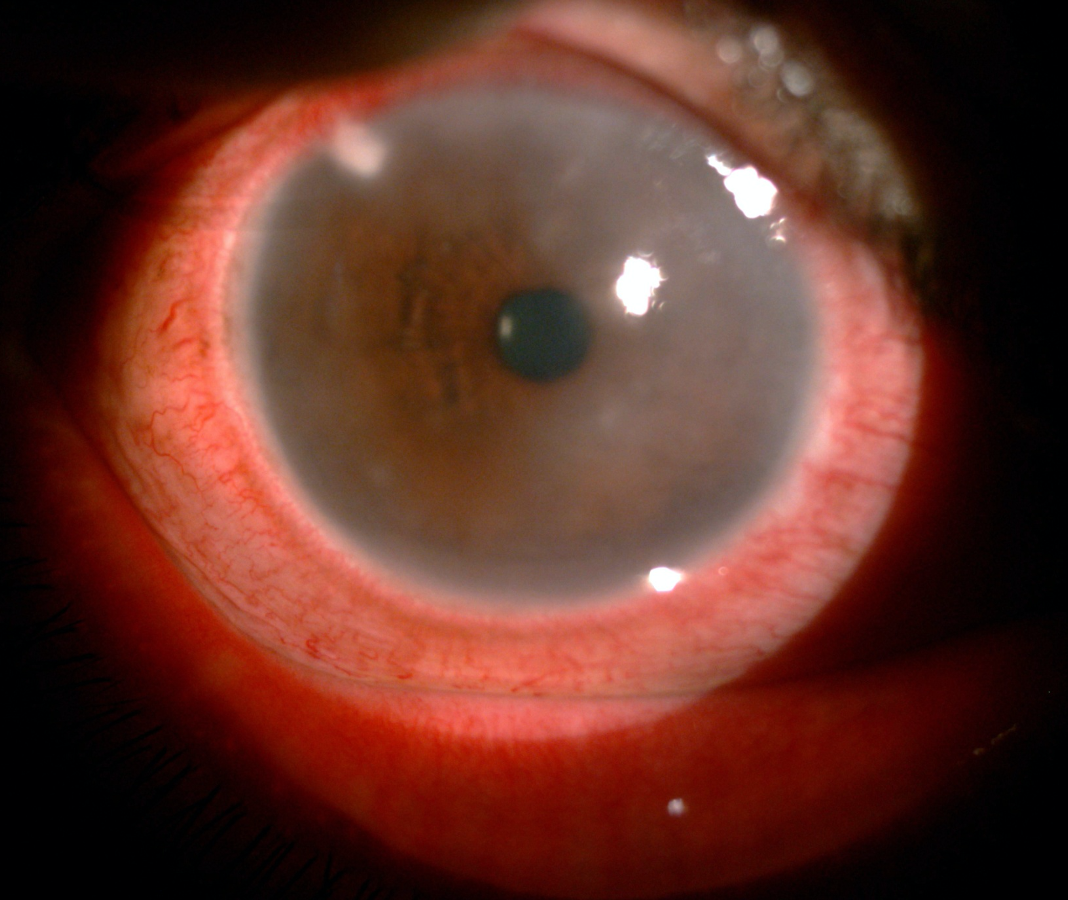
Corneal edema is defined as the increase in the thickness of cornea due to the accumulation of extracellular fluid in epithelium and stroma resulting in loss of corneal transparency.
Pathophysiology:
Cornea remains in a relatively dehydrated state maintaining a 78% hydration level. Corneal hydration depends on 5 factors.
1. Stromal swelling pressure (SP)
- This is the tendency of stroma to swell due to interfibrillary proteoglycans and other proteins.
- The normal stromal pressure is 55mmHg.
- Imbibition pressure (IP) – It is a negative pressure exerted by glycosaminoglycans by which fluid is drawn into the cornea.
