Introduction
First described in 1942, retinopathy of prematurity (ROP) is a vasoproliferative retinopathy affecting premature infants of low birth weight. As per the original description by Terry et al, this disease was labelled as retrolental fibroplasia (RLF) based on the hypothesis that it involved the proliferation of the embryonic hyaloid system[1] But Owens and Owens later described that in these infants, the hyaloid system was normal at birth and the RLF developed postnatally.[2] The term “retinopathy of prematurity” was coined later as the disease pathogenesis and clinical presentation got better understood. [3]In the 1950s, the association between supplemental oxygen and ROP pathogenesis was first established by controlled studies done in the neonatal intensive care units. {4,5]The spectrum of ROP ranges from mild cases that may resolve spontaneously with no visual impairment to advanced cases with bilateral irreversible blindness within the first few months of life.
Epidemiology
The first ROP epidemic was observed in the 1940s and 1950s in Europe and North America with the advent of unmonitored oxygen supplementation to premature infants with respiratory distress.[6] Subsequent restriction in the of oxygen use led to a drastic decrease in the ROP incidence in these countries but also led to increased morbidity and mortality in these low birth weight neonates due to other associated systemic problems like respiratory distress syndrome(RDS) and neurological disorders. The second epidemic of ROP occurred in the early 1970s with the advent of neonatal intensive care units (NICUs) in developed countries and improved neonatal survival rates and was controlled with improving standards of NICU care and the emergence of effective retinal ablation techniques, first in form of cryotherapy and thereafter, laser photocoagulation.[7,8] The third epidemic, currently seen in India and other developing countries like China, Mexico, Indonesia, Brazil, and Thailand is due to the combination of factors seen in the first and second epidemics. On one side, there is a better survival rate of preterm babies and on the other, the regulation of oxygen use is not universally followed.[9,10] In India, older and bigger babies with gestational age > 34 weeks and birthweight > 1500g have been also been observed to have a high incidence of severe ROP, largely due to varying standards of neonatal care and non-availability of an adequate number of a trained ophthalmologistfor effective screening.[11-13]Charan et al reported an overall incidence of any ROP of 47% in babies <1700g, with 13% requiring treatment.[13]
According to an estimate by Zin et al, the number of children projected to get potentially blind from ROP in India alone is likely to increase to 18000 every year unless widespread screening and treatment strategies are implemented.[14]As per a recent report from WHO, almost 15 million babies are born premature annually; nearly 1 in 10 babies are born premature worldwide.[15]Blencowe et al. estimated that nearly 32,200 infants became blind or visually impaired globally from ROP in 2010.[16]Dutta et al. suggested a high rate of prematurity in India (13% babies are born premature); improved survival of preterm babies; unrestricted use of oxygen; and lack of uniform ROP-screening protocols as a cause of ‘heavier’ and ‘more mature’ infants developing severe blinding stage 5 ROP.[17]
Pathogenesis:
To understand the pathogenesis of ROP it is important to first understand the normal retinal vascular development.
Normal Vasculogenesis of retina:
The Vascular supply network for the Retina consists of two main components.
1. The choroidal vessels that supply the outer retinal layers
2. The retinal vessels that supply the inner retinal layers
The Vascular development of choroid is complete by the third month of gestation but retinal vascular growth is still incomplete in a premature child. [18]The vascular development of retina begins by the 16th week of gestation. These developing retinal blood vessels reach the nasal ora serrata by 32-36 weeks and the temporal ora by 39-41 weeks. Thus, a premature child is born with incomplete peripheral vascularization; the area of avascular retina depending approximately upon the gestational age of the child.
Retinal vascular development occurs in two phases.
Phase 1: Vasculogenesis It is the process of de novo vessel development from endothelial precursor cells that form chords and then eventually form a lumen to form blood vessels.The primitive plexuses are thus formed from precursor cells and are not VEGF dependent.[18]
Phase 2: Angiogenesis It is the formation of blood vessels by sprouting from pre-existing vessels. These vessels are formed from pre-existing vasculature and are VEGF dependent[18.19]
It is believed that vasculogenesis forms most of the major retinal vessels of the developing human retina, while angiogenesis forms the remaining capillary network around the fovea and in the deeper and peripheral retina.[20]
Abnormal Vasculogenesis in ROP
The disease process for the development of ROP occurs in two phases; an initial phase of vessel obliteration followed by the second phase of vessel proliferation.
Phase 1: Hyperoxia-vasocessation (vaso-obliterative) phase From birth to postmenstrual age of 30 -32 wks
In humans, the retina develops in utero where there is low tissue oxygen. The metabolic demands of the developing retina are more than the oxygen supplied by the fetal choroidal circulation resulting in “physiologic hypoxia”.[21] In addition to maternally derived factors, that stimulate angiogenesis, this hypoxia triggers the release of vasoactive factors, like insulin-like growth factor (IGF)-1, vascular endothelial growth factor (VEGF), and erythropoietin.[22]
With birth, the environment for the retinal development now changes from a low oxygen tension intrauterine environment to a relatively high oxygen tension environment postnatally. A rise in PaO2 from 30–35 mmHg to 55–80 mmHg causes a decrease in the hypoxia triggered VEGF and loss of placental and maternal growth factors due to premature birth.[23]Exposure to supplemental oxygen, which is required to treat associated respiratory distress syndrome (RDS) in premature low birth weight babies, further suppresses retinal growth factors that are already compromised due to preterm birth and poor nutrition. This coupled with a lack of autoregulation of the choroidal circulation in premature babies results in movement of the oxygen from the choroidal to retinal circulation. This retinal hyperoxia and downregulation of growth factors result in phase 1 of vaso-obliteration of existing vessels, retraction of the normal developing retinal vessels, and cessation of the retinal vessel migration (Figure 1). An increase in the oxygen levels also increases the reactive oxygen free radicals, further increasing the damage.[23]
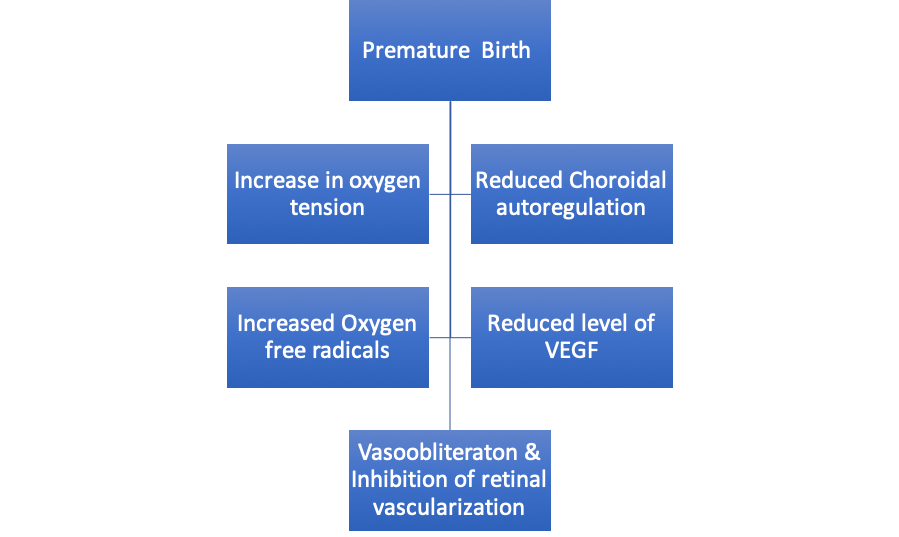
Figure 1: Flow chart depicting the sequence of events leading to phase 1 (vasocessation) of ROP pathogenesis.
