Viral ocular infections can cause sight-threatening diseases in humans either as an isolated ocular infection or as a part of systemic illness. Although many viruses may naturally be harbored in human beings, the common ones causing ocular infections include herpes simplex virus (HSV), varicella-zoster virus (VZV), and cytomegalovirus (CMV). They may remain dormant for several years with periodic asymptomatic virus shedding in immunocompetent individuals. However, in situations where the immunity is reduced due to age or, comorbidities, the symptomatic disease can occur. Ocular damage is caused by the direct cellular damage by the virus itself or the inflammatory response to the infection. Virus-specific T lymphocytes as well as humoral immunity with specific antibodies help to control the acute infection. Here we look at anterior and posterior viral uveitis in two separate sections and the common causative viruses.
Viral Anterior Uveitis ( VAU)
Introduction
Anterior uveitis (AU) is the most common form of uveitis worldwide, accounting for about three-fourth of uveitis cases reported in developed countries and around 28–50 % in developing regions [1,2,3]. Although the majority of the anterior uveitis is related to seronegative arthritis, viruses remain an important cause. Various viruses are being found to affect humans and we will be discussing the most commonly encountered viral anterior uveitis (VAU) agents like herpes simplex virus (HSV), herpes zoster virus (HZV), and cytomegalovirus (CMV).
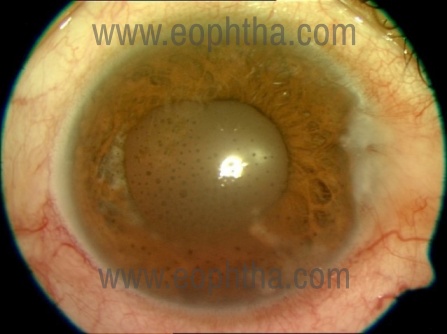
Figure 1: Viral Anterior Uveitis
Herpes Simplex Virus (HSV) associated AU (HSV-AU)
HSV is a DNA virus belonging to the Herpesviridae family. It may manifest either as a primary or a secondary infection. Being ubiquitous in nature, the virus can remain lifelong within the fifth cranial nerve sensory ganglion This state is known to be a latent infection, wherein viruses are still there in the human body without any clinical manifestation. At times, depending on a reduced host immunity due to age or comorbidities, these viruses get reactivated to give rise to a secondary infection. Transmission can be directly through skin-to-skin contact, fomites, or aerosol. Usually, HSV-AU occurs in the fourth – fifth decades of life, and both the sexes are equally affected [4]. Although both HSV 1 and 2 can cause ocular infections, HSV1 is more commonly associated with anterior uveitis or keratouveitis.
Clinical manifestations
HSV-AU not only has inflammatory reactions in the anterior chamber (AC) but can also affect the cornea and the trabecular tissues. A corneal scar is seen in around 33 % and a raised intraocular pressure (IOP) is encountered in around 38%–90% of eyes [5,6,7]. These features if present with a reduced corneal sensation are a helpful clue towards viral etiology. AC inflammation could range from a mild flare to moderate inflammation with cells and flare. Keratic precipitates (KPs) may be granulomatous or non-granulomatous. The pupil may be distorted either due to the presence of posterior synechiae or due to segmental iridoplegia causing flattening of the pupil. Amongst all these manifestations, the most striking feature is the presence of iris atrophy (Figure 1) which may be diffuse (10%) of eyes or sectoral (50%) with transillumination defects [7]. However, this might not be present earlier, in the course of the disease process, and only develop subsequently.
Herpes Zoster Virus (HZV) associated AU (HZV- AU)
Similar to HSV-AU, this also has a tendency to remain latent in the ophthalmic division of the trigeminal ganglion lifelong, only to get reactivated in periods of diminished cell-mediated immunity [8,9]. It manifests little later in life, during the sixth–seventh decade. It is usually associated with skin manifestations or dermatopathy, in the form of either skin blisters or rashes. These skin features are unique by way of their adherence to a particular dermatome, confined to the midline, seen in Herpes Zoster Ophthalmicus (HZO). However, sometimes HZV-AU can occur without any skin manifestation. This is termed as herpes zoster sine herpete. In such a scenario, other clinical features aid towards a possible diagnosis. Similar to HSV, it also manifests as either non-granulomatous or granulomatous AU with KPs and a raised IOP. KPs, in both HSV and HZV anterior uveitis, crosses the midline of the corneal endothelium. The cornea may show interstitial or nummular keratitis and ring infiltrate. Corneal scars alone may be seen in 25% of cases.[7]. The striking difference between the two homologically similar viruses is the pattern of iris atrophy. Diffuse iris atrophy has never been reported in HZV-AU. Patchy or sectoral iris atrophy with base towards the iris root occurs due to occlusive iris vasculitis and manifests in about 88%. Rarely there may be extensive iris atrophy and sphincter damage. However, like HSV-AU, iris atrophy develops later in the disease course and is found to be related closely to the aqueous viral load [10]. Trabecular meshwork appears pigmented. Secondary glaucoma may occur due to trabeculitis, posterior synechiae, and pupillary block or peripheral anterior synechiae
CMV associated AU (CMV-AU)
CMV usually is asymptomatic and may manifest systemically as a mild flu-like illness. Similar to the above, it also has the capability to remain latent in the monocytes. Inclusion bodies have been identified in the iris and ciliary body as well [11]. It has a bimodal age of distribution, affecting both younger and older age groups. Acute infection similar to Posner Schlossman Syndrome usually affects the 30 – 50 age groups whereas chronic infection affects the 50 – 70 age group of patients. Unlike the above two, CMV-AU has a male preponderance [12]. Corneal involvement is in the form of endothelitis, manifesting as round, centrally located lesions with a surrounding halo. The viruses are also found to cause trabeculitis, hence are the most commonly associated with the high IOP. KPs are usually non-granulomatous, with a more central location of occurrence. Sometimes, KPs are distributed in a ring fashion and are termed as coin-shaped lesions which are characteristic of CMV-AU [13]. However, inflammation is usually mild with minimal flare, leading to a relatively non-congested or mildly congested eye, in spite of a high IOP. The inflammation is so mild that there is no posterior synechiae formation seen. Therefore, it becomes essential to rule out the possibility of a CMV association in any hypertensive uveitic patients presenting with mild inflammation. Iris atrophy can either be in form of sectoral or diffuse, mostly diffuse atrophy, however, iris heterochromia is rare.
Special forms of VAU
Fuchs Uveitis Syndrome (FUS)
FUS was first described by Ernst Fuchs in 1906 as heterochromic iridocyclitis. However, heterochromia, though a sensitive sign, was not quite a feature of dark brown irides. Hence, the term changed from Fuchs heterochromic iridocyclitis to FUS. It accounts for nearly 20% of AU [17]. FUS has varied clinical manifestations. The classic form has an insidious onset, manifesting between third to fourth decades, and is non-granulomatous in nature. However, in a minor subset, granulomatous manifestations can also be seen.