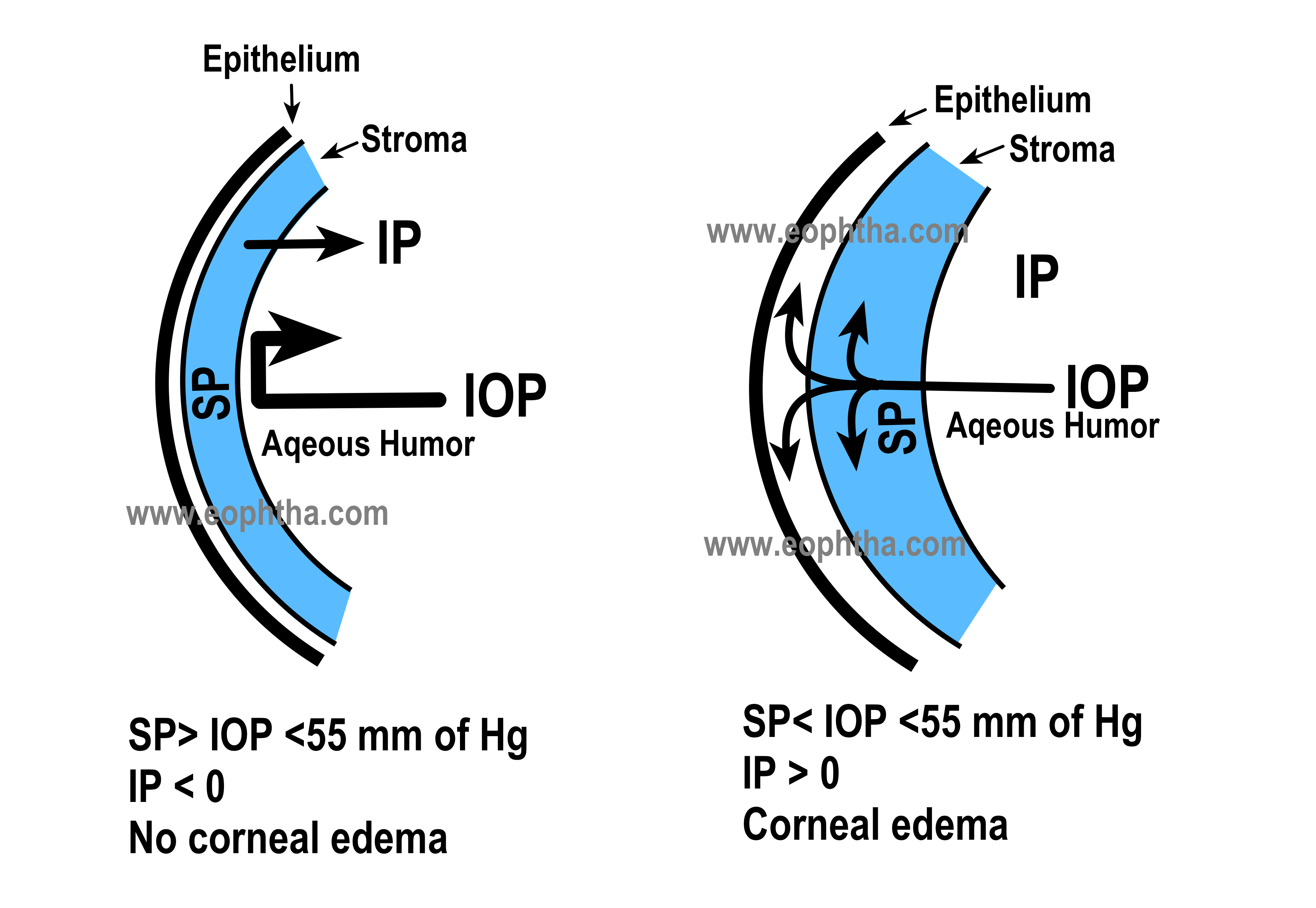
- Intraocular pressure (IOP) = SP + IP
2. Barrier function
The epithelium (Zonula occludens tight junction) offers twice the resistance to water flow compared to the endothelium (macula occludens tight junction) and the electrolyte resistance is 200 times higher in the epithelium than endothelium.1
3. Endothelial pump
- The endothelium pump function ensures, through active transport, the passage of fluid out of the corneal stroma into the aqueous humor.