Phase 2 of ROP: Relative hypoxia – revascularization (vasoproliferative) phase: begins around 32-34 weeks
Phase 2 is hypoxia driven. In this phase, the infant is weaned from supplemental oxygen. Also because of cessation of the retinal vessels’ growth, the peripheral retina now becomes a hypoxic and increased release of growth factors like VEGF, an Insulin-like growth factor is seen. This up-regulation stimulates neovascularization at the border between vascular and avascular retina (Figure 2), which leads to retinal hemorrhages, vitreous hemorrhage, fibrosis, and tractional retinal detachment leading to visual impairment.[18]
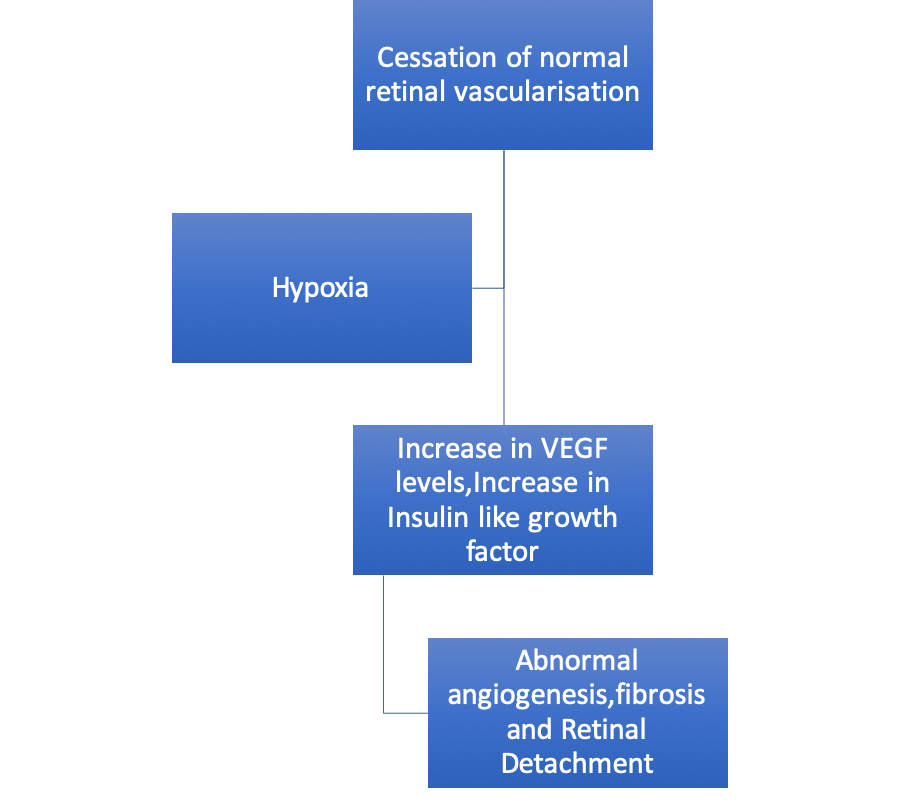
Figure 2: Flow Chart depicting Phase 2 (hypoxia-revascularization) of ROP
Risk Factors For ROP:
Definite risk factors include prematurity / lower gestational age, low birth weight, and supplemental oxygen administration.The incidence and severity of ROP are closely related to lower birth weight and lower postconceptional age. The duration of supplemental oxygen therapy is also an independent risk factor for the incidence and severity of ROP. [4-7,24]
Other risk factors include blood transfusions, failure to gain weight, respiratory distress syndrome(RDS), intermittent hypoxia, intra-ventricular hemorrhage (IVH), anaemia, multiple apnoeic spells, sepsis, hypercarbia and acidosis. Fetal hemoglobin has a greater affinity for oxygen than adult hemoglobin. Thus, a transfusion of adult hemoglobin could generate possible hyperoxia due to increased oxygen delivery to tissues.[25]Also, an increase in free radicals is seen after transfusions. The premature infant has inadequate concentrations of antioxidants at birth, as well as an impaired synthesis of antioxidants in response to hyperoxia and is, therefore, especially susceptible to reactive oxygen species(ROS) induced damage. This can lead to an increased risk for the development of ROS-induced diseases of the newborn, including ROP.[26]
Predictive models depending upon the presence of one or more risk factors have been developed to identify the neonates at risk of developing ROP.[27,28] One such model is the WINROP (Weight, Insulin-like growth factor-1, Neonatal Retinopathy of prematurity Model).[27] However additional research is required before their widespread use is advocated.[28
Role Of Oxygen
Historically, “excess oxygen” exposure was presumed to initiate the injury to the developing blood vessels in the immature retina, leading to endothelial cell death.[29]VEGF produced by the avascular retina has been incriminated in the development of ROP.
To evaluate the role of oxygen, STOP ROP study (Supplemental Therapeutic Oxygenfor Prethreshold ROP)was conducted.[30] It was a multicentral trial in which the premature infants with pre-threshold disease and median pulse oximetry less than 94% saturation were randomly assigned to conventional oxygen with pulse oximetry targeted at 89–94% saturation or supplemental oxygen with pulse oximetry targeted at 96–99% saturation. The study found that, once the ROP was established, raising the oxygen saturation mildly did not worsen the ROP, but it was not of any clear benefit as well. Current guidelines suggest the implementation of the “Oxygen with Love” (OWL) strategy for the prevention of ROP in premature neonates in NICU care.[31-33]For infants <28 weeks gestation, oxygen saturation (SpO2) target range of 85 – 93% is recommended from birth to 36 weeks’ postmenstrual age and a standardized bedside oximeter alarm limit is set at 80 – 95%. For infants born at 28-34 weeks of gestation, the SpO2 target range of 90-95% is recommended with the oxygen saturation alarm limits set at 85-98%.
Clinical Presentation:
Clinical presentation of ROP was first classified according to the international classification system of ROP(ICROP) first established in 1984[34]and later expanded in 1987.[35] Few modifications were later proposed in 2005.[36] It uses 3 parameters to describe ROP- the location (zones of involvement), the extent, as well as its stages.
ZONES OF RETINAL INVOLVEMENT IN ROP
The retina is divided into 3 concentric anterior-posterior zones that are centered on the optic disc (Figure 3). This is in contrast to the conventional retinal drawings, which mark the macula as the center and disc towards the nasal side. This difference is because of the natural course of development of the retinal vessels is from the disc towards the periphery.
- Zone I includes the posterior pole and is defined as a circle, centered on the disc, whose radius is twice the distance from the disc to the macula. It subtends an arc of about 60°. During indirect ophthalmoscopy by a 25 or 28 D lens if the disc is kept at the nasal end of the lens then the temporal extent of the retina as seen on the temporal end of the lens is the limit of the zone 1. Any disease within this area is marked as zone 1 ROP.
- Zone II extends centrifugally from the edge of zone I to the nasal ora serrata. Temporally this boundary corresponds approximately to the anatomic equator. Any disease if present nasally is termed a zone I or zone 2 disease.
- Zone III is the remaining temporal crescent of retina anterior to zone II. Zone III is the last zone to become vascularized, being farthest from the disc. There is no zone III nasally; so clinically ROP is classified as zone II if there is active ROP or immature vessels in the nasal retina beyond zone 1.
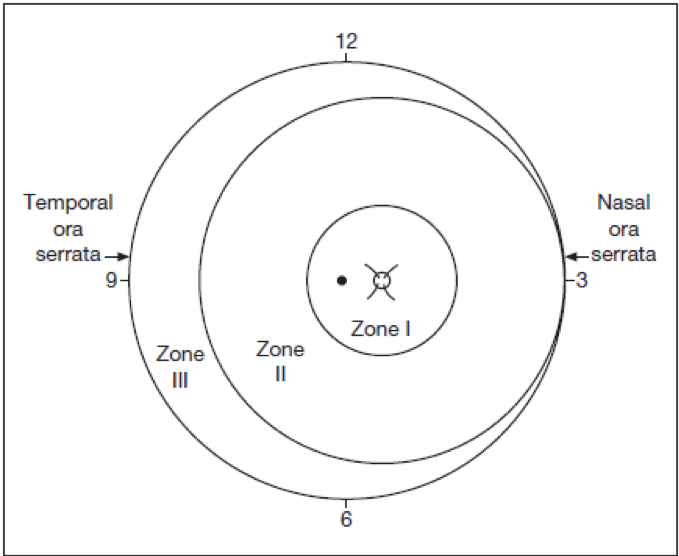
Figure 3: Zones of ROP in a Right eye.
STAGING OF ROP
The evolution of active ROP is clinically divided into 5 stages
Stage 1: Demarcation Line
The presence of a demarcation line is the first visible ophthalmoscopic sign of ROP. It is a flat, thin, definite, white line lying within the plane of the retina, separating the avascular retina anteriorly from the vascularized retina posteriorly (Figure 4a). This may or may not be accompanied by an abnormal branching in the form of frequent branching or straightening of vessels leading up to the whitish demarcation line. This stage can progress to stage II or regress to normal vascularization within several days to weeks.