Pathogenesis
The underlying pathophysiology still remains unknown, however, various theories have been proposed including sympathetic, hereditary, infectious (HSV, CMV, toxoplasmosis, rubella), and immunological theories. Of these, the infectious theory remains the most accepted till now, supported by the newer molecular diagnostic investigations like polymerase chain reactions (PCR). Amongst the various viruses implicated, recently, Mahendradas et al. reported the association of Chikungunya in FUS with the help of reverse transcriptase PCR assays 18]. However, a causative factor could not be established. The main etiologic agent in Europe the United States has been proven to be the rubella virus and CMV is a common cause in Singapore, Taiwan, and Japan.[12]. Other reported infectious aetiologies include HSV [11], toxocariasis[12] and toxoplasmosis[13]. Another widely accepted hypothesis is of immunological basis. Muhaya et al. found increased aqueous levels of interferon-gamma and interleukin – 10 (IL-10) in FUS as compared to idiopathic AU. This observation could help answer the presence of low-grade ocular inflammation [19].
Clinical features
The characteristic of FUS is the peculiarity of the KPs formed. They are translucent, white, fine to medium-sized, and stellate shaped, distributed throughout the corneal endothelium. These are thought to be an autoimmune response to corneal antigens and hence a diffuse distribution rather than the usual inferior distribution noted in other infectious uveitis. Chee et al. noted an association between CMV DNA and corneal endothelial precipitates in aqueous samples, further supporting the autoimmune theory [12]. Another sign is the depigmentation of the iris stroma leading to a depigmented iris color in the affected eye, termed as heterochromia. Inverse heterochromia is also documented when a similar loss of iris stroma occurs in blue irides, exposing the darker pigmented epithelium. Iris nodules, both Koeppe and Busacca nodules are also seen, mainly in dark irides,. Posterior synechiae are typically absent, however, neovascularization of iris and angles are consistent features of FUS. It is important to differentiate between dilated normal iris vessels and abnormal vascularization. Normal iris vessels are fine, straight, and does not cross the scleral spur, noted by gonioscopy. Usually, these vessels become prominent over an area of iris atrophy. Whereas, the neovascular network of blood vessels are fine, branching vessels crossing the scleral spur. These are filiform vessels and therefore, have a tendency for spontaneous hemorrhage whenever there is a sudden decrease of IOP, as could happen in any intraocular surgery like a cataract operation, or even AC paracentesis[20]. This special characteristic is known as the Amsler-Verrey or, more commonly, Amsler’s sign and is a useful differentiating feature from other uveitic entities [21]. Iris features and KPs characteristic look are important, however, the most diagnostic finding is the vitreous involvement. Most of the patients have ongoing inflammation in the vitreous and pars plana region for a long period of time giving rise to the rare complaints of floaters and blurring of vision. However, unlike intermediate uveitis, it does not cause cystoid macular edema
Management
Usually, the inflammation is mild and smoldering in nature. A short course of systemic corticosteroids in periods of exacerbation is sufficient mainly to control the vitreal inflammation. AU in case of exacerbation can be managed with mild topical corticosteroids. Snowballs are seen even in quiet, chronic cases, without any need for treatment. However, it is the treatment of complications that forms the major concern for these patients. The most common is the cataract, which is often considered a part of the diagnostic criteria. Small incision cataract surgery (SICS) and phacoemulsification are considered safer than extracapsular cataract extraction (ECCE), ensuring in-the-bag IOL implantation, reduced risk of sudden decompression of the chamber, and thereby, reducing chances of the postoperative spike of postoperative inflammation leading to the formation of posterior synechiae and posterior capsular opacification[22,23.24]. Whenever an intraoperative bleed happens due to sudden decompression, high-density viscoelastic agents can help to arrest the bleed. The second most common cause of vision loss in glaucoma with an incidence of 6.3-59% [25,26]. Secondary glaucoma encountered in FUS patients is usually recalcitrant to medical as well as laser therapies. Therefore, surgical management remains the mainstay. Trabeculectomy with intraoperative mitomycin-C (MMC) is the preferred procedure of choice. However, glaucoma draining devices are also found to be efficient, sometimes even superior, as compared to trabeculectomy [27,28,29,30].
Posner Schlossman Syndrome (PSS)
PSS presents as a unilateral mild anterior inflammation with a very high IOP. PSS has features very similar to FUS, like mild AC reaction and absence of posterior synechiae. However, various distinguishing features are the acute and recurrent nature of PSS with a very high IOP, often in the range of 40-60 mm of hg [31,32]. Hence, also known as the glaucomatocyclitic crisis. The mainstay of treatment is control of IOP with anti-glaucoma medications, and if required surgical management also. Association has been reported with various agents like helicobacter pyroli, CMV, HSV, and rubella. Few consider it to be the acute hypertensive form of CMV-AU [12].
Laboratory investigations
The diagnosis of VAU is usually clinical with characteristic systemic as well as ocular features. Systemic features of viral involvement in forms of blisters or rashes involving a particular dermatome as per the division of ophthalmic nerve involved and the strict adherence to midline form a major clue towards a viral etiology. However, in certain conditions like herpes zoster sine herpete, where rashes are particularly absent, ocular clues and molecular diagnostic assays form a major basis. Viral serology is only supportive evidence since the presence of IgG indicates a chronic infection and IgM indicates an active systemic infection without any proof of active ocular infection as well. Hence, molecular assays form the mainstay of diagnosis. PCR analysis of an aqueous sample, collected under sterile AC paracentesis, is most helpful if done in the acute inflammatory phase. PCR detects viral DNA, hence identify the particular causative virus. In a few cases, a combined involvement of more than one virus has also been reported [33]. Local antibody production can also be estimated by enzyme-linked immunosorbent assay (ELISA) to determine the Goldmann-Witmer coefficient (GWC). A GWC of more than 3 in immunocompetent and more than 2 in immunosuppressed is considered to be significant. However, antibody production usually takes 2 weeks and remains positive even in the chronic phase. Also, antibody production is significantly suppressed and prolonged in the case of immunosuppressed individuals. Still, a combination of GWC and PCR results improves the diagnostic yields. It is recommended to perform a quantitative PCR as a screening step, followed by a real-time PCR to confirm as well as quantify the viral load [34].
Management
HSV– AU
The role of antivirals for treatment is not as well-known as is established for HSV keratitis. However, various studies have noted a significant reduction in the duration of the acute attack and recurrences. Like other anterior uveitis, anterior segment inflammation is controlled with topical corticosteroids along with cycloplegics. Oral acyclovir (ACV) 400 mg five times a day is used for a minimum of 4 weeks as a loading dose followed by a maintenance therapy of ACV 400 mg twice daily is preferred, especially for severe or recurrent episodes. Valacyclovir (VAL), a prodrug of ACV, is preferred over ACV for its better bioavailability. A loading dose of 500 mg thrice daily followed by a maintenance dose of 500 mg twice daily is the usual regimen followed. Still unresolved is the duration of the maintenance therapy, but is advised to maintain for a long duration along with a low dose of topical corticosteroids. In severe or recurrent cases, intravenous ACV 15 mg/kg thrice daily is instituted. Cases non-responsive to oral as well as parenteral ACV are then treated with intravenous foscavir 40 mg/kg thrice daily and intravenous cidofovir 5 mg/kg once weekly [35,36].
