Introduction:
Cytomegalovirus (CMV) retinitis is an opportunistic viral posterior uveitis that occurs mostly in severely immunocompromised Acquired Immunodeficiency Syndrome (AIDS) patients or, rarely, in those on immunosuppressive therapy after organ transplantation, or who are on systemic corticosteroids.1 More than 75% of AIDS patients with CD4+ counts less than 50 cells/μL have CMV retinitis. It is the most common ocular opportunistic infection in AIDS patients and accounts for 75-85% of all CMV end-organ disease.2 Before the introduction of Highly Active Antiretroviral Treatment (HAART), 30% of patients with Human Immunodeficiency Virus (HIV) developed CMV retinitis during their lifetime, but with the advent of HAART, there is a 75% reduction in the number of new cases of CMV retinitis.3 Rarely, CMV retinitis may occur in non-HIV patients having some degree of immune dysfunction, such as advanced age, diabetes mellitus, corticosteroid, and non-cytotoxic immunosuppressive drug usage.1
Epidemiology:
CMV, or HHV-5, is a double-stranded DNA virus that belongs to the Herpesviridae family with a genome of approximately 230 kb encoding 160 genes.4 Primary CMV infection is transmitted perinatally, by close contact, sexual contact, or parenterally. CMV infections occur in approximately 1% of all live births throughout the world, making it the most common cause of intrauterine infection in humans.6 Primary maternal infection causes more frequent and severe fetal complications than reactivation of maternal disease during pregnancy. Approximately 5-10% of congenitally infected neonates have cytoplasmic inclusion disease (CID) which is characterized by lesions of the reticuloendothelial and central nervous systems.7 Ocular manifestations of CID include chorioretinitis, optic atrophy, and cataract formation. The remaining 90% of congenital infections are undiagnosed due to their asymptomatic nature or subtle deficits, such as sensorineural hearing loss or psychomotor disturbance. Prolonged viral shedding may occur for several years in asymptomatic infected children.
In adults, the seropositive prevalence in developed countries ranges from 50-70% and approaches 100% in less developed countries.7 Seropositivity increases with age and reaches the highest incidence in individuals older than 50 years of age. Most immunocompetent individuals with positive serology have asymptomatic infection. Few develop an infectious mononucleosis-like syndrome, whereas more severe diseases may manifest in immunocompromised patients in the form of encephalitis, pneumonitis, hepatitis, colitis, and uveitis.6 The virus remains latent in mononuclear lymphocytes in immunocompetent hosts and only reactivates when immunity is suppressed in patients with AIDS and immunosuppressed transplant patients.
Cytomegalovirus and HIV interactions:
CMV and HIV transactivate each other in vitro resulting in increased yields of both the viruses.8,9 Dual infection of individual retinal cells of autopsy eyes from patients with AIDS and CMV retinitis with both CMV and HIV has been demonstrated.10 CMV produces cytokine homologues that bind to host receptors and downregulate the immune system. One key cytokine homologue appears to be an interleukin (IL) 10 homologue. IL-10 is a human cytokine that inhibits T helper type-1 (Th1) immune responses, which are key to cell-mediated immunity.11 Cytomegalovirus also produces chemokine receptors, which bind chemokines and inhibit the recruitment of inflammatory and immune cells, and it interferes with natural killer cells, thereby inhibiting the host’s ability to clear viruses.
Clinical features:
CMV retinitis is commonly seen between the age group of 30 and 60 years. Males are commonly involved. Bilateral CMV retinitis is seen in about 30% of cases but it usually begins as unilateral disease, and later involves the contralateral eye within six months probably from hematogenous spread.
CMV retinitis is asymptomatic in 50% of cases. Visual complaints related to CMV retinitis closely correlates with the area of retinal involvement. Early lesions of the peripheral retina may cause mild visual impairment or may go undetected. Symptoms associated with CMV retinitis include blurred vision, floaters, scotoma, metamorphopsia, micropsia, and ocular discomfort. CMV retinitis presents with mild anterior segment inflammation, including keratic precipitates, anterior chamber cells, and minimal vitritis.
The characteristic feature is a focal or discrete lesion of full-thickness yellow-white necrotizing retinitis with granular borders.1,5,12 It has perivascular distribution and expands centrifugally. There may be mild vitritis, retinal edema, hemorrhage, and vasculitis, usually in the form of retinal arteritis, which is called “pizza pie retinopathy”. (Figure 1)
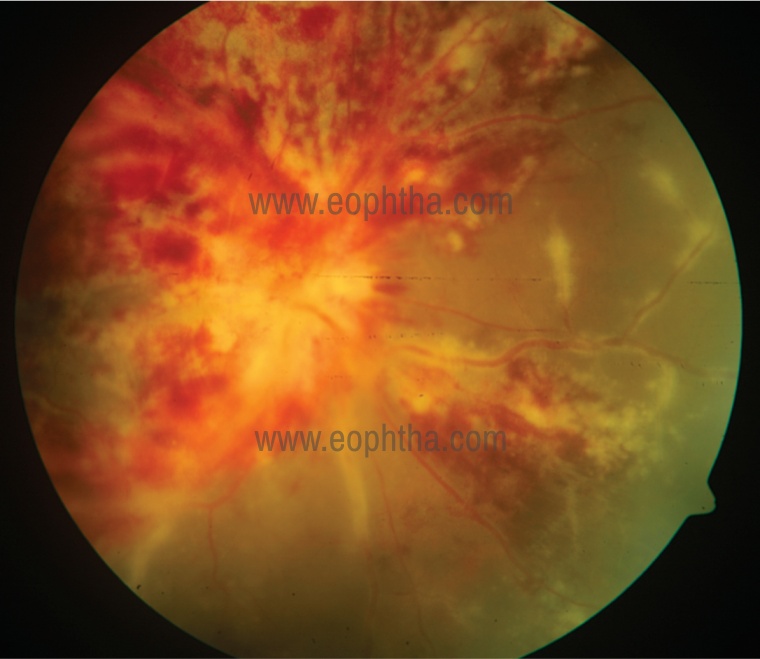
Figure 1: Pizza pie retinopathy with disc edema and retinal vasculitis.
