One of the most dreaded complications of cataract surgery is a posterior capsular tear (PCR). Its rate ranges from 1.3% to 9.6% (from skilled surgeons to trainees)1-5. Early recognition of the PCR and skilled management is necessary for good surgical outcomes and to avoid any devastating sequelae. Hence, anterior vitrectomy is an important skill that every cataract surgeon should master.
There are some risk factors that may predispose to PCR like a true posterior polar cataract, dense and mature cataracts, old age (zonular weakness, small pupil, and denser cataract), uncooperative/ anxious patients, poor visualization due to corneal disease, pre-existing ocular trauma leading to capsular rupture/ zonular damage, etc.6,7
A thorough preparation beforehand may avoid confusion and lead to a well-managed surgery. The OT staff should be well aware of the kit to be used for anterior vitrectomy and the settings to be used for the same.
Early identification of the posterior capsular tear
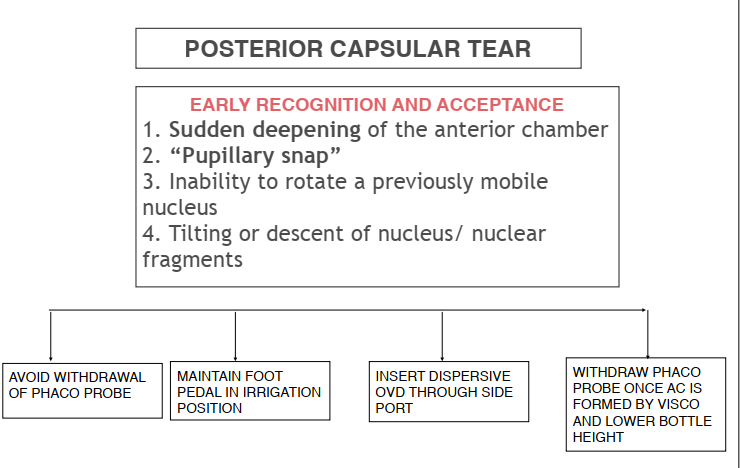
The early signs of capsular rent are:
- Sudden deepening of the anterior chamber (AC)
- “Pupillary snap” – sudden pupillary dilatation due to sudden equalization of pressure between anterior and posterior segments.
- Inability to rotate a previously mobile nucleus
- Tilting or descent of nucleus/ nuclear fragments
Once a PCR is recognized, the main aim of the surgery is to
- Avoid any further enlargement of the PCR
- Safely remove the remaining lenticular matter and avoid the remaining nuclear fragments/cortex from dislocating in the posterior segment
- To put an intraocular lens (IOL) safely either in the bag or in the sulcus
To achieve the above objectives, it is important to stabilize the anterior chamber. Any sudden pressure fluctuation may lead to enlargement of the PCR and the nucleus or cortical matter drop. To achieve this, an alert mind is extremely important. The surgeon should avoid sudden withdrawal reflex of the phaco probe. Instead, it should be maintained in the irrigation mode (usually foot pedal-1 position) and a dispersive ophthalmic viscosurgical device (OVD) should be inserted into the anterior chamber before the withdrawal of the instruments. (Fig 1)
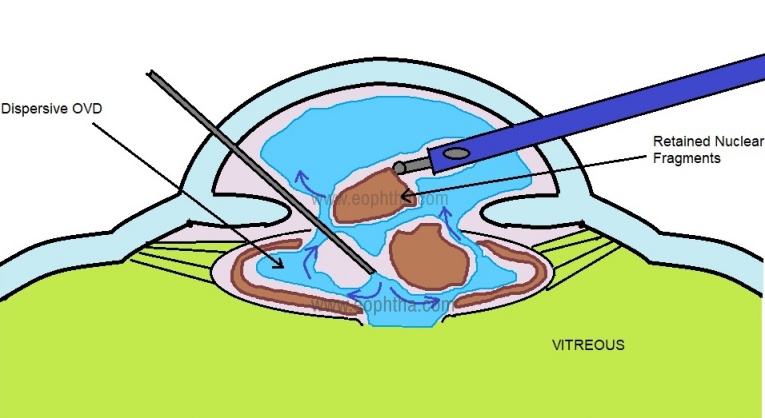
Figure 1: Stabilizing the anterior chamber with dispersive OVD before withdrawing the phaco probe to avoid any enlargement of the posterior capsular tear
Once OVD is inserted, it avoids the vitreous prolapse. This is the time to withdraw the phaco probe and reassess the situation for further management. Important questions include
- How much nucleus/cortex is still remaining in the eye?
