 2009;116:2400–2406.
- Nagase, S.; Miura, M.; Makita, S.; Iwasaki, T.; Goto, H.; Yasuno, Y. High-penetration opticalcoherence tomography with enhanced depth imaging of polypoidal choroidal vasculopathy.OphthalmicSurg. Lasers Imaging 2012;43:5–9.
- Imamura Y, Engelbert M, Iida T, Freund KB, Yannuzzi LA. Polypoidal choroidal vasculopathy: a review. SurvOphthalmol 2010; 55:501-15.
- Anantharaman G, Ramkumar G, Gopalakrishnan M, Rajput A. Clinical features, management and visual outcome of polypoidal choroidal vasculopathy in Indian patients. Indian J Ophthalmol 2010;58:399–405.
- Liu Y, Wen F, Huang S, Luo G, Yan H, Sun Z, et al. Subtype lesions of neovascular age-related macular degeneration in Chinese patients. Graefes Arch ClinExpOphthalmol 2007;245:1441–5.
- Maruko I, Iida T, Saito M, Nagayama D, Saito K. Clinical characteristics of exudative age-related macular degeneration in Japanese patients. Am J Ophthalmol 2007;144(1):15-22.
- Chang YC, Wu WC: Polypoidal choroidal vasculopathy in Taiwanese patients. Ophthalmic Surg Lasers Imaging 2009;40:576-81.
- Byeon SH, Lee SC, Oh HS, Kim SS, Koh HJ, Kwon OW. Incidence and clinical patterns of polypoidal choroidal vasculopathy in Korean patients. Jpn J Ophthalmol 2008;52:57-62.
- Lafaut BA, Leys AM, Snyers B, Rasquin F, De Laey JJ. Polypoidal choroidal vasculopathy in Caucasians. Graefes Arch ClinExpOphthalmol 2000;238:752-9.
- Ladas ID, Rouvas AA, Moschos MM, Synodinos EE, Karagiannis DA, Koutsandrea CN. Polypoidal choroidal vasculopathy and exudative age-related macular degeneration in Greek population. Eye (Lond) 2004;18:455-9.
- Scassellati-Sforzolini B, Mariotti C, Bryan R, Yannuzzi LA, Giuliani M, Giovannini A. Polypoidal choroidal vasculopathy in Italy. Retina 2001;21:121-5.
- Honda S,Matsumiya, W. &Negi, A. (2014). Polypoidal Choroidal Vasculopathy: Clinical features and genetic predisposition. Ophthalmologica, 231(2),59-74.
- Chen H, Liu K, Chen LJ, Hou P, Chen W, Pang CP. Genetic associations in polypoidal choroidalvasculopathy: a systematic review and meta-analysis. Mol Vis. 2012;18:816-29. Epub 2012 Apr 4.
- Hiroyuki Nakashizuka, Masako Mitsumata, ShigekuniOkisaka, Hiroyuki Shimada, Akiyuki Kawamura, Ryusaburo Mori, MitsukoYuzawa; Clinicopathologic Findings in Polypoidal Choroidal Vasculopathy. Invest. Ophthalmol. Vis. Sci. 2008;49(11):4729-4737. doi: 10.1167/iovs.08-2134.
- Chung SE, Kang SW, Kim JH, Kim YT, Park do Y. Engorgement of vortex vein and polypoidal choroidalvasculopathy. Retina. 2013 Apr;33(4):834-40
- Balaratnasingam, Chandrakumar, Won-Ki Lee, Hideki Koizumi, KunalDansingani, Maiko Inoue, and K. Bailey Freund. “Polypoidal Choroidal Vasculopathy: A Distinct Disease or Manifestation of Many?.” RETINA Publish Ahead of Print (9000).
- Nakashizuka H,Mitsumata M,Okisaka S,Shimada H,Kawamura A,Mori R, et al. Clinicopathologic Findings in Polypoidal Choroidal Vasculopathy.Invest. Ophthalmol. Vis. Sci2008;49(11):4729-37.
- Stanga PE, Lim JI, Hamilton P. Indocyanine greenangiography in chorioretinal diseases: indications andinterpretation: an evidence-based update. Ophthalmology. 2003;110(1):15-21.
- Desmettre T,Devoisselle JM,Mordon S. Fluorescence properties and metabolic features of indocyanine green (ICG) as related to angiography. Surv. Ophthalmo2000, 45, 15-27.
- Li X. Retina. Polypoidal choroidal vasculopathy. In: Sadda S, Hinton D, editors. RyanSJ: Retinal Imaging and Diagnostics5th ed. Philadelphia. Elsevier pp1285-1290
- Tan CS,Ngo WK,Chen JP,Tan NW,Lim TH;EVEREST Study Group. EVEREST study report 2: imaging and grading protocol, and baseline characteristics of a randomised controlled trial of polypoidal choroidal vasculopathy. Br J Ophthalmol 2015;99: 624–8.
- Japanese Study Group of Polypoidal Choroidal Vasculopathy.[Criteria for diagnosis of polypoidal choroidal vasculopathy].Nippon GankaGakkaiZasshi 2005;109(7):417–27.
- Gomi F,Sawa M,Mitarai K,Tsujikawa M,Tano Y. Angiographic lesion of polypoidal choroidal vasculopathy on indocyanine green and fluorescein angiography. Graefe’s Arch ClinExpOphthalmol 2007;245:1421-7.