There are two stages in CMV retinitis5,12:
I] Stage 1: The first stage is active retinitis. Three patterns of active retinal lesions have been described:
- Fulminant/oedematous pattern: It consists of large areas of confluent retinal necrosis with overlying retinal haemorrhage. This form progresses as a brushfire-like extension from each discrete lesion. (Figure 2)
- Indolent/granular pattern: It consists of granular satellite lesions with little or no
retinal haemorrhage. (Figure 3) - Frosted branch angiitis: It is also called as exudative pattern. This form presents as
extensive vascular sheathing .
II] Stage 2: Stage 1 can progress to extensive necrosis. This leads to development of large multiple retinal tears leading to retinal detachment. Occasionally, disease can become quiescent manifesting as disc pallor, retinal atrophy, fibrosis, calcification and sclerotic vessels. (Figure 4)
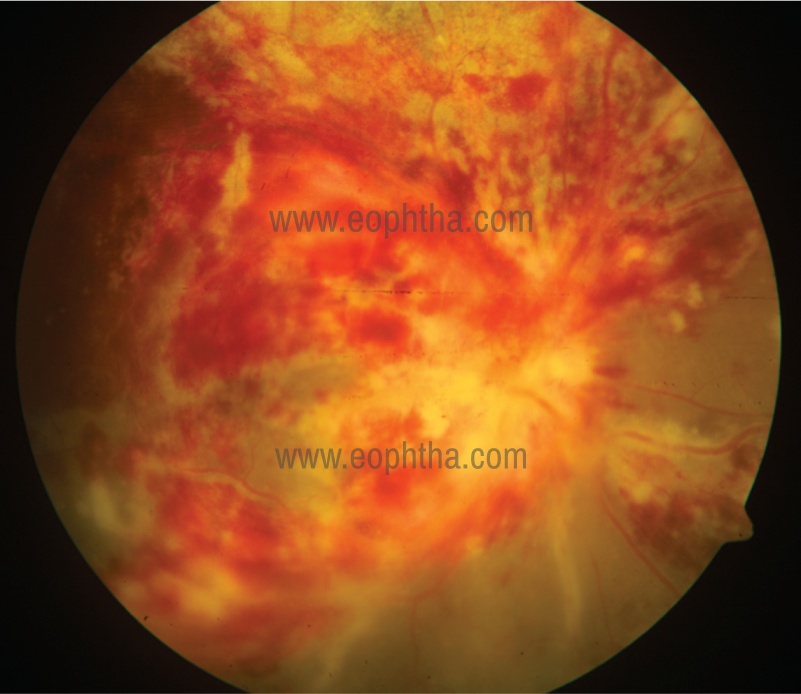
Figure 2: Fulminant pattern of CMV retinitis.
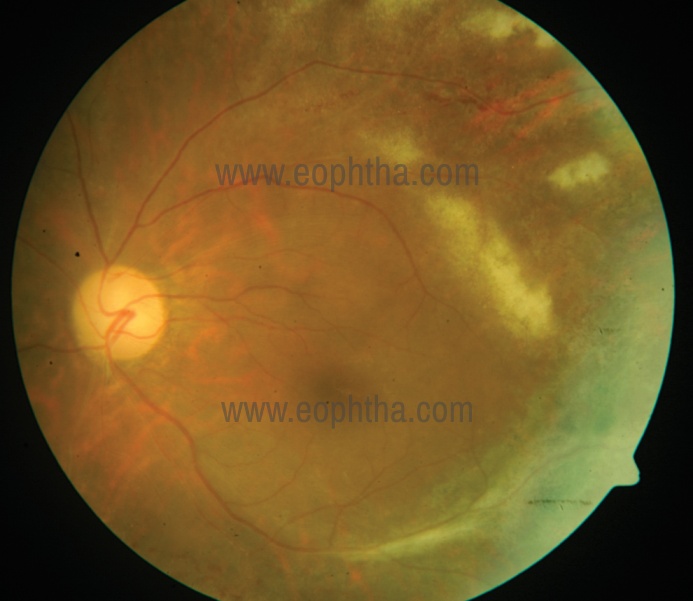
Figure 3: Granular pattern of CMV retinitis.