Stage 2: Ridge
The ridge is the hallmark of stage II ROP where the demarcation line of stage I becomes more prominent with an increase in height and width. It is also seen to extend above the plane of the retina (Figure 4b). The ridge may be white or pink in color. Small whitish tufts or buds of new vessels, also called “popcorn lesions” may be seen lying above the retinal surface just posterior to the ridge (Figure 5). These may be precursor of new vessels. Though not all these popcorn lesions progress to form new vessels, close follow up of these lesions is necessary. “Popcorn lesion” per se is not considered indication for treatment. The ridge results from the proliferation of endothelial cells with some evidence of organization into recognizable vascular channels.[37]
Stage 3: Extraretinal Fibrovascular Proliferation
Extraretinal fibrovascular proliferation is the diagnostic feature of Stage 3 ROP. These are seen to extend into the ridge as well as from the ridge into the vitreous cavity. Traction on these active and fragile new vessels can cause vitreous hemorrhage (Figure 4c). This stage 3 can be subdivided into mild, moderate and severe depending upon the extent of the fibrovascular tissue infiltrating into the vitreous.
Stage 4: Partial Retinal Detachment
Contraction of the fibrovascular tissue on the ridge may result in tractional retinal detachments. Stage 4 comprises partial tractional retinal detachment which may be present in addition to findings seen in stage 3. It can be further subdivided into 2 groups:
Stage 4A: Extrafoveal retinal detachment
Partial tractional detachments not involving the macula are included in Stage 4 A (Figure 4d). These detachments are typically tractional with a concave configuration and mostly circumferentially oriented. These tractional detachments most commonly start at the area of maximal adhesions between the fibrovascular proliferation and the retina in any zone and extent circumferentially. Progression of the disease is marked by the increasing height of the retinal detachment due to continuous contraction of the fibrous tissue, extending both posteriorly and anteriorly.
Stage 4B: Partial retinal detachment including the fovea
This can either follow the progression of stage 4A disease or may be a result of zone I ROP wherein fibrovascular tissue proliferates posteriorly and early involvement of the macula is seen in the TRD (Figure 4e). Peripapillary detachments are also seen because of the contraction of fibrovascular tissue at the disc pulling up in an anteroposterior direction.
Stage 5: Total Retinal Detachment
Total retinal detachments are included in Stage 5 ROP. These retinal detachments are generally tractional with funnel-shaped configuration and may occasionally be rhegmatogenous as well if retinal break formation is seen because of the relentless traction. Typically, these tractional RD ‘s present with a “peripheral loop formation.”The peripheral avascular retina does not get detached while the vascular retina gets pulled up to the lens forming a loop between the vascular and avascular retina, the apex of which lies at the “ridge”. In case of a retinal break formation, this loop gets obliterated because the avascular retina also now gets detached. Stage 5 detachments are associated with the proliferation of fibrous tissue behind the lens giving rise to the typical “retrolental fibroplasia” (Figure 4f).
Stage 5 detachments appear as funnel-shaped on ultrasound examination. The “funnel” can be divided into anterior and posterior parts and based on its configuration, as seen on ultrasonography, this stage can be further subdivided. The funnel may be open both anteriorly and posteriorly, closed both anteriorly and posteriorly or maybe closed posteriorly and open anteriorly. The least common configuration is that of a funnel that is narrow anteriorly and opens posteriorly. Ultrasound may also be useful in picking up the presence or absence of a peripheral trough.
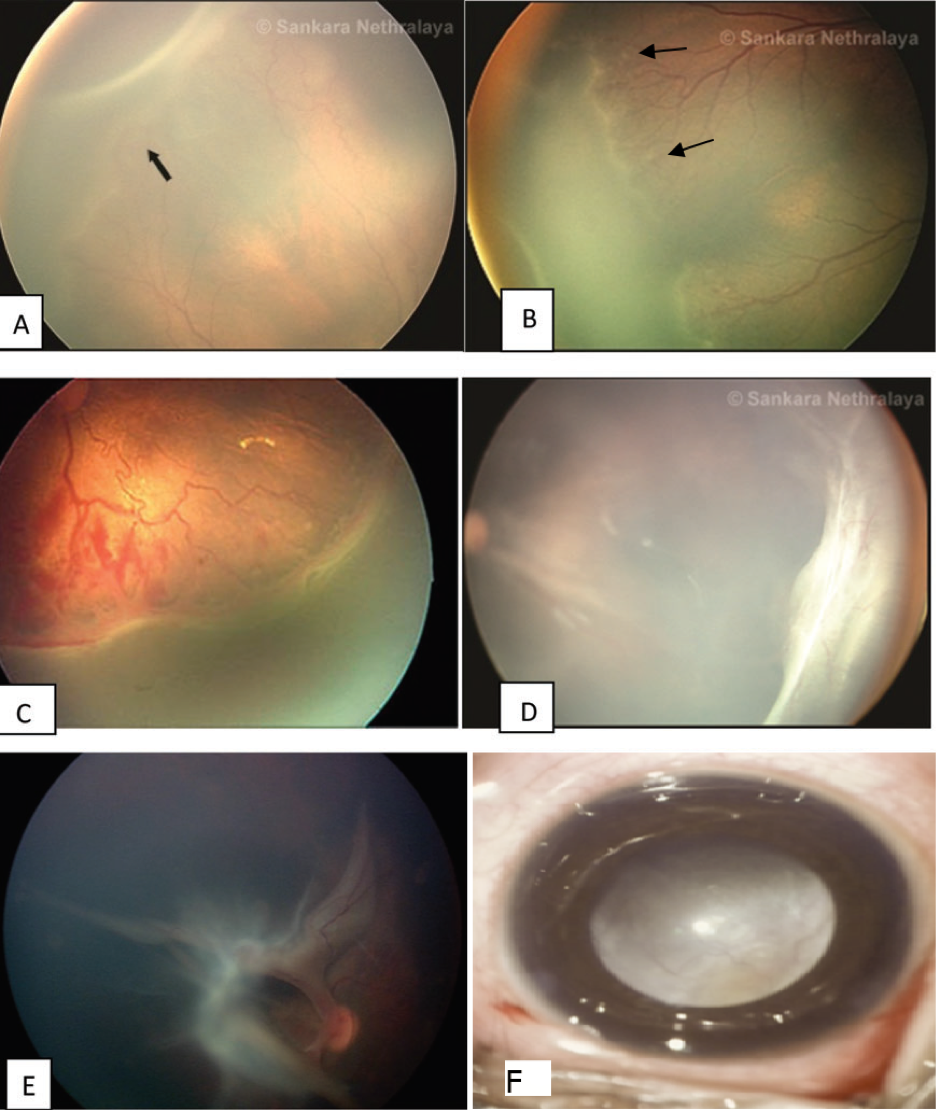
Fig. 4 (A) Stage 1: Demarcation line (arrow); (B) Stage 2: Ridge with popcorn lesions (arrows); (C) Stage 3: Ridge with extra retinal fibrovascular proliferation; (D) Stage 4A: extrafoveal retinal detachment; (E) Stage 4B: fovea involving retinal detachment; (F) Stage 5: Total retinal detachment (“Retrolental fibroplasia”)
Plus Disease
Plus disease is characterized by significant venous dilation and arteriolar tortuosity at the posterior pole involving at least 2 quadrants, vitreous haze, iris vascular engorgement, and pupillary rigidity (Figure 6 a and b).[35] This represents a more florid form of ROP, suggesting progressive vascular incompetence. It is indicated by adding a “+” sign after the disease stage number and is a sign of poor prognosis. It is important to recognize this because it is indicative of a rapidly progressive disease needing urgent treatment.

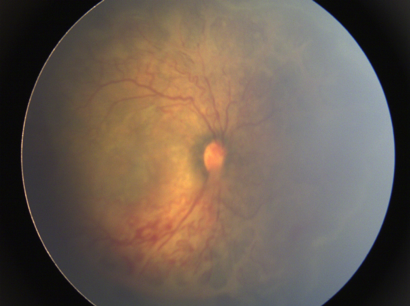
Fig 6: Plus disease: a) Rigid pupil because of the presence of engorged vessels seen at the edge of the pupil. This pupil does not dilate well; b) Fundus showing increased vessel tortuosity and dilatation
Pre Plus Disease:
It was defined by the revised ICROP in 2005 as the presence of abnormal dilatation of retinal veins and tortuosity of retinal arterioles at the posterior pole which is not severe enough to be classified as a plus disease but is more than normal. It is also written along with the disease stage eg. Stage 3 preplus.[35]
Aggressive Posterior ROP (APROP):
The revised 2005 ICROP described an uncommon severe and rapidly progressive form of ROP as “aggressive posterior ROP”.[35] This was previously termed as “Rush disease” and is characterized by a zone1 or posterior zone 2 diseases, an ill-defined ridge with rapidly progressive fibrovascular proliferation and a prominent plus disease (with increased dilation and tortuosity in all quadrants, which may be out of proportion to the apparent stage of the disease). The vascular changes are seen to progress rapidly and with formation of shunts from vessel to vessel within the retina. Flat network of neovascularization can be easily missed in absence of the typical ridge. It is apparent on careful examination at the junction of a featureless vascularized and non-vascularized retina. APROP typically progresses circumferentially and is often associated with a circumferential vessel. Both arterioles and venules show significant dilation and tortuosity making distinction difficult. This kind of disease may rapidly evolve without going through the classic stages of 1 to 3.