HZV – AU
Similar to HSV, HZV-AU is also treated with ACV. A loading dose of 800 mg five times a day for 10 days followed by a maintenance dose of 400 mg three times a day is recommended for an acute episode. For chronic uveitis, the duration is longer, a minimum of 4 weeks or longer in immunocompromised patients is preferred [10]. VAL 1 gram or famciclovir 500 mg thrice daily are other available alternatives. Topical corticosteroids and cycloplegics are used to control the anterior segment inflammation.
CMV – AU
Anti-CMV antivirals available are ganciclovir (GCV) or valganciclovir (VGV). Oral VGV is preferred for its better bioavailability. A dose of 900 mg twice daily for 6 weeks followed by a maintenance dose of 450 mg twice daily for 6 weeks or more is the general practice. Other modes of the institution are topical ganciclovir gel (0.15%) five times a day for 3 months, intravitreal VGV (2mg/0.1 ml) weekly for 3 months, and intravitreal GCV implants. Intravitreal injections and implants are usually considered in unilateral cases with severe inflammation. However, efficiency is lower with higher relapse rates. A minimum of 15 months of combined therapy with systemic antivirals and intravitreal injections is recommended [37]. It is preferable to treat till negative PCR results are reported, wherever available [38].
Complications
Cataract and glaucoma are the most common complications encountered. Other complications are optic neuropathy, muscle paresis due to viral involvement.
Cataract
Either as a part of the disease spectrum like in FUS or as a complication of management, cataract develops in 28-35% of HSV, 27-30% of VZV, 23% of acute CMV, or PSS, and 75% of chronic CMV AU[5,7,12]. A quiescent period of three months to cataract operation is considered ideal in all cases except FUS where it is not mandatory to have a quiescent waiting period. Phacoemulsification and in-the-bag IOL placement is preferred. SICS is also an option, wherever phacoemulsification is not available. However, ECCE needs to be avoided as far as possible.
Glaucoma
18-54% of HSV, 30-40% VZV, 23% acute CMV, and 36% chronic CMV are noted to develop glaucoma in the course of the disease [5,7,12]. Unlike cataract management, glaucoma management is quite difficult with regards to its recalcitrant nature to various treatment modalities. Medical management usually fails. Oral carbonic anhydrase inhibitors are used only in severe IOP spikes as seen in PSS. Prostaglandin analogs are to be avoided. HSV and HZV associated glaucoma are better responsive as compared to CMV associated glaucoma. Nearly 60% of CMV AU end up requiring glaucoma surgery[37,39]. Trabeculectomy with MMC and anti-glaucoma valves are the usual methods of management.
Viral Posterior Uveitis (VPU)
VPU can be caused by various viruses including herpes viruses, measles, rubella, and arboviruses. VPU may occur as an isolated ocular disease or as a part of systemic illness. Viruses can enter by hematogenous spread or from a break in the blood-ocular barrier. Risk factors leading to infection or reactivation of latent infection leading to VPU is mainly the immunocompromised status of the patient, especially those with multiple comorbidities [37]. Studies by various authors concluded that the immune status also modifies the presentation of various viral retinopathies. Other risk factors are local immunosuppression by intraocular injections. Several cases of viral retinitis following subtenon corticosteroid injection have been reported [38].
In general, viral posterior uveitis causes retinitis, vasculitis, macular edema, vitritis, and mild anterior uveitis with a possibility of raised intraocular pressure. The most common viruses causing posterior uveitis are Cytomegalovirus (CMV), Herpes Simplex virus(HSV), and Varicella-Zoster virus (VZV). Diagnosis is usually clinical with supportive ancillary investigations, which includes laboratory investigations and imaging modalities. Various imaging modalities used are fundus fluorescein angiography (FFA), wide-field retinal imaging (e.g Optos), and optical coherence tomography (OCT). Laboratory investigations consist of viral serology and PCR. Similar to VAU, PCR has a similar role in VPU and the sensitivity and specificity of 80-84% and 97.4-100% respectively, has been noted [39, 40]. Serial PCR analysis helps analyze the disease course and monitor response to treatment. GWC, on the other hand, has little role in VPU as compared to its role in VAU [39]. Chorioretinal biopsy plays a pivotal role in posterior viral uveitis where the diagnosis cannot be made with other common investigations [41].VPU has more vision-threatening potential as compared to VAU. Common complications are cataract, glaucoma, band-shaped keratopathy, similar to VAU, in addition to other serious complications like CME and RD, epiretinal membrane, neovascularization, optic atrophy, and phthisis bulbi [42] We will be discussing in detail the various manifestations of VPU.
Acute Retinal Necrosis (ARN)
It was first described in 1971 by Urayama in young Japanese as an acute panuveitis with retinal necrosis and retinal detachment [37]. With an incidence of 1-2 million individuals per year, it has a bimodal age of distribution, peaking around the 2nd and 5th decades of life. It does not have any sex predilection. VZV is the most common cause of ARN and is responsible for about one-third of ARN cases [46]. ARN can also be caused by HSV and CMV. VZV or HSV-1 causes ARN in patients older than 25years whereas HSV-2 causes ARN in patients younger than 25 years [43]. Both HSV-1 and HSV-2 encephalitis can be a risk factor for the development of ARN[44, 45]. Therefore along with viral meningitis or encephalitis, the risk factors to develop ARN are younger age, pre-existing chorioretinal scar, trauma, and systemic corticosteroids [47].
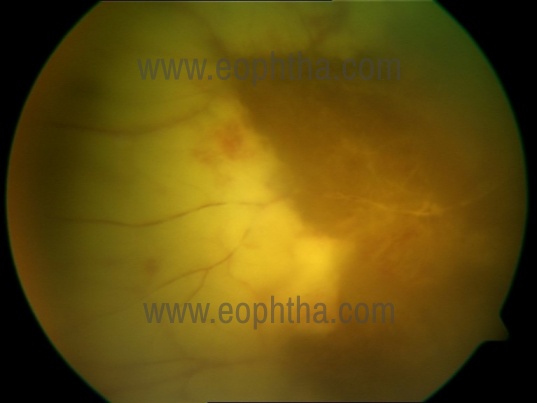
Fig 2. Acute retinal necrosis – extensive necrosis in the peripheral retina involving the retinal vessels
Clinical features
Redness, photophobia, pain, floaters, and blurring of vision are a few common symptoms with or without a recent history of infection with the herpes virus. It is usually unilateral in affection, however, bilateral involvement is reported in up to one-third of patients. Unilateral cases can subsequently affect the other eye too, hence a close watch has to be kept on the other eye [48]. Clinically it is characterized by a triad of prominent anterior chamber inflammation, intense vitritis, and occlusive retinal vasculitis. Other characteristic features are the scalloped wedge-shaped areas of peripheral necrotizing retinitis with a circumferential extension of the necrotic foci (Fig 2). These necrotic areas of the retina are highly prone to developing focal areas of full-thickness necrotic lesions to form a retinal break or hole.