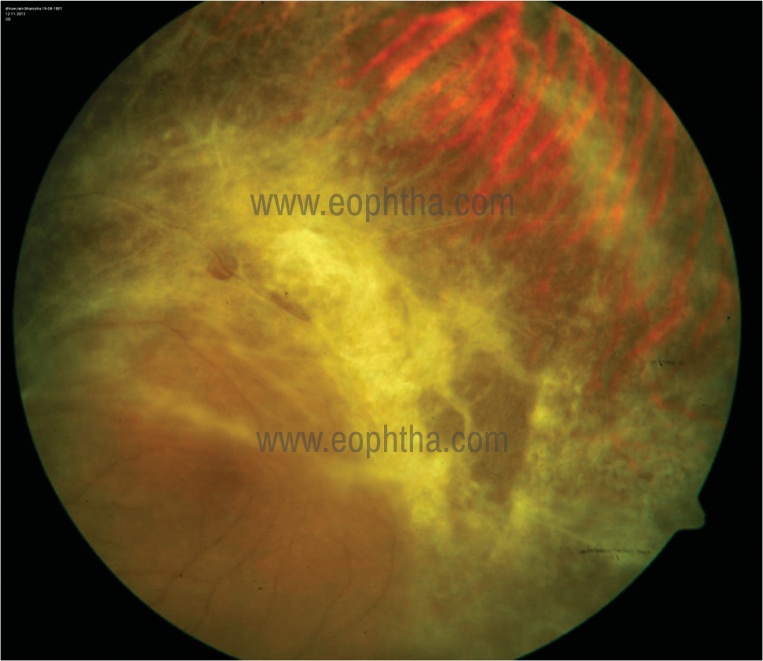
Figure 4: Healed CMV retinitis is clinically seen as retinal atrophy, fibrosis, and sclerotic vessels.
Zones of CMV Retinitis13:
Complete retina is divided into 3 zones to decide on treatment of CMV retinitis (Figure 5):
Zone 1: Area of the retina extending 3000 microns from the center of the fovea and 1500 microns from the edge of the optic nerve. Zone 1 disease is considered immediate sight threatening and require more urgent therapy than do more peripheral lesions.
Zone 2: Extends from the edge of zone 1 to a circle identified by the vortex ampullae.
Zone 3: Extends from the anterior edge of zone 2 to the ora serrata.
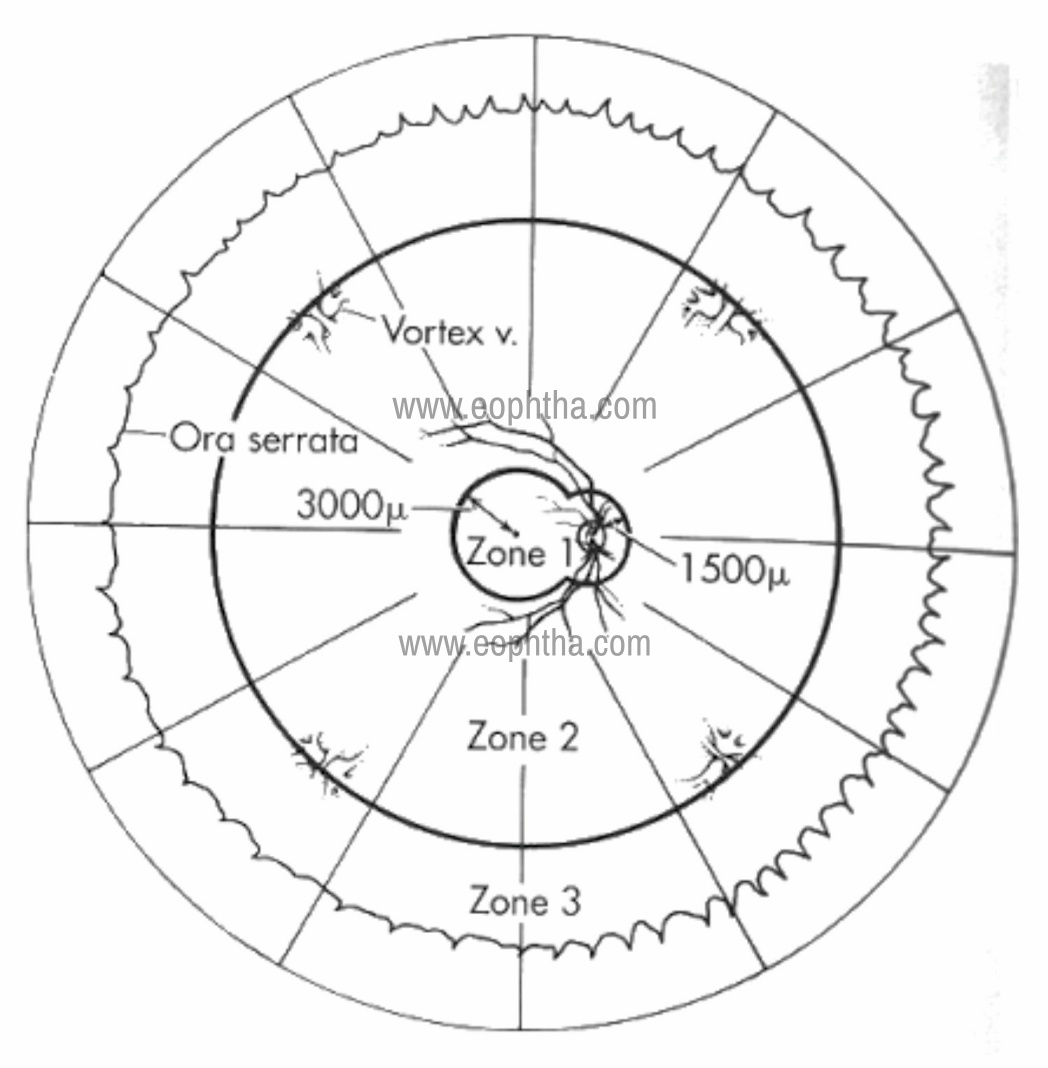
Figure 5: Various zones of CMV retinitis.