- The site and extent of a capsular tear!
- Has any vitreous prolapsed into the Anterior Chamber?
MANAGEMENT OF RETAINED NUCLEAR FRAGMENTS
Assessment starts with injecting dispersive OVD liberally behind the residual lens material through the side port. The aim is to tamponade vitreous posteriorly and holds the lens fragments anteriorly.
In case the PCR happens in later stages when smaller nuclear fragments are remaining, the surgeon has two options
- Continue phaco with a stabilized anterior chamber and using LOW VACUUM and LOW IRRIGATION (low bottle height and low flow rate) setting. (Fig 2) Minimal cracking should be done as creating smaller pieces may increase the risk of these pieces falling into the vitreous cavity. Absolute care should be taken not to engage vitreous at any time in the phaco probe. A sheet glide may be used to complete phaco.
- To direct the small nuclear pieces with OVD toward the main port for delivery. The wound may slightly be enlarged to assist in viscodelivery.
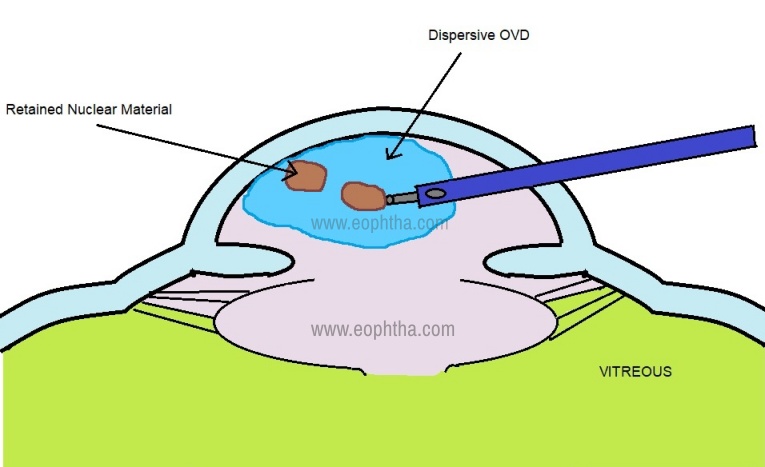
Figure 2: Controlled phacoemulsification of the small nuclear fragments retained after PCR, taking care not to engage any vitreous
However, if a larger nuclear fragment is left in the AC, it may be deemed necessary to convert to an Extracapsular Cataract Extraction (ECCE). Dispersive OVD is injected below the nucleus. Conjunctival peritomy may be done, followed by enlargement of the limbal incision. The incision is made slightly larger than the largest nuclear fragment to be removed. Careful extraction of the nucleus with a serrated lens loop or a sinskey hook and spatula can be used. The nucleus should not be pressed against the cornea and pulled out. This can cause severe endothelial loss. Care should be taken at all times to avoid engaging vitreous. Excessive external pressure must be avoided as it may prolapse more vitreous.
If the nuclear fragments or the entire nucleus has displaced posteriorly into the vitreous cavity, no blind attempts should be made to retrieve the nuclear fragments. The surgeon should proceed with anterior vitrectomy with the aim to close the wound securely and placement of stable IOL if possible. It is preferable that a vitreoretinal surgeon manages the dropped nucleus by a complete pars-plana vitrectomy. (Flowchart 1)
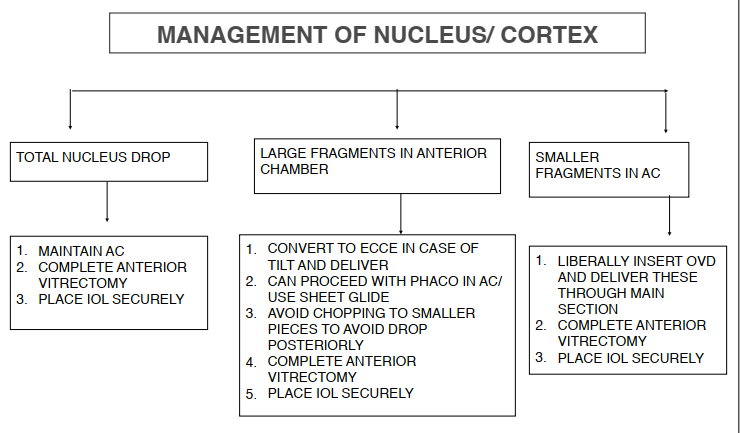
RESIDUAL CORTEX MANAGEMENT
The cortical matter should be removed with low flow bimanual aspiration. The aim is to avoid any turbulence and hence the enlargement of the PCR. The aspiration tip should be brought in contact with the cortex first before any vacuum is applied, in order to avoid aspirating the vitreous. Stripping the cortex should be done towards the tear, as stripping away will enlarge the tear. Alternately, a cutter may be used for cortex removal which is mentioned below.