Threshold and pre-threshold diseases
The CRYO-ROP study (see below) described threshold ROP as stage 3 ROP with neovascularization involving 5 contiguous or 8 non-contiguous but cumulative clock hours in zone 1 or 2 with plus disease.[38] It was considered as the level of severity at which the risk of an unfavorable outcome was predicted to approach 50% and required active intervention. The pre-threshold disease was defined as zone 1, any stage and zone 2, stage 2 ROP with “plus” disease or zone 2 stage 3 disease.[38]
For a complete description of the disease in an eye with ROP the zone, stage and the presence or absence of plus disease must be mentioned. The posterior zone in which the disease is seen is used to represent the zone of the disease.
Timing of examination and screening guidelines
The “natural history” of ROP as studied in the CRYO-ROP study, showed that ROP occurs according to the infant’s corrected age (postconceptional age/ postmenstrual age), rather than the time since birth (chronological age).[39]It showed that in infants with birthweight <1250g, the median age of developing stage 1, stage 2, and threshold disease was 34.3, 35.5, and 36.9 weeks postconceptional age respectively.
The aim of screening premature babies for ROP is to detect and timely treat all neonates needing treatment, with minimal expense of time and resources. Screening strategy should be such as not to screen those babies who are unlikely to get a severe form of ROP at the same time not miss any child who could develop the disease. The criteria for screening babies are based on two critical factors - the birth weight and the gestational age. Other additional factors contributing to the development of ROP are also taken into consideration while framing the screening criteria.
The screening criteria as outlined by the AmericanAcademy of Paediatrics[40]include
- Infants with birth weight of ≤1500 g,
- Gestational age of 30 weeks or less,
- Infants with birth weight between 1500 and 2000 g or gestational age of >30 weeks with unstable clinical course.
There have been many reports of the prevalence of ROP in heavier babies from India as well as the other developing countries.15,16Keeping in mind the Indian scenario screening guidelines were modified in 2010 by the National Neonatology Foundation(NNF) to include criteria as shown in Table 1.[41]
|
S.No |
Criteria for Screening (Indian Guidelines) |
|
1 |
Birth weight <1750 g |
|
2 |
Gestational age at birth <34 weeks |
|
3 |
Exposed to oxygen >30 days |
|
4 |
Infants born at <28 weeks and weighing <1200 g are particularly at high risk of developing a severe form of ROP |
|
5 |
Presence of other factors such as respiratory distress syndrome, sepsis, multiple blood transfusions, multiple births (twins/triplets), apneic episodes, intraventricular haemorrhage increase the risk of ROP. In these cases, screening should be considered even for babies between 34-36 weeks gestational age or a birth weight between 1750-2000g. |
Table 1: ROP Screening criteria (Indian Guidelines)
As per the National Neonatology Foundation (NNF) guidelines, the first screening should be initiated within 4 weeks (30 days) of life in infants with gestational age >28 weeks. Screening should be done earlier (2–3 weeks after birth) if gestational age is <28weeks or birth weight is <1200g.[41]These recommendations were again modified in 2017 by the National Taskforce of ROP, Government of India. These revised guidelines suggest that all preterm babies ≤2000 g BW or ≤36 weeks GA should be screened for ROP.[42]
Eye Examination Technique
Screening is most commonly done with an indirect ophthalmoscope. The child should be fed 30-60 minutes prior to examination. Pupils may be effectively dilated using a combination of 1% tropicamide and 2.5% phenylephrine. These combination drops are used every 15 minutes for 3 times. Punctum occlusion is done after instilling the drops to reduce the systemic side effects of medication. Tropicamide is preferred over cyclopentolate drops that were used in the Cryo ROP study because of reduced incidence of side effects like bradycardia, apnoea, abdominal distension, and vomiting.[43,44]Excess eye drops should also be wiped off to prevent absorption through cheek skin. If the pupil does not dilate in spite of the proper use of medication, the presence of plus disease should be suspected. Repeated installation of topical drops should be avoided to prevent systemic problems.
Preparation Of The Baby:
The examination should be done in a warm room with the child well wrapped to avoid hypothermia. Babies in incubators can be screened within the incubators in the presence of a neonatologist with constant monitoring of the heart rate and oxygen saturation. The examination is performed using an indirect ophthalmoscope and a condensing lens. Sterile Alfonso or pediatric speculum may be used to retract the lids (Figure 7). Gentle indentation is done using a wire vectis or pediatric depressor to stabilize or rotate the globe. Care should be taken to avoid excessive pressure on the globe. A detailed note of the nasal and temporal peripheral retina is made to look for complete retinal vascularisation in absence of ROP. If ROP is noted, the stage of the disease, as well as the zone involved, is noted. The presence of plus or pre-plus disease should be documented [as described above, (Figure 6)]. These features are best documented in the form a drawing as shown in Figure 3.

The subsequent follow-up schedule for the babies who need observation depends on the findings on initial examination, as shown in Table 2
|
Follow-up |
Immature vascularization, no ROP |
ROP Stage 1 or 2 |
ROP Stage 3 |
Regressing ROP |
|
1 week or less |
The immature retina in Zone I or extending into posterior zone II |
Stage 1 or 2 in the zone I |
Zone III or Zone II Pre-threshold |
|
|
1-2 weeks |
Posterior zone II |
Stage 2 in zone II |
Regressing ROP, Zone I |
|
|
2 weeks |
Zone II |
Stage 1, zone II |
Regressing ROP, Zone II |
|
|
2-3 weeks |
Stage 1 or 2 in zone III |
Regressing ROP, Zone III |
Table 2 Follow-Up Schedule For ROP Babies (As Recommended By The ETROP)11
Telescreening In ROP:
In India, there are nearly 35 lac premature babies born every annually. Of these, nearly 1 lac are at risk of developing ROP and need screening. Considering that at least 3 examinations are necessary before declaring a child risk free from ROP, almost 3 lac examinations need to be done annually.[45] With not more than a few hundreds of ROP trained specialists in India, this is an uphill task. To bridge this gap, the concept of telescreening has been brought into ROP screening in a big way. High-quality retinal images obtained using commercially available wide-angle fundus cameras like the Retcamcamera (Clarity MSI, USA) followed by telescreening by a trained ophthalmologist can be done. In developing countries like India, where majority of people live in remote areas that may not have access to tertiary-level care, telescreening may bring more children into the screening program. This model has been successfully used by Vinekar et al. in Karnataka Internet Assisted Diagnosis of Retinopathy of Prematurity (KIDROP). [46]However, the limitation of Retcam is the initial cost of the equipment.
Treatment Guidelines:
Since the stimulus for abnormal vessels comes from the avascular retina, therefore, ablating the peripheral avascular retina causes regression of the ROP. This was first done by using Cryotherapy. The CRYO-ROP study started in 1986 was the first large scale, multicentre, randomized control trial that assessed the efficacy of cryotherapy in ROP.[38] It also studied the natural history of ROP as a secondary objective. Infants with birthweight ≤1250g were recruited in the study and those having stage 3 ROP with threshold disease (as described above) were eligible for transscleral cryotherapy and were randomly assigned to treatment and control (observation) groups. The treatment group received contiguous, nonoverlapping spots of transscleral cryotherapy to the avascular retina anterior to the ridge. Serial examinations were done to look for a response to therapy and unfavorable fundus outcomes (defined as posterior retinal detachment, posterior retinal fold involving the macula or retrolental mass). At 10-year outcome assessment, unfavorable fundus outcomes were present in 27% of treated eyes versus 48% of control eyes, and visual acuity was 20/200 or worse in 44% of treated eyes versus 62% of control eyes.[47]
With the results of Early treatment for retinopathy of prematurity (ETROP) study, there has been a shift in treatment to laser therapy from earlier practiced CRYO therapy.[48]In comparison to cryotherapy, laser therapy using binocular indirect ophthalmoscopic lasers allows more precision of treatment, is technically easier to perform, produces less extensive and more discrete scars, and reduces the unfavorable side effects of the cryotherapy, such as inflammation and swelling. Also, while for cryotherapy the baby needs to be under general anesthesia, the laser can be done under topical anesthesia. Moreover, outcomes of laser treatment of threshold disease in zone I and posterior zone II were superior to cryotherapy and at least equivalent to cryotherapy results in zone II disease.[49,50]
The ETROP study
ETROP study was a multicentre randomized clinical trial conducted in 1999 to study appropriate indications for the treatment of ROP to further reduce the risk of an unfavorable outcome.[48]In the ETROP study, 401 infants with pre-threshold ROP in one or both eyes were randomized to early treatment (laser photocoagulation at high-risk pre-threshold ROP, labeled as the “new threshold” or Type I ROP) versus conventional management (observation till threshold criteria developed). Eyes with any plus disease in zone I (stage 1, 2, or 3) or plus disease with stage 2 or 3 in zone II, or stage 3 ROP without plus disease in zone 1 were labeled as high-risk pre-threshold (Table 3).