Differential diagnosis:
- In the early stage of CMV retinitis, it may mimic large cotton wool spots. Cotton wool spots don’t cause blurred vision and are not associated with vitritis and retinal haemorrhages. They have feathery borders rather than granular borders. Examining again after 1-2 weeks may help in making a diagnosis as they will not progress.
- In the later stage, it may resemble other infections of the retina mainly toxoplasmic retinochoroiditis, herpetic infection, bacterial or fungal infections like syphilitic retinitis, aspergillosis, or candida retinitis. (Table 1)
|
Sr No |
Features |
Acute Retinal Necrosis (ARN) |
Progressive outer Retinal Necrosis (PORN) |
CMV Retinitis |
Toxoplasmic retinitis |
|
1 |
Immune status |
Healthy/ rarely immunosuppressed |
Immunosuppressed |
Immunosuppressed |
Healthy but also seen in immunosuppressed |
|
2 |
Laterality |
Bilateral (30%) |
Bilateral (70%) |
Bilateral (30-50%) |
Unilateral in acquired disease |
|
3 |
Visual loss |
Initially mild, later gross |
Early loss of central vision |
Variable, depending on the site of involvement |
Early if macula involved or significant vitritis |
|
4 |
Anterior segment |
Mild to moderate uveitis May have granulomatous uveitis |
Mild non-granulomatous uveitis |
Mild non-granulomatous uveitis |
Mild to moderate granulomatous uveitis |
|
5 |
IOP |
Raised |
Normal |
Normal |
Normal/ Raised |
|
6 |
Vitreous reaction |
Significant vitritis |
Minimal/ No vitritis |
Minimal/ No vitritis |
Seen mainly overlying retinitis lesion may be significant |
|
7 |
Retinal involvement |
Full thickness |
Deep retinal involvement without granular border |
Full thickness involvement with granular borders |
Necrotising Retinochoroiditis surrounded by edema, occasionally adjacent to previous scar |
|
8 |
Pattern of involvement |
Multifocal, predominantly peripheral tongue shaped lesion progressing centripetally |
Multifocal, early macular involvement |
Usually unifocal, fovea relatively spared |
Usually unifocal, macula can get involve early |
|
9 |
Classic appearance |
Late swiss cheese |
Cracked mud |
Cottage cheese with pizza-pie |
Head light in fog appearance |
|
10 |
Vasculitis |
Common |
Uncommon |
Seen, but not common |
Seen, Kyrieleis plaques |
|
11 |
Retinal haemorrhages |
Common |
Uncommon |
Common in active lesion |
Uncommon, Rarely seen if associated with vascular occlusion |
|
12 |
Treatment |
Acyclovir Valacyclovir Famciclovir |
Limited success Acyclovir+Foscarnet Ganciclovir |
Ganciclovir, Valganciclovir, Foscarnet, Cidofovir |
Pyrimethamine+sulfadiazine, Clindamycin, Spiramycin |
Table 1: Differentiating features of various causes of Retinitis
Diagnosis:
Diagnosis of CMV retinitis is mostly based on characteristic ophthalmological findings. Retinal imaging including fundus photography, fundus fluorescein angiography, wide-field retinal imaging, and optical coherence tomography are useful in establishing the diagnosis and monitoring the response to treatment. Viral isolation, serodiagnosis, detection of the viral genome, and biopsy are ways for laboratory diagnosis.1,5
- Viral Isolation: Isolation of the virus in culture with detection of viral cytopathic effect and early nuclear antigen by immunofluorescence is an important diagnostic modality. Viral isolation is the gold standard for CMV diagnosis but is slow and often takes weeks to yield positive results. Aqueous fluid, vitreous sample, or subretinal fluid can be used for culture but the yield is low.
- Serodiagnosis: Different methods of serodiagnosis include enzyme-linked immunoassay, indirect hemagglutination, radioimmunoassay, and complement fixation. The presence of antibodies to CMV indicates prior infection but is neither indicative of concurrent clinical disease nor productive of future disease.
- Polymerase chain reaction: The tetraplex PCR analysis of intraocular fluids for HSV, VZV, and CMV are effective in confirming the etiological pathogen. It has a sensitivity of 80.9 - 84% and a specificity of 97.4 - 100%.1 An elevated CMV viral load is the most predictive risk factor for the end-organ disease.
- Biopsy: In atypical cases, endoretinal biopsy taken at the junction of necrotic and actively infected tissue may be necessary. The pathognomonic finding is the presence of intranuclear and intracytoplasmic inclusion bodies with an ‘owl’s eye’ appearance or positive CMV staining by immunohistochemistry. Electron microscopy also helps in demonstrating viral particles.