MANAGEMENT OF NUCLEUS TILT
Charles Kelman 8, proposed a popular technique called posterior-assisted levitation, or ”PAL” technique to retrieve a tilted or partially descended nucleus. It involves levitation of the nucleus by a metal spatula, inserted through a pars plan incision. The technique was subsequently modified by Richard B. Packard (9), who used Viscoat, inserted by cannula through pars plana sclerotomy, to support and levitate the nucleus, popularly known as Viscoat PAL technique.
The procedure involves, buttonholing the conjunctiva and then making a pars plan sclerotomy with a microvitreoretinal (MVR) blade, 3.5 mm behind the limbus. Thereafter, the cannula is inserted and dispersive OVD is inserted below the tilted end of the nucleus, avoiding overinflation of the globe. The aim is to support and reposition the descending nucleus. This is followed by manual levitation of the nucleus with the cannula. In case the capsulorrhexis or the pupil is small, the push of the OVD from behind helps to stretch them and accommodate a nucleus of larger size. Once in the anterior chamber, dispersive OVD is injected both in front and behind the nucleus and delivered with irrigating serrated lens loop as described earlier. Since no suction is involved in this technique, the vitreous disturbance is minimal. However, every surgeon must judge his skills and confidence, before attempting alternative maneuvers.
MANAGEMENT OF VITREOUS PROLAPSE
The major complication that happens due to PCR is improper management of the prolapsed vitreous which may be indirectly suggested by peaking of the otherwise round pupil. Any vitreous incarceration may lead to traction and retinal tears besides increasing the risk of endophthalmitis.
It is also important to clear the vitreous from the anterior chamber for the stable placement of IOL. This can be achieved by both anterior and posterior routes.
1. BIMANUAL ANTERIOR VITRECTOMY
It is essential to maintain adequate anterior chamber pressure in order to hold the vitreous posteriorly. Hence an infusion line is placed first in the anterior chamber through the existing limbal side port and directed towards the angle, always above the pupillary plane. This is very important because a posteriorly directed irrigation causes hydration of the vitreous body and further prolapse of the vitreous anteriorly, which must be completely avoided.
The stability of the main port is assessed. In case of any doubt, it should always be closed with a suture. This also helps in case an additional vitreoretinal procedure has to be planned for dropped fragments. A second side port needs to be made for the cutter. In case the main port was superior, the side port is planned around 9 o'clock and in case it is temporal, the port for the cutter is made at 6 o'clock. Cutter is inserted through the second side-port, maintaining a closed system. It is directed through the tear posterior to the capsule (below the capsular plane), with the tip pointing inwards towards the optic nerve and the aspiration port facing the cornea. With the cutter tip under the posterior capsule and infusion cannula pointed towards the angle, the vitreous bands are severed at the level of capsular tear and the vitreous is aspirated towards the cutter. The cutter tip should be kept stable at a place, as excess movement may tend to cut the posterior capsule and exert undue traction on the vitreous.
The cutter tip should be deep enough to avoid any engagement with the posterior capsule, but superficial enough to be under direct visualization of the operating microscope. Care is taken to cut the vitreous at first before applying any vacuum. This is done till the anterior segment is free of vitreous strands.