The ETROP study results published in 2003 showed a significant benefit of earlier treatment intervention as measured by visual acuity outcome at a corrected age of 9 months and in the structural outcome of the retina at corrected ages of 6 and 9 months.[48]In the selected high-risk eyes that were studied, unfavorable functional results were reduced by earlier treatment intervention to 14.5%, from 19.5% in the conventionally treated control group (P = 0.01). Unfavorable structural outcomes were reduced from 15.6% in the control group to 9.1% in the early treatment eyes (P <0.001). The results produced a new clinical algorithm as a guide for treatment in eyes with severe ROP.It divided active ROP (stages 1-3) into Type 1 and Type 2 ROP, with prompt laser treatment being indicated for eyes with type 1 ROP (“the new threshold”) and continued serial observations without treatment recommended for eyes with type 2 ROP, as shown in table 3.
|
Table 3. ETROP treatment recommendations48[48] |
||
|
Type of active ROP |
Zones of involvement |
Recommendation |
|
Type 1 ROP (“new threshold”) |
Zone I Plus Stage 1, 2, 3 No plus Stage 3 Zone II Plus Stage 2,3 |
Treat with peripheral laser ablation |
|
Type II ROP |
Zone I No Plus Stage 1, 2 Zone II Plus Stage 1 No plus Stage 1,2, 3 |
Follow (wait and watch for progression) |
Table 3 :ETROP treatment protocol
Laser Treatment guidelines
Once the decision to do laser has been taken, laser photocoagulation is done using indirect laser ophthalmoscope under topical anesthesia after pupillary dilatation under the care of an anesthetist in the operation theatre. Lids are kept apart by using a speculum and depression is done using a wire vectis. Diode laser or frequency-doubled Nd-YAG laser can be used. The entire avascular retina up to the ora serrata should be ablated with near confluent burns (0.5–1 burn width apart) up to the ridge. In severe forms of disease not responding to this laser photocoagulation, further laser to the ridge as well as posterior to ridge has also been performed in some severe cases of ROP.[51]Heart rate, apnoea spells, and oxygen saturation should be monitored throughout the laser.[52]If frequent bradycardia or apnoea is seen treatment may need to be done in a staged manner. Care must also be taken to avoid hypothermia and hypoglycemia in the babies during the procedure. Follow-up visits after laser treatment are usually weekly till the ROP regresses and involution of all tractional elements is seen and vascularization reaches the temporal ora. Rapid regression of the disease is seen in 94-95% of the eyes with adequate treatment (Figure 8 a-c).
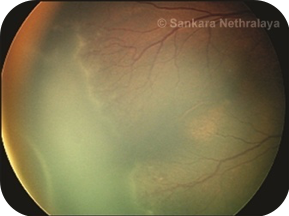 Fig 8a
Fig 8a
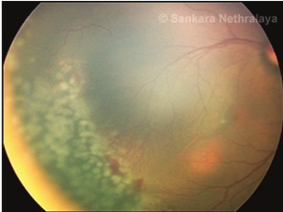 Fig 8b
Fig 8b
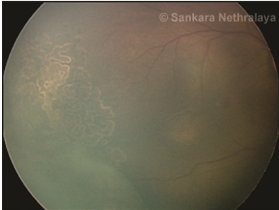 Fig 8c
Fig 8c
Fig 8: LASER in ROP. (a) Stage 3 zone 2 diseases; (b) nearly confluent burns are put in the avascular retina anterior to the ridge; c) regression of ridge seen following adequate laser.
Role of anti-vascular endothelial growth factor (VEGF):
In recent times, anti-VEGF has been used frequently in severe forms of ROP like zone 1 disease or APROP as well as in eyes that show a progression of disease in spite of maximal laser photocoagulation. Its role, however, is controversial. VEGF is needed in premature babies for the normal organogenesis and vasculogenesis. Systemic absorption may cause vascular development delay in other organs in these premature babies. Therefore, it is not recommended by many as first-line therapy.
BEAT ROP study which compared bevacizumab monotherapy with conventional laser therapy showed promising results for stage 3+ ROP in zone I but not in zone II.[53] In this study, peripheral retinal vessels continued in normal fashion after treatment with intravitreal bevacizumab. This study was too small to assess the safety profile in these babies. The follow-up period after mono therapy is unpredictable as recurrence of neovascularization has been seen in eyes treated only with anti-VEGF even beyond 54 weeks of post gestational age. It is recommended that follow-up should be continued until the vascularization reaches ora. In Zone 1 ROP, the Laser treatment outcomes are poorer. Treatment with anti-VEGF followed by a 4–5 days later with laser treatment in these cases has improved the efficacy of the laser along with a reduced need for extensive laser especially at the posterior pole which can permanently affect the visual outcome in these babies.[54] In a study by Chen et al, both bevacizumab and ranibizumab had similar efficacy at the end of 1 year in terms of ROP regression and visual acuity.[55]
On the basis of available literature definite indications for anti-VEGF therapy in ROP can be enumerated as:
1. The primary therapy for aggressive posterior zone 1 disease or APROP.
2. Aggressive anterior ROP with rubeosis iridis and rigid non-dilating pupil that does not allow adequate laser photocoagulation.
3. Media haze due to aggressive posterior disease to improve visualization for laser treatment.
4. Inadequate response to laser treatment leading to persistent neovascularization and tractional elements
5. As an adjunctive tool prior to surgery to reduce the risk of intraoperative bleeding.
In a recently concluded randomized clinical trial (the RAINBOW trial), 225 treatment naïve Type 1 ROP babies were recruited. Structural outcomes, as well as systemic safety outcomes with both treatment modalities (monotherapy with 0.2mg Ranibizumab and laser photocoagulation), were comparable[56]
Technique and dose of Anti-VEGF Injection:
The anti-VEGF injection is given in the operation theatre under sterile precautions under topical anesthesia under anesthetists’ care. Sterile cleaning and draping is done as for Intravitreal injection in adults. Half the adult dose i.e., 0.025 ml (0.625mg) of Bevacizumab is injected at 1.5 mm from the limbus into the vitreous cavity. Care is taken to avoid touching the lens by directing the needle towards the mid vitreous cavity. The injection is given once the tip of the needle is visualized inside the vitreous cavity. The indirect ophthalmoscopic examination should be done following the injection to look for retinal vascular pulsations at the optic disc. Anterior chamber paracentesis may be done only if the IOP is high digitally. The eyes is patched for 2 hours, followed by a dilated exam the next day.
Role of surgery:
Surgery is done for tractional retinal detachment (TRD) repair as seen in Stages 4 and 5 ROP. The aim of surgical intervention in Stage 4 ROP is to prevent the progression of retinal detachment. All surgeries are done under general anesthesia.
Scleral Buckling:
Scleral buckling is done for some cases of Stage 4A ROP with only peripheral tractional retinal detachment.
Procedure:
360-degree conjunctival limbal peritomy is done. This is followed by the tagging of all the four recti. Scleral tunnels are made in each quadrant and a2.5 mm width 240 encircling band is placed at the height of the TRD as determined by indirect ophthalmoscopy. This surgery does not involve the removal of membranes formed on the retina but by causing peripheral scleral indentation reduces the effective TRD. This procedure can be combined with cryotherapy or laser to any peripheral persistent new vessels in active disease. The advantage of the procedure is the absence of intraocular manipulation but the encircling band needs to be removed once the child is 1 year of age to allow normal growth of the eyeball and to reduce the amount of anisometropia that may be induced by the buckle.
Vitrectomy:
Pars plicata vitrectomy needs to be performed for stage 4B, Stage 5 and some cases of Stage 4 A, wherein fibrovascular proliferation causing the tractional detachment is too severe and extensive to be relieved by scleral buckling alone. Lens sparing vitrectomy can be done for Stage 4A and 4 B. Stage 5 ROP with retrolental fibroplasia however necessitates the removal of the lens as well.