Treatment:
The mainstay of treatment in CMV retinitis is the reversal of immunodeficiency. Prior to the introduction of HAART, patients often experienced a catastrophic loss of vision due to CMV retinitis. The development of HAART in 1996 has dramatically changed the incidence rates of CMV retinitis and associated complications.15-16 As per Longitudinal Study of Ocular Complications of AIDS (LSOCA) study research groups (2012), the four-year cumulative incidence of CMV retinitis in AIDS patients with lowered CD4 counts was 7%, a 72% reduction in the post-HAART era.17 Kempen et al showed a 30% reduction in mortality in patients treated with systemic anti-CMV therapy along with HAART.18
FDA had approved four drugs for the treatment of CMV retinitis: ganciclovir, valganciclovir, foscarnet, and cidofovir; all are selective viral DNA polymerase inhibitors. All these drugs are virostatic, not virucidal. These drugs are generally commenced at higher induction doses for two to three weeks followed by maintenance doses to prevent relapses of retinitis. Individual drugs with doses and side-effects are given in Table 2. Anti-CMV drugs should be continued in the maintenance phase until patients have complied with HAART for at least 18 months, there are documented CD4+ counts ≥100 cells/μL for at least 3 months and the retinitis is quiescent with prolonged relapse-free interval.19
|
Drugs |
Induction phase |
Maintenance phase |
Laboratory tests |
Side effects |
Consideration |
|
Ganciclovir (Intravenous) |
5mg/kg IV BD x 14 days |
5 mg/kg IV per day |
1. Complete blood counts twice a week during induction, weekly during maintenance. 2. Serum creatinine monthly |
Anaemia, thrombocytopenia, neutropenia |
1. Dose adjustment for renal disease 2. Avoid other bone marrow suppressing medications 3. Cytokine support: (granulocyte colony- stimulating factor) for neutropenia (absolute neutrophil count <500/mm3) |
|
Ganciclovir (Oral) |
Not recommended |
1 gram PO TDS |
1. Complete blood count every1–2 weeks; 2. Serum creatinine monthly |
Anaemia, thrombocytopenia, neutropenia |
1. Dose adjustment for renal disease 2. Avoid other bone marrow suppressing medications 3. Cytokine support: (granulocyte colony- stimulating factor) for neutropenia (absolute neutrophil count <500/mm3) |
|
Ganciclovir (Intravitreal) |
2 mg/0.1 mL twice a week for 3 weeks |
2 mg/0.1 mL per week |
None |
Risk of endophthalmitis, cataract, increases IOP, uveitis, vitreous haemorrhage, retinal detachment |
Mainly used in Zone 1 disease |
|
Valganciclovir (Oral) |
900mg PO BD x 14 days |
900 mg PO OD per day |
Complete blood count weekly |
Bone marrow suppression, nausea, diarrhoea |
1. Avoid other bone marrow suppressing medications 2. Cytokine support for neutropenia |
|
Foscarnet (Intravenous) |
90 mg/kg BD x 14 days or 60 mg/kg TID |
90-120 mg/kg per day |
Serum creatinine, potassium, magnesium, calcium, phosphorous biweekly during induction, and weekly during maintenance |
Nephrotoxicity, hypokalaemia, hypocalcaemia, hypomagnesemia, genital ulcers, anaemia, headache |
1.Dose adjustment for renal disease 2. IV pre-hydration with 1 L 0.9% NaCl 3. Slow infusion if hypocalcaemia symptoms 4. Electrolyte replacement |
|
Foscarnet (Intravitreal) |
2.4 mg biweekly for 3 weeks |
2.4 mg weekly |
None |
Risk of endophthalmitis, cataract, increases IOP, uveitis, vitreous haemorrhage, retinal detachment |
Mainly used in Zone 1 disease and ganciclovir resistant cases |
|
Cidofovir (Intravenous) |
5 mg/kg per week x 14 days |
3–5 mg/kg IV every 2 weeks |
Serum creatinine, urinary protein, complete blood count before each infusion |
Nephrotoxicity, proteinuria, Fanconi- like syndrome, hypocalcaemia, neutropenia, uveitis, hypotony |
1. IV prehydration with 1 litre 0.9% NaCl 2. Oral probenecid 2 gram PO 3 hours before, 1 gram 2 hour after and 1 gram 8 hour after each infusion 3. Discontinue if IOP < 50% of baseline 4. Discontinue if dipstick proteinuria > 2+ or serum Creatinine increases by 0.5 mg/dL |
|
Fomivirsen (Intravitreal) |
300 microgram every 2 weeks for 4 weeks |
300 microgram every 4 weeks |
None |
Risk of endophthalmitis, cataract, increases IOP, uveitis, vitreous haemorrhage, retinal detachment |
Resistant cases |
Table 2: List of anti-CMV drugs
In a large randomized trial Foscarnet-Gancyclovir CMV Retinitis Trial, designed to compare both drugs in terms of relative efficacy and safety in patients with AIDS, revealed no difference in terms of the rate of retinitis progression.20 In 1996, HPMPC trial demonstrated the efficacy of cidofovir in 64 newly diagnosed CMV retinitis patients randomized to treatment and deferred arms.21 Further, the Spanish Cidofovir Study Group highlighted the efficacy of compassionate use of cidofovir in 51 patients on HAART with previously treated retinitis who had relapsed or were unresponsive to induction with other anti-CMV agents.22 Cidofovir has the advantage of weekly dose in the induction phase and fortnightly dose in maintenance phase because of its prolonged intracellular half-life but is highly nephrotoxic. Hence, it is used in ganciclovir resistant cases.