- Corneal endothelial permeability gradually increases as central endothelial cell density decreases below 2000 cells/mm2
- Compensatory metabolic pump mechanisms maintain the dehydrated state until a central endothelial density of 500cells/mm2 is reached.
4. Tear evaporation
- Normal tear evaporation (rate 2.5ml/cm2/hour ) makes the tear hypertonic resulting in osmotic extraction thus thinning the cornea by 5%. But this loss is readily replaced by aqueous.
- Evaporation can be a factor in maintaining epithelial dehydration, as is observed in the diurnal variations of visual acuity in patients with the early stage Fuchs’ endothelial dystrophy.
5. Intraocular Pressure
When the IOP (> 50 – 60 mm Hg) exceeds the stromal pressure epithelial edema occurs. e.g. High IOP and normal SP – Acute glaucoma, normal IOP and low SP – endothelial dystrophy
Figure: First one- When the IOP does not exceed the level of stromal swelling pressure (SP), negative imbibition pressure (IP), which is dependent upon an intact endothelial barrier, prevents humor movement toward the corneal stroma and the subepithelial space. Second one-When IOP increases and exceeds the level of SP, the corneal endothelial barrier is disturbed, resulting in water accumulation in the stroma and subepithelial layers (corneal edema).
Etiology
|
Dystrophic |
|
|
|
|
|
Inflammatory |
|
|
|
|
|
Toxic |
|
|
|
|
|
Mechanical |
|
|
Acute hydrops in keratoconus does not fit neatly into any of the above headings, as the basic pathology does not involve the endothelium, but rather the stroma. With progressive stretching of the stroma, sudden breaks in Descemet’s membrane may appear, exposing the underlying stroma directly to the aqueous.
Symptoms
Decrease in vision
- Slight epithelial edema may cause significant visual disturbance
- 70% stromal edema also can keep vision unaffected
Pain
Photophobia
Colored Haloes
Post Cataract Surgery Corneal Oedema
Every cataract surgeon is apprehensive of this dreaded complication.The causes for post-cataract surgery corneal edema can be multifactorial and can be subdivided into :
Mechanical Injury
- Surgical Trauma - Operative factors associated with the corneal endothelial injury include Ultrasound energy
- The turbulence of the irrigating solution
- Ricocheting of nuclear fragments
- IOL contact
- Phacoemulsifier tip trauma to the endothelium
- Desçemet’s detachment
- Prolonged phacoemulsification time
- Need for anterior vitrectomy and irrigation/aspiration
Inflammation/ Infection
- Endophthalmitis
- Herpetic Endothelitis
- Retained lens material
- Brown- McLean Syndrome
Chemical Injury - TASS – Toxic Anterior Segment Syndrome 2-9
It is the sterile postoperative anterior segment inflammation within 12–48 hours of surgery. It is associated with minimal pain, diffuse limbus-to-limbus corneal edema, fibrinous AC reaction, iris atrophy, trabecular meshwork damage but the absence of vitritis.
The typical offending agents are
- Antiseptic solutions used for pre-surgical preparation
- Topical and intracameral anesthetics
- Irrigating solution
- Intraocular medications and preservatives
- Viscoelastic
- Detergent residues on ophthalmic surgical instruments
Concurrent Eye Disease
Primary Corneal Endothelial Disease: Pre-operative low endothelial cell count as in patients with Fuchs’ endothelial dystrophy can result in endothelial decompensation after cataract surgery due to low reserve.
A small portion of the population has a low endothelial cell density that is not heralded by the presence of corneal guttae. The other eye should be examined for the endothelial count.