Although Vitrectomy should be thorough, overzealous vitrectomy should be avoided as excessive manipulation in the anterior vitreous body may cause peripheral retinal injuries. The cut-rate should be the maximum that the machine offers (varying from 600 cuts/ min to 5000 cuts/min) and vacuum can range from 100-150 mm Hg depending upon the cut-rate. Following this, the cutter is used to remove any cortical lens matter in the bag. “IA- Cut mode” is used during this maneuver. The cut-rate is reduced and the vacuum is slightly increased. Most of the softer lens matter is just aspirated, however, some firm matter can be cut. The cortex is freed from the capsular bag and drawn into the center, the cutting action is used to remove it.
The visualization of the vitreous may pose a challenge for a good vitrectomy. In such a situation preservative-free intracameral triamcinolone acetone may be used to stain vitreous and identify any residual thin strands (10). Triamcinolone acetate (40mg/ml) is commercially available. This is diluted with BSS (1:1) before using it in the anterior chamber. Triamcinolone granules stain the vitreous and also provide an intraocular anti-inflammatory effect. One must use this technique in case a good anterior vitrectomy has not been achieved and the pupil still looks peaked.

2. SINGLE PORT PARS PLANA VITRECTOMY
Although the anterior segment surgeon may find it challenging, the Pars Plana route is definitely a more effective route to completely clear the vitreous from the anterior chamber. A single port is made 3- 3.5 mm posterior to the limbus, with infusion placed through the side port. In the case of 20 G, the conjunctiva is reflected in that area while for 23 G/ 25 G, transconjunctival placement of trocar is done. A bevelled entry is preferred, to create a self-sealing incision. The tip of the cutter must be visualized all the time, through the pupil, as the vitrectomy proceeds. The advantage of this technique is that the vitreous flow from the anterior chamber to the posterior segment getting cleared quickly,(Fig 3) unlike the bimanual anterior vitrectomy where the vitreous keeps getting hydrated and prolapsing into the AC. The technique is simpler than it is believed to be and the skill must be acquired by all anterior segment surgeons.
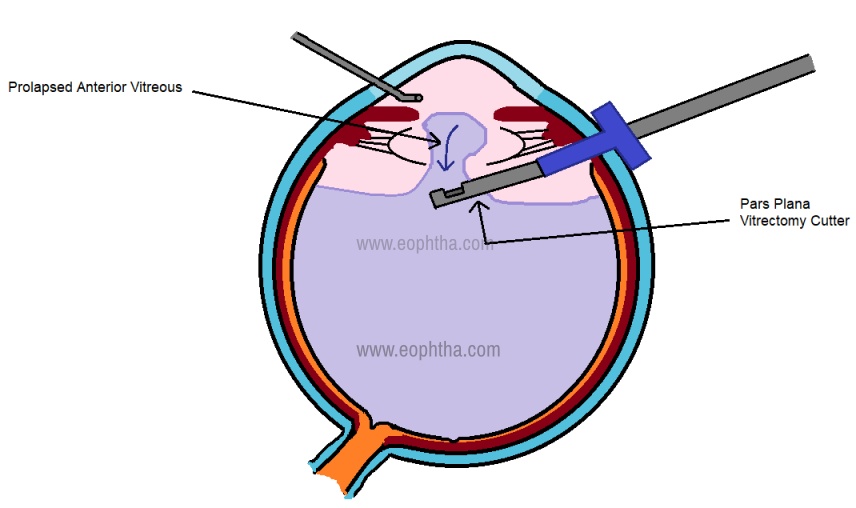
Figure 3: Single port pars plana vitrectomy, showing how vitreous moves anterior to posterior and gets cleared effectively from the anterior chamber.
IOL IMPLANTATION
An IOL implantation must be done by the cataract surgeon if safely possible, regardless of any dropped fragments, to be removed later by the vitreoretina surgeon. The options include:
- If the PCR is small and central, then using the Utrata capsulorrhexis forceps, the capsular tear can be converted to smooth posterior capsulorrhexis (PCC), and then in the bag IOL placement can be done.
- In case the PCR is large, IOL can be placed in the sulcus. The overall diameter of the lens to be implanted in the sulcus should be preferably more than 13 mm to ensure stability. The IOL power also needs to be reduced by 0.5D while being placed in the sulcus for best visual results. In case the anterior capsulorrhexis is small and central, the optic may actually be tucked behind it creating an optic capture with haptic in the sulcus, providing better lens stability. However, if it is eccentric then this may not be possible.