Lens sparing vitrectomy (LSV)
Surgical Technique:
For 20-gauge surgery, limited peritomy in the superonasal, inferonasal, and inferotemporal quadrant is made. Three ports are made through the pars plicata about 1.5 mm from the limbus making sure that the MVR is directed posteriorly towards the mid vitreous cavity to avoid touching the lens. With the advent of MIVS more and more ROP surgeries are being done by this technique due to the natural advantage of small instruments for smaller eyes, minimal surgical trauma, more patient comfort, and early postoperative recovery.[57] Smaller instruments of 25- gauge vitrectomy has additional advantages in ROP surgery like better access to the retrolental space as well as in between retinal folds without causing lens touch or retinal break formation. For ROP sclerotomies are fashioned at 1-1.5 mm from the limbus. The location of the sclerotomies is decided after indirect ophthalmoscopy. Sclerotomies are not made in the clock hours that have the presence of anterior retinal folds to prevent retinal break formation. The viewing system used is the Binocular Indirect Ophthalmic Microscope (BIOM). To facilitate bimanual dissection, combination instruments like infusion light pipe or infusion cannula with the light pipe are useful during membrane dissection (Figure 9a). Unlike diabetic vitrectomy, the aim of vitrectomy in ROP is not complete removal of the vitreous or the fibro-vascular proliferation. An attempt is made to relieve the traction between “ridge to ora”, “ridge to ridge” and “ridge to the lens”. By relieving this traction the retina falls back and removal of vitreous also helps by reducing the VEGF load in the vitreous cavity. Induction of posterior vitreous detachment may not be possible in these eyes. Modification of the technique in the form of conjunctival dissection as well as suturing of the sclerotomies at the end of surgery may be necessary.[58]
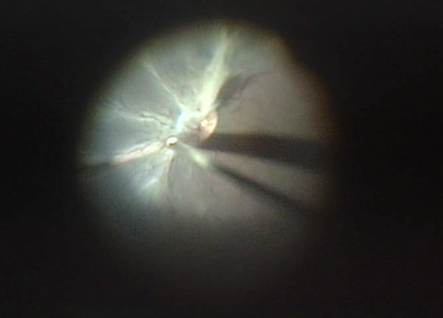 Fig 9a
Fig 9a
Fig. 9: Surgical management of ROP: (a) Intraoperative picture of LSV in a baby with Stage 4 B ROP;
Lens sparing vitrectomy has shown promising results in Stages 4A and 4B. Long-term results with lens sparing vitrectomy were favorable in a study by Trese et al: 82.1% (Stage 4A), 69.5% (Stage 4B), and 42.6% (Stage 5) showing successful anatomical reattachment of the retina with the lens being clear for at least the first decade of life.[59] Bhende et al. also reported 82% anatomical success in 4A stage ROP and 50% in Stage 4B ROP after a single procedure.[60] Modern vitreoretinal surgical tools like the infusion light pipe, binocular indirect ophthalmoscopy (BIOM) which allows wide-angle viewing has reduced the risk of creating iatrogenic retinal breaks and also sparing of the lens, allowing easy visual rehabilitation in these children.[61]Reports of MIVS in ROP have stated a success rate of 82% -100% for stage 4 ROP.[57-60]Figure 10 shows an algorithm for the surgical management of ROP.

Figure 10: An algorithm for Surgical management of ROP
Surgery for Stage 5 ROP with Retrolental Fibroplasia:
Preoperative evaluation should be done to determine the extent of the disease and the presence of any negative prognostic factors such as corneal edema, shallow anterior chamber, or pupillary block glaucoma. The presence of extensive subretinal membranes, exudates, or blood is considered as an indicator of inoperability.
Surgical technique
Lensectomy is done for complete access to the retrolental tissue. Sclerotomies are fashioned 0.5 mm from the limbus or can even be at the limbus. The aim of the surgery for Stage 5 ROP is to clear all pre-retinal tissue up to the disc and open the peripheral trough all round. In most instances, bimanual surgery under viscoelastic is performed through the superior two sclerotomies. Anterior chamber (AC) maintainer to keep the IOP under control during surgery is also used. Fixation of infusion cannula through pars plicata is impossible in these cases because the retina is pulled up right behind the lens.
Coaxial illumination of the operating microscope is used for the most part of surgery without the wide-angle viewing system due to the anterior location of the fibrovascular tissue. In case of shallow anterior chamber(AC) or presence of peripheral anterior synaechiae (PAS), viscoelastic Is used to form the AC and iris spatula is used to release the anterior and posterior iris synechiae. The lens is generally clear and is removed using a vitrectomy probe under low suction. The retrolental fibrous membrane is then opened by making full thickness radial cuts from centre towards the equator in a cruciate manner to expose the underlying retina (Figure 9b). This dissection needs to be done carefully to avoid iatrogenic retinal breaks. The preretinal tissue is completely removed by delaminated using intraocular forceps and scissors. The dissection is continued till the posterior funnel is opened and the optic disc is visible. Now the peripheral trough is opened up all around by carefully dissecting into the fold formed by the peripheral attached avascular retina and the retinal fold from the ridge. This may need the use of a wide-angle viewing system.
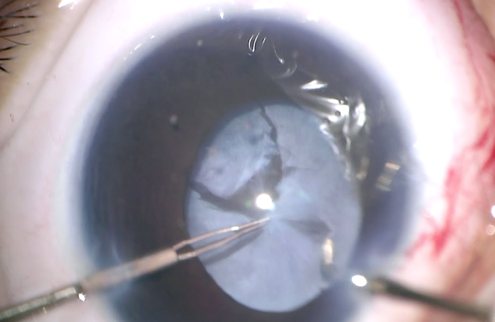
Fig 9b: Intraoperative picture of Lensectomy + Vitrectomy in a baby with stage 5 ROP. Cruciate incisions followed by complete removal of the Retrolental fibroplasia is done
Complications of the surgery include retinal break formation, vitreous haemorrhage, secondary glaucoma and corneal clouding.
Open sky vitrectomy (through a trephined corneal opening) has also been advocated in the past by some especially in cases with corneal opacity or intraoperative corneal clouding.[62] It has the advantage of allowing two hand dissection from a large anterior incision but maintenance of intraocular pressure is difficult and there is risk of corneal rejection postoperatively.
In Stage 5 ROP, the results of surgical intervention are poor. Gopal et al. reported anatomical success with the attachment of posterior pole in 22.5% of cases with lens sacrificing closed globe vitrectomy though visual results were unsatisfactory with only two children showing mobile vision out of 96 eyes.[63]
Differential diagnosis
In the neonatal intensive care unit (NICU) set up, a typical fundus picture in a premature infant, the differential diagnosis is almost exclusively limited to ROP. But several conditions can mimic ROP with similar findings, especially in older children. Familial exudative vitreoretinopathy (FEVR), Norrie disease, incontinentia pigmenti (IP) can present with peripheral avascular retina, neovascularization, and tractional retinal detachment. The absence of a history of prematurity can rule out ROP in these cases. Retinoblastoma can also present with retinal detachment, although it is often exudative and has a convex surface as opposed to the concave surface seen in tractional RD. The tumors associated with exudative detachments are typically quite large and would be picked up on the ultrasound.
Persistent fetal vasculature (PFV) can have a similar appearance to ROP, but is usually unilateral. Other features in PFV include the smaller size of the globe, visualization of the pulled up ciliary processes and absence of a history of prematurity. Coats disease is also generally unilateral and has substantial lipid exudates which are not typically seen in ROP.
Natural Regression of ROP
Some cases of mild ROP regress spontaneously by a process of involution or evolve from a vascular phase to a fibrotic phase as the disease dies out. Natural regression typically begins at 38 weeks of postconceptional/ post menstruation age and is characterized by the downgrading of staging with or without progressive advancement of retinal vasculature into a more peripheral region of the retina on serial examinations.[64]Involution from early stages of the disease may happen without any sequelae of ROP while the more severe acute forms of ROP are more likely to have more severe involutional changes that are marked by fibrosis and termed as “cicatricial” phase.[65,66]
Involutional sequelae include a wide spectrum of peripheral and posterior retinal and vascular changes. Ocular findings in milder forms of regressed ROP include vascular anomalies like small areas of peripheral vascular a vascularity, abnormal branching or straightening of retinal vessels, areas of irregular pigmentary changes (increased or decreased pigmentation) in the peripheral retina, vitreoretinal membranes and interface changes and high myopia. More prominent changes include straightening and temporal dragging of vessels and macula (macular heterotropia)(Figure 11a), avascularity, and lattice degeneration in the peripheral retina with large areas of pigmentary changes. Falciform retinal folds can be seen in severe cases (Figure 11b). The most advanced end-stage cases can have a totally detached retina seen as a thickened fibrous membrane behind the lens, referred to as retrolental fibroplasia.