Valganciclovir was a useful addition to the existing list of anti-CMV drugs in 2000. It is an L-valyl ester prodrug of ganciclovir, with high oral bioavailability (60%) and convenient once daily dosing. In a randomized study involving 80 patients with CMV retinitis, a satisfactory response to induction therapy was achieved in 77% of patients assigned to intravenous ganciclovir and 72% assigned to oral valganciclovir.23 Compared with intravenous therapy, oral valganciclovir equally prevents and treats non-ocular CMV disease, and hence has become a preferred treatment option for most experts. (Figure 6)
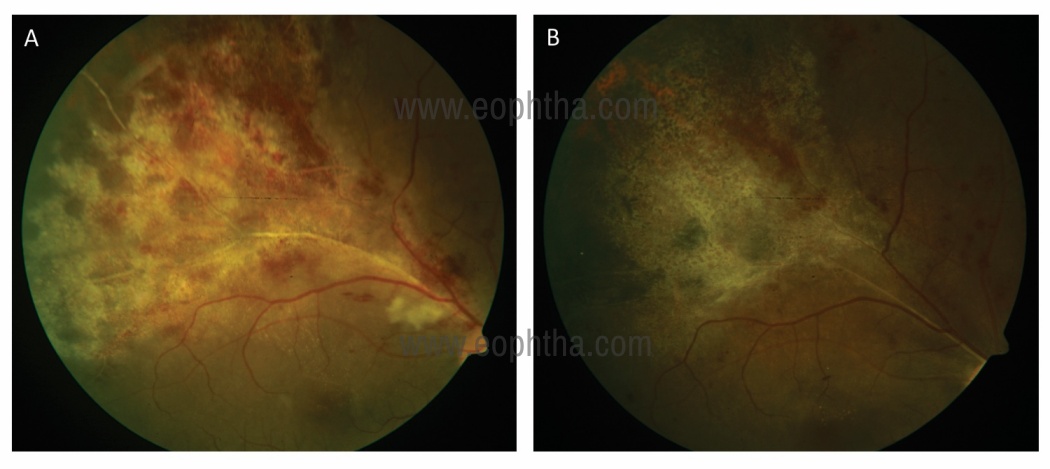
Figure 6: A 34-years-old patient suffering from AIDS (CD4 count: 41 cells/μL) presented with CMV retinitis in Zone 2 (A). He received Oral Valgancyclovir 900 mg twice a day for 14 days followed by 900 mg once a day. At 6 weeks on a maintenance dose, his CMV lesions healed with the atrophic retina and pigmentary changes.
Apart from systemic anti-CMV therapy, intravitreal drug injection of available drugs are important options especially in sight-threatening zone 1 disease.13,24 Various reports have documented the efficacy of intravitreal injections of ganciclovir and foscarnet solution. Cidofovir is no longer recommended due to its ocular toxicities like uveitis, hypotony, and occasionally, irreversible severe visual loss. In 1996, a sustained-release ganciclovir implant was approved for intraocular use.25 It releases intraocular ganciclovir at the rate of 1 μg/hour for up to eight months obviating the need for weekly injections during the maintenance phase.Although combining the ganciclovir implant with oral ganciclovir therapy has shown synergistic efficacy against the progression of retinitis, it is no longer commercially available due to the favorable ease of use of oral valganciclovir. Intravitreal therapy alone is associated with a higher risk of retinitis to the fellow eye, extraocular CMV-related diseases, and increased morbidity. Hence, systemic anti-CMV therapy should be added to local therapy. Table 3 shows the treatment algorithm for CMV retinitis.
|
Retinitis Location |
Highly Active Antiretroviral Therapy (HAART) |
|
|
Experienced |
Naive |
|
|
Zone 1 |
Oral Valgancyclovir + intravitreal ganciclovir |
Oral Valgancyclovir + intravitreal ganciclovir |
|
Zone 2 and 3 only |
Oral Valgancyclovir +/- intravitreal ganciclovir |
Oral Valgancyclovir |
Table 3: Treatment algorithm for CMV Retinitis
Resistance and Relapse:
Resistance is defined as persistent active retinitis lesions despite six weeks of induction therapy. It occurs in 25 to 35 percent of patients by nine months of therapy. Management includes combining therapies such as foscarnet with ganciclovir or adding an intravitreal injection to systemic therapy. In some patients, there may be a need to switch therapies altogether. Importantly subtherapeutic doses should not be confused with resistance.
The chance of relapse increases if CD4 counts are less than 100. In order to detect early relapses comparison of serial fundus photos is necessary. Relapse is defined as a quiescent CMV retinitis that becomes active. UL97 mutation may plan an important role in resistance and relapses. Hence, PCR sequencing for the detection of the UL97 mutation should be done. In case of relapse, current therapy should be continued at the induction schedule and compliance with HAART should be checked.