High IOP/ Glaucoma
Preoperative Risk Assessment
Juan G et al17 has proposed an endothelial risk matrix as below:
- Low risk (0-5)
- Moderate risk (6-10)
- High risk (11-19)
- Very high risk (20+)
|
Endothelial Risk Score |
0 |
1 |
2 |
3 |
4 |
|
Krachmer Classification of FED |
0 |
1-2mm area of guttae |
3-4mm area of guttae |
5mm area of guttae |
6mm area of guttae |
|
Cell Count |
>2000 |
< 2000 |
<1500 |
<1000 |
<500 |
|
CV & Hexagonality |
<33 & >50% |
<40 & >45% |
>40 & <39% |
>50 & <30% |
>60 & <20% |
|
Ultrasound Pachymetry |
<540 |
540-575 |
576-624 |
625-649 |
>650 |
|
AC Depth |
>4mm |
>3.5mm |
<3.5mm |
<2.7mm |
<2mm |
|
LOCS III |
NO 1 or 2 |
NO 3 |
NO 4 |
NO 5 |
NO 6 |
Other factors include advanced age, systemic diseases (DM, COPD, Renal insufficiency)18 etc.
Clinical Examination
A careful Slit Lamp examination is of utmost importance to ascertain the cause of edema.
Extent of edema
Localized edema – Descemet Membrane Detachment, Instrument touch, Vitreous wick, Herpetic keratitis
Predominantly stromal edema – Check IOP
Limbus to limbus edema – TASS
Shape of pupil
Peaked Pupil – look carefully for the presence of vitreous
Dilated pupil – TASS – iris sphincter muscle damage
Patchy atrophy of iris – Herpetic etiology
Anterior Chamber
Reaction / Hypopyon – Endophthalmitis, TASS
Vitreous strands
Hyphema
Retained lens material
Keratic Precipitates
Investigations
Pachymetry
There are many devices that can be used to estimate corneal thickness. But corneal edema offers some significant challenges for the machines to record corneal thickness accurately. Nida et al19 published the Comparison of central corneal thickness measurements in corneal edema using ultrasound pachymetry, Visante anterior-segment optical coherence tomography, Cirrus optical coherence tomography, and Pentacam Scheimpflug camera tomography in 2018 in which the conclusion was that “all devices reliably measured the CCT 650 μm. In eyes with edema exceeding 650 μm, CCT measurements from the Visante OCT, Cirrus OCT, and ultrasound pachymetry devices showed good reproducibility and were well correlated, while the Pentacam overestimated the values compared to the other devices. Pentacam and ultrasound pachymetry should not be used in eyes with extreme corneal edema and opacity.”
Specular Microscopy
In corneas with significant corneal edema often the specular microscopy fails to give any reading. In such cases, the other eye can be checked for the following factors. It is a very useful tool for preoperative screening in suspected corneas.
ASOCT
This is the single most important investigation to find the clue for the diagnosis of corneal edema like descemet membrane detachment if not seen clinically. This is a reliable method to ascertain the corneal thickness in huge corneal edema.
Treatment
Medical
Eliminating the treatable causes such as inflammation, infection, high IOP, IOL contact, and Descemet’s membrane detachment.
- In mild cases only, hypertonic solutions, typically 5% sodium chloride ophthalmic preparation or 6% ointment., can improve corneal edema by enhancing surface dehydration.
- Bandaged contact lenses (BCL), especially extended-wear hydrophilic contact lenses are useful in reducing pain associated with epithelial bullae.10 They act by creating an effective precorneal protective layer that shields the swollen epithelium from the lid movement and prevents the rupture of bullae.11,12 Considering the potential risk of infection, prolonged use of BCL is discouraged, and a broad-spectrum antibiotic must be used along with these lenses.
- Raised IOP can be managed with topical antiglaucoma medications or surgical options such as trabeculectomy with mitomycin C or a glaucoma drainage implant in cases not controlled with topical therapy. carbonic anhydrase inhibitors and Prostaglandin analogues (PGA) must be avoided.13Inhibition of corneal endothelial carbonic anhydrase pumps can lead to decreased fluid flow from stroma to aqueous, resulting in precipitation of corneal edema. PGA, theoretically, can flare up the intraocular inflammation.13
- Associated inflammation must be treated with topical steroids.
Surgical
Descemetopexy for Descemet Membrane Detachment
The standard treatment option remains descemetopexy.In the immediate postoperative phase, intracameral air or gas injection is practiced .14
Kumaret al.15proposed HELP (height, extent, length, and pupil-based) treatment protocol for DMD.