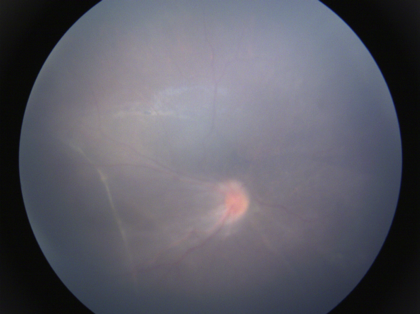 Fig 11a
Fig 11a
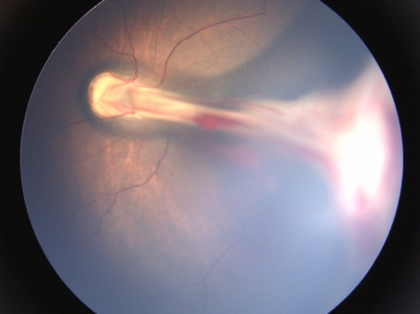 Fig 11b
Fig 11b
Fig. 11: ROP sequelae a) Straightening of vessels with ectopic fovea;b)Falciform fold
Children with ROP are also at increased risk of developing associated ocular problems like myopia, strabismus and amblyopia, angle-closure glaucoma, cataract, and retinal detachment. The incidence of these complications increases with the extent and stage of ROP and lower birth weight.[63-65]
In patients with a history of prematurity and suboptimal best-corrected visual acuity, particular attention should be given to the temporal periphery of the retina to look for the tell-tale signs of regressed ROP. These patients and their parents should be alerted to the symptoms of rhegmatogenous retinal detachment and glaucoma as soon as the child is old enough to appreciate and report them.
Medicolegal Aspects:
There is a need to create ROP awareness among parents and concerned healthcare providers. It is important for pediatricians, neonatologists, obstetricians, and ophthalmologists to be aware of the need for screening and follow-up of premature infants at risk and timely intervention/referral when required.
Conclusion
Considering the poor outcome of surgery in end stages of ROP, and the social and economic burden of bringing up a blind child, screening and timely intervention in the form of laser treatment is the best treatment option. There is a need to increase the awareness of the disease to facilitate teamwork amongst ophthalmologists, neonatologists, health care workers and government institutions to ensure timely treatment of these helpless babies.
References
- Terry T.L. Extreme prematurity and fibroblastic overgrowth of persistent vascular sheath behind each crystalline lens. I. Preliminary report. Am J Ophthalmol1942;25:203-204
- Owens WC, Owens EU. Retrolental fibroplasia in premature infants. Am J Ophthalmol 1949;32:1–29.
- Rubaltelli DM, Hirose T. Retinopathy of prematurity update. Int Ophthalmol Clin. 2008 Spring;48(2):225-35.
- Patz A, Hoeck LE, De La Cruz, E. Studies on the effect of high oxygen administration in retrolental fibroplasia. I. Nursery observations. Am J Ophthalmol 1952;35:1248–53.
- Kinsey VE. Retrolental fibroplasia: cooperative study of retrolental fibroplasia and the use of oxygen. Arch Ophthalmol 1956;56:481–543.
- Silverman WA. Retrolental fibroplasia: a modern parable. New York: Grune and Stratton, Inc; 1980. p. 207.
- Multicenter trial of cryotherapy for retinopathy of prematurity. Three-month outcome. Cryotherapy for Retinopathy of Prematurity Cooperative Group. Arch Ophthalmol. 1990 Feb;108(2):195-204.
- Good WV, Hardy RJ, Dobson V, et al. The incidence and course of retinopathy of prematurity: findings from the early treatment for retinopathy of prematurity study. Pediatrics 2005;116(1):15–23.
- Gilbert C. Retinopathy of prematurity: a global perspective of the epidemics, population of babies at risk and implications for control. Early Hum Dev 2008; 84(2):77–82
- Kong L, Fry M, Al-Samarraie M, et al. An update on progress and the changing epidemiology of causes of childhood blindness worldwide. J AAPOS 2012;16(6): 501–7.
- Charan R, Dogra MR, Gupta A, et al. The incidence of retinopathy of prematurity in a neonatal care unit. Indian J Ophthalmol 1995;43(3):123–6.
- Shah PK, Narendran V, Kalpana N, Gilbert C. Severe retinopathy of prematurity in big babies in India: history repeating itself? Indian J Pediatr 2009; 76: 801-804.
- Patwardhan SD, Azad R, Gogia V, et al. Prevailing clinical practices regarding screening for retinopathy of prematurity among pediatricians in India: a pilot survey. Indian J Ophthalmol 2011;59(6):427–30.
- Zin A, Gole GA. Retinopathy of prematurity-incidence today. Clin Perinatol. 2013;40:185-200.
- World Health Organization. Born too soon: the global action report on preterm birth. http://www.who.int/pmnch/media/news/2012/201204_borntoosoon-report.pdf
- Blencowe H, Lawn JE, Vazquez T, Fielder A, Gilbert C. Preterm associated visual impairment and estimates of retinopathy of prematurity at regional and global levels for 2010. Pediatr Res. 2013;74: 35–49.
- Dutta S, Raghuveer T, Vinekar A, Dogra MR. Can we stop the current epidemic of blindness from Retinopathy of Prematurity? Indian Pediatr. 2016;53:80–4.
- Mintz-Hittner HA. Retinal development and the pathophysiology of retinopathy of prematurity. In: Pollin RA, Fox WW, Abman SH, eds. Fetal and neonatal physiology, Vol 2. Philadelphia: Saunders; 2011. p. 1875–1881.
- Hartnett ME. Studies on the pathogenesis of avascular retina and neovascularization into the vitreous in peripheral severe retinopathy of prematurity (an American Ophthalmological Society thesis). Trans Am Ophthalmol Soc.2010;108: 96–119.
- Hughes S, Yang H, Chan-Ling T. Vascularization of the human fetal retina: roles of vasculogenesis and angiogenesis. Invest Ophthalmol Vis Sci 2000; 41: 1217–1228.
- Chan-Ling T, Gock B, Stone J. The effect of oxygen on vasoformative cell division. Evidence that ‘physiological hypoxia’ is the stimulus for normal retinal vasculogenesis. Invest Ophthalmol Vis Sci. 1995;36(7):1201–1214.
- Pharmacologic interventions for the prevention and treatment of retinopathy of prematurity. Beharry KD, Valencia GB, Lazzaro DR, Aranda JV. Semin Perinatol. 2016 Apr;40(3):189-202.
- Cavallaro G, Filippi L,et al. The pathophysiology of retinopathy of prematurity: an update of previous and recent knowledge. Acta Ophthalmol. 2014 Feb;92(1):2-20.
- Rekha S, Battu RR. Retinopathy of prematurity: incidence and risk factors. Indian Pediatr. 1996 Dec;33(12):999-1003
- Dutta S, Narang S et al. Risk factors of threshold retinopathy of prematurity. Indian Pediatr. 2004 Jul;41(7):665-71.
- Lee J, Dammann O. Perinatal infection, inflammation, and retinopathy of prematurity. Semin Fetal Neonat Med.2012;17:26–29. 37–46.
- Wu C, Hellström A, et al. WINROP Consortium. Importance of early postnatal weight gain for normal retinal angiogenesis in very preterm infants: a multicenter study analyzing weight velocity deviations for the prediction of retinopathy of prematurity. Arch Ophthalmol. 2012 Aug;130(8):992-9.
- Hutchinson AK, Melia M et al. Clinical Models and Algorithms for the Prediction of Retinopathy of Prematurity: A Report by the American Academy of Ophthalmology. Ophthalmology. 2016 Apr;123(4):804-16.
- Alon T, Hemo I, Itin A, et al. Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity. Nature Med 1995;1:1024–8.
- STOP-ROP Multicenter Study Group. Supplemental therapeutic oxygen for prethreshold retinopathy of prematurity (STOP-ROP), a randomized, controlled trial. I. Primary outcomes. Pediatrics 2000;105:295–310.
- Castillo A, Sola A, Baquero H, Neira F, Alvis R, Deulofeut R, et al. Pulse oxygen saturation levels and arterial oxygen tension values in newborns receiving oxygen therapy in the neonatal intensive care unit: is 85% to 93% an acceptable range? Pediatrics 2008;121:882e9.