Two phases of the infection described are – acute herpetic phase and the late cicatricial phase. In the acute herpetic phase, panuveitis with circumferential necrotizing retinitis, occlusive vasculitis with hemorrhages are the usual manifestations. The retinitis spreads rapidly, both circumferentially and centrally within few days with focal and confluent patches of necrotizing retinitis. Whereas, the late cicatricial phase is characterized by retinal atrophy, proliferative vitreoretinopathy, and serous or rhegmatogenous retinal detachment [49].
Diagnosis
Although the diagnosis of ARN is usually clinical, laboratory investigations help identify the causative virus. Viral antibodies are detected by various methods as complement fixation method, immunofluorescence, or enzyme-linked immunosorbent assay (ELISA) techniques. PCR can help identify the genome of the virus in the intra-ocular fluids. Ocular imaging plays an important role in identifying the extent of retinal involvement, neovascularization, and other complications like CME or vascular occlusion, vitreous hemorrhage (VH), and optic atrophy. FFA helps identify vasculitis, occlusion, or presence of any abnormal new vessels in the form of capillary non-perfusion areas and dye leakage, and OCT helping in identifying the presence of CME or any retinal hole formation [50].
OCT shows areas of inner retinal hyperreflectivity in areas of retinal necrosis with a disorganization of the retinal architecture in the acute phase. In the late cicatricial phase, there might be inner as well as outer retinal thinning within the areas of retinal necrosis with the resolution of earlier hyperreflectivity. This helps in monitoring early signs of retinal detachment (RD) [51].
Management
Antivirals are the mainstay of treatment. Owing to its severity and rapid progression, the standard of care is usually by intravenous (IV) administration of acyclovir (ACV) 10 mg/kg every 8 hours or 1500 mg/m2 per day for 5 to 10 days, followed by maintenance with oral acyclovir 800 mg 5 times daily for an additional 6 weeks. It is important to prevent the risk of the virus spreading to the other eye, and since most such spread occurs within six weeks, it appears to be rational to give a maintenance dose for at least 6 weeks. Newer antivirals as Valacyclovir (VAL) and famciclovir (FCV) have better bioavailability, having a systemic concentration similar to intravenous ACV and therefore can be used in cases where hospital admission is difficult [52]. Oral VAL 1 g 3 times daily or oral FCV 500 mg 3 times daily are the standard dose used. Higher doses of VAL up to 2 g 4times/day can also be used in severe cases.
The severity of the disease is classified according to the surface area affected– mild if less than 25% of the retinal area is involved, moderate if 25%–50%, and severe if more than 50% of the retinal surface area is involved [53]. Apart from systemic administration in form of oral or parenteral administration, the intravitreal route is also available in cases of severe retinitis or non-responding refractory cases to systemic antivirals. Intravitreal foscarnet 2.4 mg/0.1 mL and intravitreal ganciclovir 4 mg/0.1 mL are the available options [54, 55]. Systemic corticosteroids are to be added only after initiation of the antivirals as they may promote viral replication, at the dose of 1-1.5 mg/kg/day[56].
Surgical interventions are reserved for complications like a macular hole, VH, or RD. Prophylactic laser barrage is controversial with regard to its preventive role for RD. Laser photocoagulation does not prevent the progression of retinitis but forms a barrier posterior to the involved retina to form a stronger chorioretinal adhesion, hence preventing potential retinal detachment. However, strong evidence is lacking and prospective studies are required [57].
Progressive Outer Retinal Necrosis ( PORN)
As opposed to ARN, PORN is usually seen in immunocompromised individuals with a rapidly progressive destructive involvement of the outer retina. Hence, a history of organ transplantation, chemotherapy, HIV status, or idiopathic CD4 lymphocytopenia has to be considered
Clinical features
It is usually bilateral, with a male predominance affecting between 20-50 years of age [58]. The progressive blurring of vision, scotoma with the absence of pain or photophobia are typically seen. VZV is the usual virus associated with PORN, hence the presence of zoster blisters could be a clue towards it. Anterior uveitis is usually non-granulomatous and minimal with the presence of minimal or no vitreous involvement. Unlike ARN, retinal necrosis starts at the posterior pole spreading towards the peripheral retina. The retinal lesions are white, necrotic patches with perivascular lucency giving a characteristic “cracked mud” appearance (Fig 3). There is an absence of retinal vascular inflammation with minimal hemorrhage [59].

Fig 3. Progressive outer retinal necrosis – extensive outer retinal necrosis with sparing of the retinal vessels giving rise to a typical “cracked mud” appearance (Photo Courtesy: Dr. Jyotirmay Biswas)
Diagnosis
Retinal involvement in the form of well-demarcated, multifocal, deep areas of retinal necrosis in immunocompromised patients, supported with laboratory testing of viral serology, and PCR analysis of intraocular fluids establish the diagnosis. Assessment of immune status is mandatory for proper treatment. OCT shows total loss of retina layers corresponding to areas of retinal necrosis and helps distinguish from central retinal artery occlusion (CRAO) [60].
Management
Owing to its rapid progression, early aggressive treatment with antiviral therapy along with the institution of HAART therapy in cases of HIV-positive individuals is the mainstay of treatment. Oral corticosteroids help in reducing associated inflammation. Systemic antiviral therapy includes IV foscarnet 24 mg/mL 3 times/day or ganciclovir 5 mg/kg/day or cidofovir or acyclovir. PORN usually shows a poor response to IV acyclovir therapy alone, hence a combination therapy with intravitreal ganciclovir combined with IV acyclovir has a better response. After the initial loading dose, a maintenance dose of oral VAL 1 g 3 times/day or valganciclovir, or famciclovir usually suffice [58]. Surgical management is similar to that of ARN, with a controversial role of prophylactic laser photocoagulation in preventing RD.
CMV Retinitis (CMVR)
It is an opportunistic infection seen in severely immunosuppressed individuals. It is usually seen in HIV patients with a CD4 count of less than 50 cells/μL. However, CMVR may also manifest in immunocompetent individuals as well with some immune dysfunction as diabetes mellitus, use of any immunosuppressive agents like corticosteroids [61].
Clinical features
CMVR usually begins as a unilateral disease with progression towards a bilateral involvement through a hematogenous route [62, 63]. Males between 30 – 60 years are most commonly affected. It is usually asymptomatic with some patients complaining of blurred vision, floaters, and scotoma. Pain, redness, and photophobia are typically absent.
The classical findings are retinal hemorrhages with a whitish, granular appearance to the retina give rise to the characteristic “pizza pie” retinopathy (Fig 4a and 4b). It follows the retinal vasculature, spreading centrifugally. The ocular inflammatory response is minimal as it mainly affects immunosuppressed individuals [64].