- AlshahraniST,Al Shamsi HN,Kahtani ES,Ghazi NG.Spectral-domain optical coherence tomography findings inpolypoidalchoroidalvasculopathysuggest a type 1 neovascular growth pattern.ClinOphthalmol2014;8:1689-95.
- De SalvoG,Vaz-Pereira S,Keane PA,Tufail A,Liew G. Sensitivity and specificity of spectral-domainoptical coherence tomography in detecting idiopathicpolypoidalchoroidalvasculopathy. Am J Ophthalmol2014;158(6):1228-38.
- Liu R,Li J,Li Z,Yu S,Yang Y,Yan H, et al. Distinguishingpolypoidal choroidal vasculopathy from typical neovascular age-related macular degeneration based on spectraldomain optical coherence tomography. Retina2015;0:1-9.
- Okubo A, Sameshima M, Sakamoto T. Plasticity of polypoidal lesions in polypoidal choroidal vasculopathy.Graefes Arch ClinExpOphthalmol. 2004;242(11):962-5.
- Uyama M, Matsubara T, Fukushima I, et al. Idiopathic polypoidal choroidal vasculopathy in Japanese patients.Arch Ophthalmol. 1999;117(8):1035-42.
- Uyama M, Wada M, Nagai Y, et al. Polypoidal choroidalvasculopathy: natural history. Am J Ophthalmol. 2002;133(5):639-48.
- Kwok AK,Lai TY,Chan CW,Neoh EL,Lam DS. Polypoidal choroidal vasculopathy in Chinese patients.Br. J. Ophthalmol2002;86:892-7.
- KohAH;ExpertPCVPanel,Chen LJ,Chen SJ,Chen Y,Giridhar A,Iida T,et al. Polypoidal choroidal vasculopathy: evidence-basedguidelinesfor clinical diagnosis and treatment. Retina2013;33(4):686-716.
- Koh A,Lee WK,Chen LJ,Chen SJ,Hashad Y,Kim H, et al. EVEREST study: efficacyand safety of verteporfin PDT in combination with ranibizumabor alone versus ranibizumab monotherapy in patientswith symptomatic macular polypoidal choroidal vasculopathy.Retina 2012;32:1453–64.
- Otani A,Sasahara M,Yodoi Y,Aikawa H,Tamura H,Tsujikawa A, et al. Indocyanine green angiography: guided photodynamic therapy for polypoidal choroidal vasculopathy. Am J Ophthalmol 2007;144(1):7-14.
- Wong R, Lai T. Polypoidal Choroidal Vasculopathy: AN Update on Therapeutic Approaches. J. Ophthalmic Vis Res 2013;8(4):359-71.
- Lai TY, Chan WM. An update in laser andpharmaceutical treatment for polypoidal choroidalvasculopathy. Asia-Pac J Ophthalmol 2012;1:97-104.
- Nishijima K, Takahashi M, Akita J, Katsuta H, Tanemura M, Aikawa H,et al. Laser photocoagulation of indocyanine green angiographically identified feeder vessels to idiopathic polypoidal choroidal vasculopathy. Am J Ophthalmol 2004;137(4):770-3.
- Yuzawa M, Mori R, Haruyama M. A study of laser photocoagulation for polypoidal choroidal vasculopathy. Jpn J Ophthalmol 2003;47(4):379-84.
- Lee MW, Yeo I, Wong D, Ang CL. Argon laser photocoagulationfor the treatment of polypoidal choroidal vasculopathy. Eye (Lond) 2009;23:145-8.
- Yuzawa M, Mori R, Haruyama M. A study of laser photocoagulation for polypoidal choroidal vasculopathy. Jpn J Ophthalmol 2003;47(4):379-84.
- Gomi F, Sawa M, Sakaguchi H, TsujikawaM, Oshima Y, Kamei M, et al. Efficacy ofintravitreal bevacizumab for polypoidal choroidalvasculopathy. Br J Ophthalmol 2008;92:70-73.
- Kokame GT, Yeung L, Lai JC. Continuous anti-VEGF treatment with ranibizumab for polypoidalchoroidal vasculopathy: 6-month results. Br JOphthalmol 2010;94:297-301.
- Lai TY, Chan WM, Liu DT, Luk FO, Lam DS.Intravitreal bevacizumab (Avastin) with or withoutphotodynamic therapy for the treatment ofpolypoidal choroidal vasculopathy. Br J Ophthalmol2008;92:661-666.
- Hikichi T, Higuchi M, Matsushita T, Kosaka S,Matsushita R, Takami K, et al. Results of 2 years oftreatment with as-needed ranibizumab reinjectionfor polypoidal choroidal vasculopathy. Br JOphthalmol 2013;97:617-621.
- Hara C, Sawa M, Sayanagi K, et al. One-year results of intravitrealAflibercept for polypoidalChoroidal vasculopathy. Retina 2015;1–9.
- Inoue M,Arakawa A,Yamane S,Kadonosono K. Short-term efficacy of intravitrealafliberceptin treatment-naive patients withpolypoidalchoroidalvasculopathy. Retina2014;34(11):2178-84.
- Yamamoto A,Okada AA,Kano M,Koizumi H,Saito M,Maruko I, et al. One-Year Results of IntravitrealAfliberceptforPolypoidalChoroidalVasculopathy. Ophthalmology2015;122(9):1866-72.