- Sink D, Thomas P, Bober B, Hagadorn JI. Oxygen With Love (OWL) improves oxygen saturation target achievement and eye outcomes in preterm newborn. Available fromhttp://www.docstoc.com/docs/73625317/OxygenwithLove_OWL_ ImprovesOxygenSaturationTarget October 24, 2012.
- Garcia-Serrano JL, Uberos Fernández J, Anaya-Alaminos R, Jerez-Calero A, Padilla-Torres JF, Ramírez-Garcia MC, Piñar-Molina R. "Oxygen with love" and diode laser treatment decreases comorbidity and avoidable blindness due to retinopathy of prematurity: results achieved in the past 12 years. Pediatr Neonatol. 2013 Dec;54(6):397-401.
- Committee for the Classification of Retinopathy of Prematurity. An international classification of retinopathy of prematurity. Arch Ophthalmol 1984;102:1130–4.
- An international classification of retinopathy of prematurity II. The classification of retinal detachment. The International Committee for the Classification of the Late Stages of Retinopathy of Prematurity. Arch Ophthalmol. 1987 Jul;105(7):906-12.
- The International Classification of Retinopathy of Prematurity Revisited. Arch Ophthalmol. 2005;123:991-99
- Garner A. The pathology of retinopathy of prematurity. In: Silverman WA, Flynn JT, editors. Retinopathy of prematurity. Boston: Blackwell; 1985. pp. 19–52.
- Cryotherapy for Retinopathy of Prematurity Cooperative Group. Multicenter trial of cryotherapy for retinopathy of prematurity: preliminary results. Arch Ophthalmol 1988;106:471–9.
- Palmer EA, Flynn JT, Hardy RJ, et al. Incidence and early course of retinopathy of prematurity. The Cryotherapy for Retinopathy of Prematurity Cooperative Group. Ophthalmology. 1991 Nov;98(11):1628-40.
- Fierson WM. Screening examination of premature infants for retinopathy of prematurity. Pediatrics. 2013;131:189–95
- Pejawar R, Vinekar A, Bilagi A. National Neonatology Foundation’s Evidence-based Clinical Practise Guidelines (2010), Retinopathy of Prematurity, NNF India, New Delhi 2010:253–62.
- Rashtriya Bal Swasthya Karyakram, Ministry of Health & Family Welfare, Government of India. Guidelines for Universal Eye Screening in Newborns including Retinopathy of Prematurity, 2017. https://nhm.gov.in/images/pdf/programmes/RBSK/Resource_Documents/Revised_ROP_Guidelines-Web_Optimized.pdf
- Bolt B, Benz B, Koerner F, Bossi E. A mydriatic eye-drop combination without systemic effects for premature infants: a prospective double-blind study. J Pediatr Ophthalmol Strabismus. 1992 May-Jun;29(3):157-62.
- Mitchell AJ, Green A, Jeffs DA, Roberson PK. Physiologic Effects of Retinopathy of Prematurity Screening Examinations.Advances in neonatal care : official journal of the National Association of Neonatal Nurses. 2011;11(4):291-297. doi:10.1097/ANC.0b013e318225a332.
- Blencowe H, Moxon S, Gilbert C. Update on Blindness Due to Retinopathy of Prematurity Globally and in India. Indian Pediatr. 2016 Nov 7;53 Suppl 2:S89-S92.
- Vinekar A, Gilbert C, Dogra M, Kurian M, Shainesh G, Shetty B, Bauer N. The KIDROP model of combining strategies for providing retinopathy of prematurity screening in underserved areas in India using wide-field imaging, tele-medicine, non-physician graders and smart phone reporting. Indian J Ophthalmol. 2014;62(1):41–9.
- Cryotherapy for Retinopathy of Prematurity Cooperative Group. Multicenter trial of cryotherapy for retinopathy of prematurity: ophthalmological outcomes at 10 years. Arch Ophthalmol 2001;119:1110–8.
- Early Treatment of Retinopathy of Prematurity Cooperative Group. Revised indications for treatment of retinopathy of prematurity: results of early treatment of retinopathy of prematurity randomized trial. Arch Ophthalmol. 2003;121:1684–96.
- O’Keefe M, Burke J, Algawi K, et al. Diode laser photocoagulation to the vascular retina for progressively advancing retinopathy of prematurity. Br J Ophthalmol 1995;79:1012–4.
- White JE, Repka MX. Randomized comparison of diode laser photocoagulation versus cryotherapy for threshold retinopathy of prematurity. Three-year outcome. J Pediatr Ophthalmol Strabismus 1997;34:83–7
- Uparkar M, Sen P, Rawal A, Agarwal S, Khan B, Gopal L. Laser photocoagulation (810 nm diode) for threshold retinopathy of prematurity: a prospective randomized pilot study of treatment to ridge and avascular retina versus avascular retina alone. Int Ophthalmol. 2011;31(1):3–8.
- Jalali S, Azad R, Trehan HS, Dogra MR, Gopal L, Narendran V. Technical aspects of laser treatment for acute retinopathy of prematurity under topical anesthesia. Indian J Ophthalmol. 2010;58(6):509–15.
- Mintz-Hittner HA, Kennedy KA, Chuang AZ; BEAT-ROP Cooperative Group. Efficacy of intravitreal bevacizumab for stage 3+ retinopathy of prematurity. N Engl J Med. 2011;364 (7):603–15.
- Mota A, Carneiro A, Breda J, Rosas V, Magalhaes A, Silva R, Falcao-Reis F. Combination of intravitreal ranibizumab and laser photocoagulation for aggressive posterior retinopathy of prematurity. Case Rep Ophthalmol. 2012;3(1):136–41.
- Chen SN, Lian I, Hwang YC, Chen YH, Chang YC, Lee KH, Chuang CC, Wu WC. Intravitreal anti-vascular endothelial growth factor treatment for retinopathy of prematurity: comparison between ranibizumab and bevacizumab. Retina. 2015;35(4):667–74.
- Stahl A, Lepore D, Fielder A, Fleck B, Reynolds JD, Chiang MF, Li J, Liew M, Maier R, Zhu Q, Marlow N. Ranibizumab versus laser therapy for the treatment of very low birthweight infants with retinopathy of prematurity (RAINBOW): an open-label randomised controlled trial. The Lancet. 2019 Oct 26;394(10208):1551-
- Sen P, Bhende P, Sharma T, Gopal L, Maitray A, Shah P, Oke Y. Surgical outcomes of microincision vitrectomy surgery in eyes with retinal detachment secondary to retinopathy of prematurity in Indian population. Indian journal of ophthalmology. 2019 Jun;67(6):889.
- Gonzales CR, Boshra J, Schwartz SD. 25-Gauge pars plicata vitrectomy for stage 4 and 5 retinopathy of prematurity. Retina. 2006;26(7 Suppl):S42–6.
- Nudleman E, Robinson J, Rao P, Drenser KA, Capone A, Trese MT. Long-term outcomes on lens clarity after lens-sparing vitrectomy for retinopathy of prematurity. Ophthalmology. 2015;122(4):755–9.
- Bhende P, Gopal L, Sharma T, Verma A, Biswas RK. Functional and anatomical outcomes after primary lens-sparing pars plana vitrectomy for Stage 4 retinopathy of prematurity. Indian J Ophthalmol. 2009;57(4):267–71.
- Trese MT, Capone A Jr. Surgical approach to infant and childhood retinal diseases: invasive methods. In Hartnett ME, Tresse MT, Capone A Jr, et al, eds. Pedatric Retina. Philadelphia: Lipponcott Williams& Williams; 2005, pp. 359–64.
- Tasman W, Borrone RN, Bolling J. Open sky vitrectomy for total retinal detachment in retinopathy of prematurity. Ophthalmology. 1987;94(4):449–52.
- Gopal L, Sharma T, Shanmugam M, Badrinath SS, Sharma A, Agraharam SG, Choudhary A. Surgery for stage 5 retinopathy of prematurity: the learning curve and evolving technique. Indian J Ophthalmol. 2000;48(2):101–6.
- Repka MX, Palmer EA. Involution of retinopathy of prematurity. Arch Ophthalmol 2000;118:645–9.
- Cryotherapy for Retinopathy of Prematurity Cooperative Group. The natural ocular outcome of premature birth and retinopathy: status at one year. Arch Ophthalmol 1994;112:903–12.
- Cryotherapy for Retinopathy of Prematurity Cooperative Group. Multicenter trial of cryotherapy for retinopathy of prematurity: Snellen acuity and structural outcome at 5 ½ years. Arch Ophthalmol 1996;114:417–24.