Complications:
1. Immune recovery uveitis (IRU):
The reported incidence ranges widely from 0.109 cases per person-year to 0.83 cases per person-year.26 Patients initially treated with cidofovir may be at higher risk for IRU. IRU usually occurs 2-16 weeks after the elevation of the CD4+ lymphocyte counts above 100 cells/μL, which is typically noted 2.5-8 months after starting HAART.27 Symptoms include painless decreased vision and floaters without any redness. The vision loss is generally mild. Ocular examination reveals moderate to severe vitritis, papillitis, cystoid macular edema, and /or epiretinal membrane. Rarely, the patient might present with vitreous haemorrhage, retinal neovascularization, optic disc neovascularisation, vitreomacular traction, proliferative vitreoretinopathy, posterior and anterior subcapsular cataracts, posterior synechiae, and angle-closure glaucoma.28 Eyes with inactive retinitis involving over 30% of the retinal area are at a higher risk of developing immune recovery uveitis when compared to eyes with less extensive involvement, presumably because those eyes with a larger area of CMV retinitis may generate a greater antigenic stimulus, prompting a more aggressive inflammatory response. Few small cohort studies have reported improvement in vision with periocular or systemic steroids. Contrarily, Zegans found that the vitreous cells resolved and vision improved within 6 weeks with or without anti-inflammatory therapy.29 Role of systemic steroids is controversial considering severe immunodeficiency in AIDS.
2. Retinal detachment:
Extensive retinal necrosis can lead to multiple large retinal tears and subsequently retinal detachment within 3-6 months of diagnosis.30 Retinitis lesions located in the periphery where the vitreous body is firmly adherent to the retina are more likely to be associated with retinal detachment than those located in the posterior pole. Eyes with large lesions and a longer duration of active lesions have a higher risk of retinal detachment. (Figure 7) Vitrectomy with long term tamponade with high-density oil is often necessary due to multiple holes in atrophic or gliotic retina.30,31
Prevention:
Reconstitution of immune system of the patient with HAART had dramatically reduced the morbidity and mortality in AIDS. Regular CD4 counts must be monitored in these patients with instituting secondary prophylaxis in the presence of immunological failure. Routine fundus screening should be done with indirect ophthalmoscopy inpatient with CD4 counts of < 50 cells/μL to detect peripheral lesions early. Routine use of primary prophylaxis with anti-CMV therapy is not recommended due to its high cost, potential toxicity, and concern about the development of drug resistance.
CMV Retinitis in non-HIV patients:
CMV retinitis in non-HIV cases has been observed in patients undergoing immunosuppressive therapies for hematologic malignancies, autoimmune disease, bone marrow and solid organ transplantation, and after local steroid administration.32,33 The clinical presentation is more or less similar in HIV and non-HIV patients. It has also been observed that vitreous inflammation, arteritis, and vascular occlusion outside of the areas of active retinitis can be more severe in non-HIV patients and the degree may vary depending on the level of immune suppression in non-HIV immunosuppressed patients. The treatment of CMV retinitis in HIV and non-HIV is similar requiring anti-CMV drugs in both the induction and maintenance phases. Induction therapy is typically administered for 14 to 21 days. The maintenance phase should be continued till there are signs of immune recovery. This can be achieved by either stopping or decreasing the dose of immunosuppressive drugs. The recurrence rate of 33 to 44% has been reported in various studies after discontinuation of anti-CMV therapy. The overall visual prognosis and clinical features of CMV retinitis have not been found to differ between HIV and non-HIV patients. Despite advances in anti-viral treatment, the visual prognosis of CMV retinitis remains poor. Poor prognosis is associated with retinal detachment during treatment, macular involvement, and poor general health of the patient. The incidence of retinal detachment also has not been found to significantly differ comparing HIV and non-HIV CMV retinitis patients.
References:
- Lee J, Agarwal A, Mahendradas P, et al. Viral Posterior Uveitis. Surv Ophthalmol 2017; 62:404-45.
- Jabs, DA, Van Natta, ML, Thorne, JE, et al. Course of cytomegalovirus retinitis in the era of highly active antiretroviral therapy. Ophthalmology 2004;111:2224-39.
- Jacobson MA, Stanley H, Holtzer C,
et al: Natural history and outcome of new AIDS-related cytomegalovirus retinitis diagnosed in the era of highly active antiretroviral therapy. Clin Infect Dis 2000; 30:231-3. - Yoser, SL, Forster, DJ, Rao, NA. Systemic viral infections and their retinal and choroidal manifestations. Surv Ophthalmol. 1993;37:313-52.
- Hennis H, Scott A, Apple D. Cytomegalovirus Retintis. Surv Ophthalmol 1989; 34:193-203.
- Alford CA: Chronic congenital infections of man. Yale J Biol Med 55:187-192, 1982.
- Rafailidis PI, Mourtzoukou EG, Varbobitis IC, et al. Severe cytomegalovirus infection in apparently immunocompetent patients: a systematic review. Virol J 2008;5:47.
- Davis MG, Kenney SC, Kamine J, et al. Immediate-early gene region of human cytomegalovirus trans-activates the promoter of human immunodeficiency virus. Proc Natl Acad Sci USA 1987;84:8642-6.