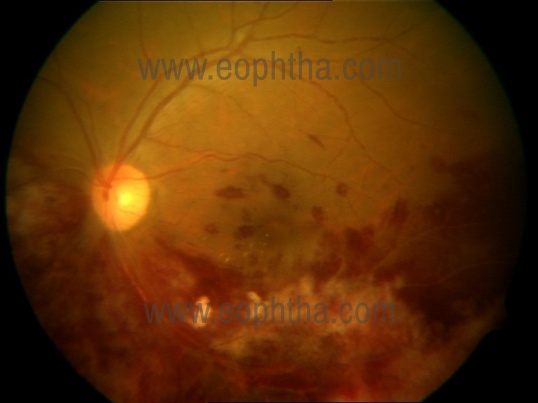
Fig 4 a. Active Pizza – pie appearance of CMV retinitis
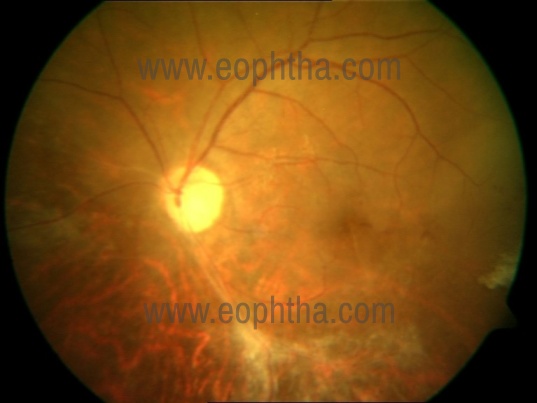
Fig. 4 b. As the retinitis resolves it leaves behind areas of the atrophic retina
In the acute stage, CMV has three patterns of retinal involvement as fulminant or edematous, indolent or granular, and exudative [65]. The fulminant type of retinal lesion consists of areas of retinal hemorrhages in the background of retinal necrosis. The granular form has granular satellite lesions with minimal or no hemorrhage. The exudative form has extensive vascular sheathing manifesting as frosted branch angiitis (Fig 5). As the disease heals it leaves behind large areas of necrosis and retinal tears. Eventually, there can be retinal and optic atrophy, retinal fibrosis, calcification, and sclerotic vessels. In patients with limited immune dysfunction (e.g diabetes), there can be the atypical presentation in the form of a combined panretinal occlusive vasculopathy and peripheral granular retinitis [61].
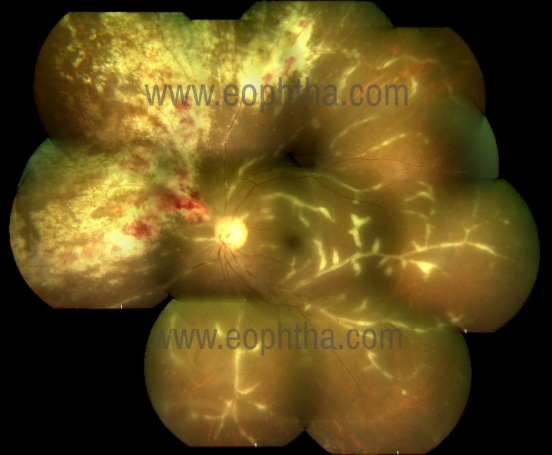
Fig 5. CMV retinitis with frosted branch angitis
Diagnosis
Diagnosis of CMVR is similar to that of PORN, basically clinical with supportive laboratory investigations and ocular imaging. PCR analysis of intraocular fluid helps in identifying the causative virus and also help in monitoring response to the therapy.
Management
Antivirals such as ganciclovir, valganciclovir, cidofovir, and foscarnet are used in the treatment of CMV retinitis because these antivirals competitively inhibit CMV DNA polymerase. However, the mainstay of treatment is addressing the immunodeficient state with the institution of HAART. It is important to note that HAART therapy could lead to immune recovery uveitis (IRU), reported in up to 63% of patients [66]. Therefore, it is recommended to delay HAART until the institution of antiviral therapy.
Non-necrotizing Retinitis (NNR)
NNR is a new disease entity that can affect any age group, with a slight male preponderance, unilateral in manifestation. It is usually associated with VZV (75%) with the rest 25% affected with HSV [67]. Ocular pain with the blurring of vision is the usual complaint. There is mild anterior chamber inflammation with vitreous inflammation as well. Retinal lesions are usually peripheral retinitis with diffuse occlusive retinal vasculitis. It progresses slowly without any necrosis. Diagnosis is similar to clinical features, ELISA, and PCR analysis. There is usually no response to systemic immunosuppressive agents. Systemic antivirals are the treatment of choice [68, 69]. Systemic antivirals include oral acyclovir 800 mg 5 times daily or oral valacyclovir 2–3 g/day are usually employed. Surgical management is similar to any other viral necrotizing retinitis. It usually has a good prognosis with less risk of CME and RD.
Dengue Retinitis
Dengue virus is a mosquito-borne, single positive-stranded RNAvirusof the family Flaviviridae. Five serotypes of thevirushave been found, all of which can cause the full spectrum of disease which can range from a nonspecific illness to fatal hemorrhagic fever.
Ocular complaints following Dengue usually occur after 1-2 weeks after the fever, myalgia and rashes. Patients complain of blurred vision and metamorphopsia. Typically there is chorioretinitis with hemorrhagic retinopathy usually involving the macula. There may be associated with retinal edema, cotton wool spots, and neuroretinitis [70]. The anterior chamber inflammation and vitritis are minimal.
Chikungunya Retinitis
Chikungunya is a mosquito-borne RNA virus (arbovirus) infection.
Ocular manifestations include mild to moderate anterior uveitis. The posterior segment shows retinitis with retinal edema and hemorrhages usually involving the posterior pole, choroiditis, neuroretinitis with mild vitritis [71].
Both Dengue and Chikungunya virus infections can be diagnosed by virus isolation, serological tests, and molecular techniques.
In both cases, early treatment with systemic steroids helps in the reduction of the inflammation. However, if there is retinal ischemia, the visual recovery may be poor.
Human Immunodeficiency Virus (HIV) retinitis
HIV is a single-stranded RNA retrovirus. The prevalence of HIV retinopathy appears to be lesser in India (6.28%) as compared to the Western countries (50%) probably because HIV patients do not undergo routine ocular examination In India [79]. Retina shows bilateral cotton wool spots, retinal hemorrhages and microaneurysms. Diagnosis is based on positive serology for HIV and the exclusion of other etiologies. Treatment with HAART improves the CD4 count and prevents progression to ocular opportunistic infections.
Other Viral retinitis
Measles- Measles is a single-stranded, enveloped (nonsegmented) RNA Morbillivirus from the Paramyxoviridae family. There is profound visual loss, usually 6–12 days after the appearance of the characteristic rash.