- In case of extensive capsular loss, the surgeon may sometimes need to place an anterior chamber lens (open loop), an iris-claw lens or sulcus fixated/ glued IOL depending upon the familiarity of the surgeon to the procedure. The IOL powers need to be adjusted according to the site chosen for lens placement. These procedures preferably should be done later and not as a primary procedure.
CLOSURE
After the three steps are over - nuclear and cortical cleanup, anterior vitrectomy, and placement of the intraocular lens, all the wounds need to be closed well. The viscoelastic should be washed thoroughly. Triamcinolone can be reused at this stage to identify any residual vitreous. Dry anterior vitrectomy may be performed for a thin strand identified at this stage. This can be achieved by pushing the visco simultaneously through the side port, as a vitrectomy is done to prevent the collapse of the anterior chamber. All wounds must be checked for any vitreous strand incarceration. The suture must be placed in case of wound instability or if a vitrectomy is planned soon for dropped fragments.
POSTOPERATIVE MANAGEMENT
The three main issues to be tackled include:
- Postoperative Inflammation: Due to excess manipulation, the inflammation may be exaggerated and may need management with frequent topical steroids and cycloplegics.
- Raised Intraocular pressure (IOP): The main causes of raised IOP could be excess inflammation, retained viscoelastic and the triamcinolone used to stain vitreous. Most of them can be managed medically with appropriate anti-glaucoma medications.
- Retinal status: Even with the most efficient management of PCR, the retina may develop tears and lead to catastrophic events of retinal detachment. Hence, a detailed retinal examination must be done at the earliest when the view is clear and must be continued into the postoperative period. Late retinal sequelae can happen in complicated cataract surgeries and hence patient must be explained about the warning signs. The sudden appearance of floaters, excessive flashing of light, and appearance of the fixed curtain in the field of view must be reported to the doctor at the earliest possible.
A posterior capsular rent during cataract surgery is not an unusual occurrence. A surgeon must always be prepared to handle such a situation with calm and efficient management. A well-managed case leads to a very satisfactory outcome in the majority of cases.
10 step approach to the management of posterior capsular tear
- Go bimanual - use both hands effectively
- Maintain a closed stable chamber always
- Infusion line to be re-secured through side port with flow directed toward the angle
- Staining of vitreous with triamcinolone acetonide
- Removing all vitreous from the anterior chamber and checking for any incarceration in the wound with repeat staining
- Keep bottle height low
- Use a higher cut-rate for cutting
- Keep vacuum low to moderate for aspiration
- Preserve as much capsule as possible
- Ensure safe and stable placement of an intraocular lens
REFERENCES
1. Martin KRG, Burton RL. The phacoemulsification learning curve: Per-operative complications in the first 3000 cases of an experienced surgeon. Eye. 2000; 2. Hashemi H, Mohammadpour M, Jabbarvand M, Nezamdoost Z, Ghadimi H. Incidence of and risk factors for vitreous loss in resident-performed phacoemulsification surgery. J Cataract Refract Surg. 2013; 3. Ionides A, Minassian D, Tuft S. Visual outcome following posterior capsule rupture during cataract surgery. Br J Ophthalmol. 2001; 4. Zare M, Javadi MA, Einollahi B, Baradaran-Rafii AR, Feizi S, Kiavash V. Risk factors for posterior capsule rupture and vitreous loss during phacoemulsification. J Ophthalmic Vis Res. 2014; 5. Turnbull AMJ, Lash SC. Confidence of ophthalmology specialist trainees in the management of posterior capsule rupture and vitreous loss. Eye. 2016; 6. Blomquist PH, Morales ME, Tong L, Ahn C. Risk factors for vitreous complications in resident-performed phacoemulsification surgery. J Cataract Refract Surg. 2012; 7. Narendran N, Jaycock P, Johnston RL, Taylor H, Adams M, Tole DM, et al. The Cataract National Dataset electronic multicentre audit of 55 567 operations: Risk stratification for posterior capsule rupture and vitreous loss. Eye. 2009; 23(1):31-7. 8. Kelman C. New PAL method may save difficult cataract cases. Ophthalmology Times.1994;19:51. 9. Chang DF, Packard RB. Posterior assisted levitation for nucleus retrieval using Viscoat after posterior capsular rupture. J Cataract Refract Surg. 2003;29:1860-1865. 10. Burke, S.E. et al. J Cataract Refract Surg 2003;29:645-651.