- Skolnik PR, Kosloff BR, Hirsch MS. Bidirectional interactions between human immunodeficiency virus type 1 and cytomegalovirus. J Infect Dis 1988;157:508-14.
- Skolnik PR, Pomerantz RJ, de la Monte SM, et al. Dual infection of retina with human immunodeficiency virus type 1 and cytomegalovirus. Am J Ophthalmol 1989;107; 261-72.
- Kotenko SV, Saccani S, Izotova LS, et al. Human cytomegalovirus harbors its own unique IL-10 homolog (cmvIL-10). Proc Natl Acad Sci U S A 2000;97:1695-700.
- Vrabec, TR. Posterior segment manifestations of HIV/AIDS. Surv Ophthalmol 2004;49:131-57.
- Jabs D. Cytomegalovirus Retinitis and the Acquired Immunodeficiency Syndrome – Bench to Bedside: LXVII Edward Jackson Memorial Lecture. Am J Ophthalmol 2011;151:198-216.
- Harper, TW, Miller, D, Schiffman, JC, et al. Polymerase Chain Reaction Analysis of Aqueous and Vitreous Specimens in the Diagnosis of Posterior Segment Infectious Uveitis. Am J Ophthalmol 2009;147:140-7.
- Deayton JR, Wilson P, Sabin CA, et al. Changes in the natural history of cytomegalovirus retinitis following the introduction of highly active antiretroviral therapy. AIDS 2000;14:1163-70.
- Skiest DJ, Chiller T, Chiller K, et al. Protease inhibitor therapy is associated with markedly prolonged time to relapse and improved survival in AIDS patients with cytomegalovirus retinitis. Int J STD AIDS 2001;12:659-64.
- Sugar EA, Jabs DA, Ahuja A, et al. Incidence of cytomegalovirus retinitis in the era of highly active antiretroviral therapy. Am J Ophthalmol 2012;153:1016-24.
- Kempen JH, Jabs DA, Wilson LA, et al. Mortality risk for patients with cytomegalovirus retinitis and acquired immune deficiency syndrome.Clin Infect Dis. 2003;37:1365-73.
- Guidelines for prevention and treatment of opportunistic infections in HIV-infected adults and adolescents. MMWR Morb Mortal Wkly Rep 2009;58(RR-4):55.
- Studies of ocular complications of AIDS. Foscarnet-ganciclovir cytomegalovirus retinitis trial: 1. Rationale, design, and methods. Control Clin Trials 1992;13:22-39.
- Parenteral cidofovir for cytomegalovirus retinitis in patients with AIDS: The HPMPC Peripheral Cytomegalovirus Retinitis Trial. A randomized, controlled trial. Ann Intern Med 1997;126:264-74.
- Berenguer J, Mallolas J. Intravenous cidofovir for compassionate use in AIDS patients with cytomegalovirus retinitis. Spanish Cidofovir Study Group. Clin Infect Dis 2000;30:182-84.
- Martin DF, Sierra-Madero J, Walmsley S, et al. A controlled trial of valganciclovir as induction therapy for cytomegalovirus retinitis. N Engl J Med 2002;346:1119-26.
- Stewart MW. Optimal management of cytomegalovirus retinitis in patients with AIDS. Clin Ophthalmol. 2010;4:285-99.
- Musch DC, Martin DF, Gordon JF, Davis MD, Kuppermann BD. Treatment of cytomegalovirus retinitis with a sustained-release ganciclovir implant. The Ganciclovir Implant Study Group. N Engl J Med 1997;337:83-90.
- Kempen JH, Min YI, Freeman WR, et al: Risk of immune recovery uveitis in patients with AIDS and cytomegalovirus retinitis. Ophthalmology 2006; 113:684-94.
- Karavellas MP, Lowder CY, MacDonald JC, et al: Immune recovery vitritis associated with inactive cytomegalovirus retinitis. A new syndrome. Arch Ophthalmol 1998; 116:169-75.
- Karavellas MP, Azen SP, MacDonald JP, et al: Immune recovery vitritis and uveitis in AIDS: clinical predictors, sequelae, and treatment outcomes. Retina 2001; 21:1-9
- Zegans ME, Walton RC, Holland GN, et al: Transient vitreous inflammatory reactions associated with combination antiretroviral therapy in patients with AIDS and cytomegalovirus retinitis. Am J Ophthalmol 1998; 125:292-300.
- Davis JL, Serfass MS, Lai MY, et al: Silicone oil in repair of retinal detachments caused by necrotizing retinitis in HIV infection. Arch Ophthalmol 1995; 113:1401-9.
- Singh R, Bhalekar S, Parchand S, et al. Outcome of surgery in post-cytomegalovirus retinal detachment: experience before and in the era of highly active anti-retroviral therapy in Indian eyes.Indian J Ophthalmol. 2013;61:636-9.
- Shapira Y, Mimouni M, Vishnevskia-Dai V. Cytomegalovirus retinitis in HIV-negative patients - associated conditions, clinical presentation, diagnostic methods and treatment strategy.Acta Ophthalmol 2018;96:e761-7.
- Parchand S, Barwad A. Cytomegalovirus Retinitis as a Presenting Feature of Multisystem Disorder: Dyskeratosis Congenita.Middle East Afr J Ophthalmol. 2017;24:219-21.