There is usually retinal edema, optic disc edema, retinal hemorrhages, and resolution optic atrophy and secondary pigmentary retinopathy with a “salt and pepper” appearance. In Subacute Sclerosing Pan Encephalitis usually, there is focal necrotizing retinitis or chorioretinitis. Ocular findings of retinitis very often precede the neurological symptoms in SSPE, usually within 1–8 months [78].There is no definitive treatment for measles retinopathy or SSPE and the role of oral corticosteroids is debatable
Rubella -Rubella is a single-stranded RNA Rubivirus of the Togaviridae family. Congenital rubella syndrome is characterized by a triad of auditory, ocular and cardiac defects. Ocular involvement includes cataracts, microphthalmos and pigmentary retinopathy. The retinopathy is described as a “salt and pepper” fundus in the posterior pole and macula with areas of depigmentation and hyperpigmentation.[72] The disc may have a waxy pallor. In acquired disease, it causes diffuse chorioretinitis with retinal detachment. The disc and retinal vessels may appear normal. Rarely rubella neuroretinitis has also been reported [73]. The retinopathy usually does not affect vision unless choroidal neovascularization develops in the macula. Systemic corticosteroids may be given in severe inflammation.
Rift valley virus - Rift Valley fever virus is a single-stranded RNA Phlebovirus of the Bunyaviridae family. It has been reported from Africa and Saudi Arabia. The fundus picture shows necrotizing retinitis and retinal edema, hemorrhages involving the posterior pole with occlusive vasculitis, optic disc edema, and mild vitritis. Treatment is mainly supportive with IV fluids, antimicrobials, blood transfusion, hemodialysis, or mechanical ventilation with 70% of patients becoming legally blind after the resolution of the retinitis[74].
West Nile virus -The most common ocular manifestation of West Nile virus infection is bilateral multifocal chorioretinitis, found in more than 80% of patients. There are linear clusters of whitish-yellow chorioretinal scars with a “target-like” appearance, following the course of the retinal nerve fibers and retinal vasculitis, optic neuritis, and minimal vitritis [75].
Ebola Virus - Ebola virus is an RNA virus from the Filoviridae family. It regained attention because of the re-emergence in West Africa in December 2013. There may be hypertensive anterior uveitis or posterior uveitis with vitreous opacities, multiple, peripheral chorioretinal scars with hypopigmented halos, and small intraretinal hemorrhages [76].
Zika Virus —Zika virus is a mosquito-borne Flavivirus that showed a re-emergence in human infection in 2015 in Brazil. Infants were born with microcephaly and were found to have macular pigment mottling and optic nerve hypoplasia. In acquired cases there is no report of posterior uveitis, however, there is one case of PCR – proven anterior uveitis [77].
Novel coronavirus 2019 (COVID -19)A novel coronavirus, a single-stranded RNA virus, designated as COVID-19 (coronavirus disease of 2019), emerged in Wuhan, China, at the end of 2019. Initial reports of anterior uveitis and retinitis in feline and murine models suggested that this virus was capable of producing a wide spectrum of ocular manifestations. Subsequently, anecdotal reports of anterior uveitis and retinitis in humans with COVID -19 have been published [80, 81, 82]. Acute anterior uveitis was seen in one patient with diffuse pigmentary and whitish precipitates over the anterior lens capsule [80]. OCT was used to demonstrate retinal changes in patients with COVID-19, which included hyperreflectivelesions at the level of ganglion cell and inner plexiform layers more prominently at the papillomacular bundle, cotton wool spots, and microhemorrhages.
References:
- Chang JH, Wakefield D. Uveitis: a global perspective. Ocul Immunol Inflamm. 2002; 10:263–79.
- Rathinam SR, Namperumalsamy P. Global variation and pattern changes in epidemiology of uveitis. Indian J Ophthalmol. 2007; 55:173–183.
- Biswas J, Narain S, Das D, Ganesh SK. Pattern of uveitis in a referral uveitis clinic in India.Int Ophthalmol1996-97; 20:223-28.
- Young RC, Hodge DO, Liesegang TJ, Baratz KH. Incidence, recurrence, and outcomes of herpes simplex virus eye disease in Olmsted County, Minnesota, 1976–2007: the effect of oral antiviral prophylaxis. Arch Ophthalmol. 2010; 128:1178–1183
- Miserocchi E, Fogliato G, Bianchi I, Bandello F, Modorati G. Clinical features of ocular herpetic infection in an Italian referral center. Cornea. 2014;33(6):565–570.
- Van der Lelij A, Ooijman FM, Kijlstra A, Rothova A. Anterior uveitis with sectoral iris atrophy in the absence of keratitis: a distinct clinical entity among herpetic eye diseases. Ophthalmology. 2000;107(6):1164–1170.
- Wensing B, Relvas LM, Caspers LE, et al. Comparison of rubella virus- and herpes virus-associated anterior uveitis: clinical manifestations and visual prognosis. Ophthalmology. 2011;118(10):1905–1910
- Wareham DW, Breuer J. Herpes zoster. BMJ. 2007;334(7605): 1211–1215.
- Liesegang TJ. Herpes zoster virus infection. Curr Opin Ophthalmol. 2004;15(6):531–536.
- Kido S, Sugita S, Horie S, et al. Association of varicella zoster virus load in the aqueous humor with clinical manifestations of anterior uveitis in herpes zoster ophthalmicus and zoster sine herpete. Br J Ophthalmol. 2008;92(4):505–508.
- Daicker B. Cytomegalovirus panuveitis with infection of corneo-trabecular endothelium in AIDS. Ophthalmologica. 1988;197(4):169–175
- Chee SP, Jap A. Presumed fuchs heterochromic iridocyclitis and PosnerSchlossman syndrome: comparison of cytomegalovirus-positive and negative eyes. Am J Ophthalmol. 2008;146(6):883–889.
- Hwang YS, Shen CR, Chang SH, et al. The validity of clinical feature profiles for cytomegaloviral anterior segment infection. Graefes Arch Clin Exp Ophthalmol. 2011;249:103–110.
