Intravitreal injections involve injecting therapeutic agents (drugs/air/gases) inside the vitreous cavity throughpars planaunder aseptic precautions
Surgical anatomy
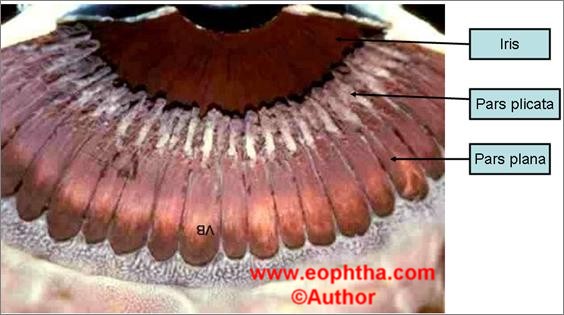
- Site of injection: Pars plana
- Ciliary body: Consists of two parts
a) anterior pars plicata
b) posterior pars plana
Pars plicata
Circumferential zone
2.5mm in anteroposterior diameter
Pars plana
Varies in anteroposterior diameter nasally 3mm wide and temporally 4.5mm wide. This variation is because of the posterior globe on temporal side curves posteriorly & laterally more than on the nasal side. Pars plana extend from pars plicata anteriorly to ora serrata (termination of the retina) posteriorly. Blood vessels in pars plana are radially oriented but circumferential incisions across these vessels cause no bleeding. Also, lens zonules insert into ciliary body avoiding pars plana
Thus, needle punctures 3.5 mm posterior to limbus enter globe through pars plana
- Not damaging lens
- Not puncturing the retina
- Not tethering / damaging the vitreous base
Ciliary body, lens & retina attain adult size by 7years of age
Linear relation exists between the axial length of eye & pars plana during development
Hence in paediatric eyes site of injection varies taking into consideration small size of eye, anterior placement of pars plana & relatively greater size of crystalline lens.
|
Age |
Distance of injection from limbus |
|
1- 6 months |
1.5 mm |
|
6 mths—1year |
2 mm |
|
1—2yr |
2.5 mm |
|
2—6yr |
3 mm |
|
7 yr onwards |
4 mm phakic, |
Table 1: Distance of site of injection from limbus according to age
|
|
Indication |
Standard dosage |
|
|
Absolute |
Relative |
||
|
Triamcinolone Acetonide |
Refractory CSME, refractory pseudophakic CME, CRVO, BRVO. |
- |
2 mg in 0.05 ml / 4mg in 0.1ml |
|
Macugen |
CNVM |
Refractory CSME, PDR, NVG, CRVO, BRVO. |
0.3 mg in 90 µl |
|
Lucentis |
CNVM |
Refractory CSME, PDR, NVG, CRVO, BRVO. |
0.3 mg in 0.05 ml |
|
Avastin |
CNVM |
Refractory CSME, PDR, NVG, CRVO, BRVO. |
1.25 mg in 0.05 ml |
|
Vancomycin |
Endophthalmitis |
- |
1 mg 0.1ml |
|
Amikacin |
Endophthalmitis |
- |
0.4 mg 0.1ml |
|
Ceftazidime/cefazoline/cefotaxime |
Endophthalmitis |
- |
2.25 mg in 0.1ml |
|
Amphotericin B |
Endophthalmitis |
- |
5 mcg in 0.1ml |
|
Air |
Pneumatic retinopexy |
- |
0.5-0.8cc |
|
SF6 (100%) |
Pneumatic retinopexy |
- |
0.5 ml |
|
C3F8 (100%) |
Pneumatic retinopexy |
- |
0.3 ml |
Table 2: Drugs, indications and dosage
CSME: clinical significant macular edema, CRVO: central retinal vein occlusion, BRVO: branch retinal vein occlusion, CNVM: choroidal neovascular membrane, AMD: age related macular degeneration, PDR: proliferative diabetic retinopathy, NVG: neovascular glaucoma.
Technique
Informed consent clearly explaining what the treatment involves/what to expect/what the risks are 1-3
Equipment
Eyelid speculum, gloves, toothed microforceps, cotton buds, eye drape, callipers, povidone iodine, 0.5% proparacaine, 26 or 30 gauge needle, tuberculin syringe
Preparation & Administration:4-7
- Pupil-- dilated
- Prepare periocular area with 10% iodine solution
- Instill 5% povidone iodine drops (3 mins)
- Instill topical 0.5% Proparacaine
- Under aspetic precautions drape placed, speculum inserted
- Patient is asked to look away from site of injection (superonasally)
- Mark the injection site using the caliper (the entry site of the needle should be 3.0-3.5 mm from the limbus in aphakic/pseudophakic patients, and 3.5-4.0 mm in phakic patients).
- Avoid the horizontal meridians of the globe; the inferotemporal quadrant is preferred
- Using forceps to steady the eye, the needle is inserted perpendicular through sclera with the tip aimed towards the centre of the globe (to avoid any contact with the posterior lens)
- One can even go 15 degrees tangential to the sclera for a few millimeters and then insert perpendicularly so as to create a small self-sealing tunnel. The conjunctiva can also be retracted before injection so that the scleral and conjuctival entry do not overlap after completing the injection.
- Remove needle slowly and carefully.
- A sterile cotton-tipped applicator is used to prevent reflux and to steady the eye.
Check that the patient is able to see objects immediately after injection to ensure that the retinal artery is perfused.
Post-procedure
Advise patient to administer antibiotic drops QDS for a minimum of 5 days postinjection.
They should be instructed to report immediately if any symptoms like eye pain or discomfort, increased redness or additional blurring of vision arise. Floaters are common and should be explained to patients (especially with triamcinolone acetonide)
Follow-up
24 hours after injection. In cases of triamcinolone injections patient is asked to follow-up after 2 wks for Intra ocular pressure (IOP) measurement.
In cases of anti-VEGF injections follow-up after 4 wks
Complications
- Defined risks (should be included in every informed consent)
- Infection (0.16%, 2-year incidence in over 10,000 injections)
- Retinal detachment (0.15%, 2-year incidence in over 10,000 injections);
- Lens trauma/acute cataract (0.07%, 2-year incidence in over 10,000 injections).
- Sub-conjunctival / vitreous hemorrhage
- Vitreous wick syndrome
- Precipitated angle closure
- Elevated IOP with perfusion compromise (CRAO: Paracentesis to be done immediately)
- Wound leak and hypotony
- Anaphylactic reaction to either the agent or peri-procedural materials.
References:
- Jager RD, Aiello LP, Patel SC Cunningham ET. Risks of intravitreous injection: a comprehensive review. Retina 2004; 24:676-698.
- Speaker MG, Milch FA, Shah MK et al. Role of external bacterial flora in the pathogenesis of acute post-operative endophthalmitis. Ophthalmology 1991; 98:639-649.
- Gragoudas ES, Adamis AP, Cunningham AP et al. Pegaptanib for neovascular age-related macular degeneration. N Eng J Med 2004; 351:2805-2816.
- Aiello LP, Brucker AJ, Chang S, Cunningham ET et al. Evolving guidelines for intravitreous injections. Retina 2004; 24: S3-S19.
- Speaker MG, Menikoff JA. Prophylaxis of endophthalmitis with topical povidone-iodine. Ophthalmology 1991;98:1769-1775.
- Cuilla TA, Starr MB, Masket S. Bacterial endophthalmitis prophylaxis for cataract surgery: an evidence-based update. Ophthalmology 2002;109:13-24.
- Ta CN. Minimizing the risk of endophthalmitis following intravitreous injections. Retina 2004; 24:699-705