- Abano J.M., Galvante P.R., Siopongco P., Dans K., Lopez J. Review of epidemiology of Uveitis in Asia: pattern of Uveitis in a tertiary hospital in the Philippines. Ocul Immunol Inflamm.2017;25:S75-S80
- Mahendradas P., Shetty R., Malathi J.,Madhavan H.N. Chikungunya virus iridocyclitis in Fuchs' heterochromic iridocyclitis. Indian J Ophthalmol.2010;58:545-547
- Muhaya M., Calder V., Towler H.M., Shaer B., McLauchlan M., Lightman S. Characterization of T cells and cytokines in the aqueous humour (AH) in patients with Fuchs' heterochromic cyclitis (FHC) and idiopathic anterior uveitis (IAU). Clin Exp Immunol.1998;111:123-128
- Srinivasan S., Lyall D., Kiire C. Amsler-Verrey sign during cataract surgery in Fuchs heterochromic uveitis. BMJ Case Rep.2010;2010
- Bloch-Michel E., Frau E., Chhor S., Tounsi Y. Amsler's sign associated significantly with Fuch's heterochromic cyclitis (FHC). Int Ophthalmol.1995;19:169-171
- Bhargava R., Kumar P., Sharma S.K., Arora Y. Phacoemulsification versus manual small incision cataract surgery in patients with Fuchs heterochromic iridocyclitis. Asia Pac J Ophthalmol (Phila).2016;5:330-334
- Bhargava R., Kumar P., Bashir H., Sharma S.K., Mishra A. Manual suture less small incision cataract surgery in patients with uveitic cataract. Middle East Afr J Ophthalmol.2014;21:77-82
- Keles S., Ondas O., Ates O. et al. Phacoemulsification and core vitrectomy in Fuchs' heterochromic uveitis. Eurasian J Med.2017;49:97-101
- Norrsell K., Sjodell L. Fuchs' heterochromic uveitis: alongitudinal clinical study. Acta Ophthalmol.2008;86:58-64
- Tugal-Tutkun I., Guney-Tefekli E., Kamaci-Duman F., Corum I. Across-sectional and longitudinal study of Fuchs uveitis syndrome in Turkish patients. Am J Ophthalmol.2009;148:510-515
- Monteiro L.G.O.F. Ciclite heterocromica de Fuchs. in:Orefice F.Uvéltes—Cĺınica e Cirúrgica. 1st ed.Cultura Médica,Rio de Janeiro2000:796-806(2000)
- Kok H., Barton K. Uveitic glaucoma. Ophthalmol Clin North Am.2002;15:375-387
- Esfandiari H., Loewen N.A., Hassanpour K. et al. Fuchs heterochromic iridocyclitis-associated glaucoma: a retrospective comparison of primary Ahmed glaucoma valve implantation and trabeculectomy with mitomycin C. F1000Res.2018;7:876
- Voykov B., Doycheva D., Deuter C., Leitritz M.A., Dimopoulos S., William A. Outcomes of Ahmed Glaucoma Valve Implantation for Glaucoma Secondary to Fuchs Uveitis Syndrome. Ocul Immunol Inflamm.2017;25:760-766
- Megaw R. Agarwal P.K. Posner-Schlossman syndrome. Surv Ophthalmol.2017;62:277-285
- Osner A., Schlossman A. Syndrome of unilateral recurrent attacks of glaucoma with cyclitic symptoms. Arch Ophthal.1948;39:517-535
- Kasetsuwan N, Tangmonkongvoragul C. Concomitant herpes simplex virus and cytomegalovirus endotheliitis in immunocompetent patient. BMJ Case Rep. 2013; 9:2013
- Sugita S, Shimizu N, Watanabe K, et al. Use of multiplex PCR and real-time PCR to detect human herpes virus genome in ocular fluids of patients with uveitis. Br J Ophthalmol. 2008; 92(7):928–932.
- The Herpetic Eye Disease Study Group. A controlled trial of oral acyclovir for the prevention of stromal keratitis or iritis in patients with herpes simplex virus epithelial keratitis. The Epithelial Keratitis Trial. Arch Ophthalmol. 1997; 115(6):703–712.
- Cobo LM, Foulks GN, Liesegang T, et al. Oral acyclovir in the treatment of acute herpes zoster ophthalmicus. Ophthalmology. 1986; 93:763–770
- Sobolewska B, Deuter C, Doycheva D, Zierhut M. Long-term oral therapy with valganciclovir in patients with Posner-Schlossman syndrome. Graefes Arch Clin Exp Ophthalmol. 2014;252(1):117–124.
- Chee SP, Jap A. Cytomegalovirus anterior uveitis: outcome of treatment. Br J Ophthalmol. 2010;94(12):1648–1652.
- Accorinti M, Gilardi M, Pirraglia MP, et al. Cytomegalovirus anterior uveitis: long-term follow-up of immunocompetent patients. Graefes Arch Clin Exp Ophthalmol. 2014;252(11):1817–1824.
- Shah AM, Oster SF, Freeman WR. Viral retinitis after intravitreal triamcinolone injection in patients with predisposing medical comorbidities. Am J Ophthalmol. 2010; 149(3):433–40.
- D’alessandro L, Bottaro E. Reactivation of CMV retinitis after treatment with subtenon corticosteroids for immune recovery uveitis in a patient with AIDS. Scand J Infect Dis. 2002; 34(10):780–2.
- Rothova A, Boer JHD, Loon NHTDV, et al. Usefulness of aqueous humor analysis for the diagnosis of posterior uveitis. Ophthalmology. 2008; 115(2):306–11
- Scheepers MA, Lecuona KA, Rogers G, Bunce C, Corcoran C, Michaelides M. The value of routine polymerase chain reaction analysis of intraocular fluid specimens in the diagnosis of infectious posterior uveitis. ScientificWorldJournal. 2013; 2013:1–8.
- Rutzen AR, Ortega-Larrocea G, Dugel PU, et al. Clinicopathologic study of retinal and choroidal biopsies in intraocular inflammation. Am J Ophthalmol. 1995; 119(5):597–611
- Yoser SL, Forster DJ, Rao NA. Systemic viral infections and their retinal and choroidal manifestations. Surv Ophthalmol. 1993; 37(5):313–52.
- Ganatra JB, Chandler D, Santos C, Kuppermann B, Margolis TP. Viral causes of the acute retinal necrosis syndrome. Am J Ophthalmol. 2000; 129(2):166–72.
- Kianersi F, Masjedi A, Ghanbari H. Acute retinal necrosis after herpetic encephalitis. Case Rep Ophthalmol. 2010; 1(2):85–9
- Stevens J, Herbert E, Johnston S, Fathers E. 075 herpes simplex retinitis in a patient with prior herpes simplex encephalitis. J Neurol Neurosurg Psychiatry. 2012; 83(3):e1.
- Wong RW, Jumper JM, Mcdonald HR, et al. Emerging concepts in the management of acute retinal necrosis. Br J Ophthalmol. 2013; 97(5):545–52.
- Cochrane TF, Silvestri G, Mcdowell C, Foot B, Mcavoy CE. Acute retinal necrosis in the United Kingdom: results of a prospective surveillance study.
- Roy R, Pal BP, Mathur G, Rao C, Das D, Biswas J. Acute retinal necrosis: clinical features, management and outcomes – a 10 year consecutive case series. Ocul Immunol Inflamm. 2014; 22(3):170–4.
- Lau CH, Missotten T, Salzmann J, Lightman SL. Acute retinal necrosis: features, management, and outcomes. Ophthalmology. 2007; 114(4):756–62
- Gupta V, Al-Dhibi HA, Arevalo JF. Retinal imaging in uveitis. Saudi J Ophthalmol. 2014; 28(2): 95–103
- Ohtake-Matsumoto A, Keino H, Koto T, Okada AA. Spectral domain and swept source optical coherence tomography findings in acute retinal necrosis. Graefes Arch Clin Exp Ophthalmol. 2015; 253(11):2049–51.
- Aizman A, Johnson MW, Elner SG. Treatment of acute retinal necrosis syndrome with oral antiviral medications. Ophthalmology. 2007; 114(2):307–12
- Meghpara B, Sulkowski G, Kesen MR, Tessler HH, Goldstein DA. Long-term follow-up of acute retinal necrosis. Retina. 2010; 30(5):795–800.
- Kishore K, Jain S, Zarbin MA. Intravitreal ganciclovir and dexamethasone as adjunctive therapy in the management of acute retinal necrosis caused by Varicella zoster Virus. Ophthalmic Surg Lasers Imaging. 2011; 42:e87–90
- Luu KK, Scott IU, Chaudhry NA, Verm A, Davis JL. Intravitreal antiviral injections as adjunctive therapy in the management of immunocompetent patients with necrotizing herpetic retinopathy. Am J Ophthalmol. 2000; 129(6):811–3.
- Weller JM, Bergua A, Mardin CY. Retinopathy in a patient with acute Epstein–Barr virus infection. Retin Cases Brief Rep. 2015; 9(1):72–7.
- Park JJ, Pavesio C. Prophylactic laser photocoagulation for acute retinal necrosis. Does it raise more questions than answers? Br J Ophthalmol. 2008; 92(9):1161–2.
- Tseng CC, Chen SN, Hwang JF, Lin CJ, Chen HS. Progressive outer retinal necrosis associated with occlusive vasculitis in acquired immunodeficiency syndrome. J Formos Med Assoc. 2015; 114(5):469–72
- Khot A, Dignan F, Taylor S, Potter M, Cubitt D, Treleaven JG. Another case of PORN (bilateral progressive outer retinal necrosis) after allogeneic stem cell transplantation. Bone Marrow Transplant. 2006; 37:113–4.
- Almony A, Dhalla MS, Feiner L, Shah GK. Macular optical coherence tomography findings in progressive outer retinal necrosis. Can J Ophthalmol. 2007; 42(6):881.
- Schneider EW, Elner SG, van Kuijk FJ, et al. Chronic retinal necrosis: cytomegalovirus necrotizing retinitis associated with panretinal vasculopathy in non-HIV patients. Retina. 2013; 33(9):1791–9.
- Kempen JH, Jabs DA, Wilson LA, Dunn JP, West SK. Incidence of cytomegalovirus (CMV) retinitis in second eyes of patients with the acquired immune deficiency syndrome and unilateral CMV retinitis. Am J Ophthalmol. 2005; 139(6):1028–34.
- Kuo IC, Kempen JH, Dunn JP, Vogelsang G, Jabs DA. Clinical characteristics and outcomes of cytomegalovirus retinitis in persons without human immunodeficiency virus infection. Am J Ophthalmol. 2004; 138(3):338–46.
- Pathanapitoon K, Tesavibul N, Choopong P, et al. Clinical manifestations of cytomegalovirusassociated posterior uveitis and panuveitis in patients without human immunodeficiency virus infection. JAMA Ophthalmol. 2013; 131(5):638.
- Vrabec TR. Posterior segment manifestations of HIV/AIDS. Surv Ophthalmol. 2004; 49(2):131– 57
- Kempen JH, Min YI, Freeman WR, et al. Risk of immune recovery uveitis in patients with AIDS and cytomegalovirus retinitis. Ophthalmology. 2006; 113(4):684–94.
- Wensing B, de Groot-Mijnes JD, Rothova A. Necrotizing and nonnecrotizing variants of herpetic uveitis with posterior segment involvement. Arch Ophthalmol. 2011; 129(4):403.
- Albert K, Masset M, Bonnet S, Willermain F, Caspers L. Long-term follow-up of herpetic nonnecrotizing retinopathy with occlusive retinal vasculitis and neovascularization. J Ophthal Inflamm Infect. 2015; 5(1):6
- Bodaghi B, Rozenberg F, Cassoux N, Fardeau C, Lehoang P. Nonnecrotizing herpetic retinopathies masquerading as severe posterior uveitis. Ophthalmology. 2003; 110(9):1737–43.
- Gupta P, Jain C, Aggarwal A, Gupta SC. Dengue fever presenting with macular hemorrhages. Retin Cases Brief Rep. 2011; 5(3):213–8. [PubMed: 25390167]
- Mahendradas P, Avadhani K, Shetty R. Chikungunya and the eye: a review.J Ophthalmic Inflamm Infect. 2013;3(1):35. Published 2013 Feb 11. doi:10.1186/1869-5760-3-35
- . Collis WJ, Cohen DN. Rubella retinopathy: a progressive disorder. Arch Ophthalmol. 1970; 84(1): 33–5. [PubMed: 5423604]
- Hayashi M, Yoshimura N, Kondo T. Acute rubella retinal pigment epitheliitis in an adult. Am J Ophthalmol. 1982; 93(3):285–8. [PubMed: 7072790
- Al-Hazmi A, Al-Rajhi AA, Abboud EB, et al. Ocular complications of Rift Valley fever outbreak in Saudi Arabia. Ophthalmology. 2005; 112(2):313–8. [PubMed: 15691569]
- Khairallah M, Yahia SB, Attia S, Zaouali S, Ladjimi A, Messaoud R. Linear pattern of West Nile Virus-associated chorioretinitis is related to retinal nerve fibres organization. Eye. 2006; 21(7): 952–5. [PubMed: 16628235]
- Kibadi K, Mupapa K, Kuvula K, et al. Late ophthalmologic manifestations in survivors of the 1995 Ebola virus epidemic in Kikwit, Democratic Republic of the Congo. J Infect Dis. 1999; 179(Suppl 1):S13–4. [PubMed: 9988158]
- Furtado JM, Espósito DL, Klein TM, Teixeira-Pinto T, Fonseca BAD. Uveitis associated with Zika virus infection. N Engl J Med. 2016; 375(4):394–6. [PubMed: 27332784]
- Shah A, Rajesh B,Biswas J. Retinitis as the presenting feature of subacute sclerosing panencephalitis in an Indian male: A case report. Indian J Ophthalmol; 2018 Oct; 66(10): 1491–1493
- Sudharshan S, Kaleemunnisha S, Banu AA, Shrikrishna S, George AE, Babu BR, Devaleenal B, Kumarasamy N, Biswas J. Ocular lesions in 1,000 consecutive HIV-positive patients in India: a long-term study. J Ophthalmic Inflamm Infect. 2013 Jan 3;3(1):2.
-
Mazzotta C, Giancipoli E. Anterior Acute Uveitis Report in a SARS-CoV-2 Patient Managed with Adjunctive Topical Antiseptic Prophylaxis Preventing 2019-nCoV Spread Through the Ocular Surface Route.Int Med Case Rep J. 2020;13:513-520
https://doi.org/10.2147/IMCRJ.S260252 - Bettach E, Zadok D, Weill Y, Brosh K, Hanhart J. Bilateral anterior uveitis as a part of a multisystem inflammatory syndrome secondary to COVID-19 infection [published online ahead of print, 2020 Jun 27].J Med Virol. 2020;10.1002/jmv.26229. doi:10.1002/jmv.26229
-
Paula M Marinho, Allexya A A Marcos, André C Romano, Heloisa Nascimento, Rubens Belfort. Retinal findings in patients with COVID-19, The Lancet, Volume 395, Issue 10237,2020,Page 1610,ISSN0140-6736,https://doi.org/10.1016/S0140-6736(20)31014-X.
(http://www.sciencedirect.com/science/article/pii/S014067362031014X