DMD <1.0 mm long and <100 μm high in any zone, medical management was considered.
DMD 1–2 mm in length and 100–300 μm high in zone 2 (paracentral 5–8 mm) and 3 (periphery >8 mm) were managed medically.
DMD >2 mm in length and >300 μm high in zone 1 (central 5 mm) and 2 (paracentral 5–8 mm) were managed surgically and in zone 3 (periphery >8 mm) were managed medically.
Surgical intervention included intracameral gas (14% perfluoropropane [C3F8] or 20% sulfur hexafluoride [SF6]) or sterile air injection.
Toxic Anterior Segment Syndrome
In the acute phase, the intensive use of topical corticosteroids and systemic steroids (in severe cases) are advocated along with the use of antiglaucoma drugs. 16In nonresolving cases, for at least 3 months endothelial keratoplasty is advised.
Pearls for keratoplasty in corneal edema
Timing
If no underlying etiology is identified a minimum waiting period of 3 months is observed before opting for keratoplasty.
Type of surgery
Visibility of anterior camber details and anterior chamber depth are two most important determinants in choosing the type of keratoplasty needed.
- If the anterior chamber is very shallow with the dense stromal scar is present, penetrating keratoplasty is the best option for visual recovery.
- If anterior chamber depth is reasonable then even with some haze in the anterior stroma, DSEK/ DSAEK is the best option for visual recovery.
- DMEK requires good visibility of anterior chamber so no haze or minimal haze in anterior stroma yields the best visual outcome.
In all cases of corneal edema with vitreous loss, a good vitrectomy has to be performed along with grafting. IOP issues to be tackled efficiently to get the best results.
References
- Kreutziger GO. Lateral membrane morphology ad gap junction structure in rabbit corneal endothelium. Exp Eye Res 1976; 23: 285
- MacRae SM, Brown B, Edelhauser HF. The corneal toxicity of presurgical skin antiseptics. Am J Ophthalmol. 1984;97:221–232.
- Judge AJ, Najafi K Lee DA, Miller KM. Corneal endothelial toxicity of topical anesthesia. Ophthalmology. 1997;104:1373–1379.
- Kadonosono K, Ito N, Yazama F et al. Effect of intracameral anesthesia on the corneal endothelium. J Cataract Refract Surg. 1998;24:1377–1388.
- Edelhauser HF, Van Horn DL, Schultz RO, Hyndiuk RA. Comparative toxicity of intraocular irrigating solutions on the corneal endothelium. Am J Ophthalmol. 1976;81:473– 481.
- Hyndiuk RA, Schultz RO. Overview of the corneal toxicity of surgical solutions and drugs and clinical concepts in corneal edema. Lens Eye Tox Res. 1992;9:331–350.
- Kim JH. Intraocular inflammation of denatured viscoelastic substance in cases of cataract extraction and lens implantation. J Cataract Refract Surg. 1987;13:537–542.
- Olson RJ. Air and the corneal endothelium: an in vivo specular microscopy study in cats. Arch Ophthalmol. 1980;98: 1283–1284.
- Breebaart AC et al. Toxic endothelial cell destruction of the cornea after routine extracapsular cataract surgery. Arch Ophthalmol. 1990;108:1121–1125.
- Narayanan R, Gaster RN, Kenney MC. Pseudophakic corneal edema: A review of mechanisms and treatments. Cornea 2006;25:993-1004
- Arora R, Jain S, Monga S, Narayanan R, Raina UK, Mehta DK,et al. Efficacy of continuous wear PureVision contact lenses for therapeutic use. Cont Lens Anterior Eye 2004;27:39-43
- Lim L, Tan DT, Chan WK. Therapeutic use of Bausch and lomb PureVision contact lenses. CLAO J 2001;27:179-85
- Bodh SA, Kumar V, Raina UK, Ghosh B, Thakar M. Inflammatory glaucoma. Oman J Ophthalmol 2011;4:3-9
- Sharma N, Gupta S, Maharana P, Shanmugam P, Nagpal R, Vajpayee RB,et al. Anterior segment optical coherence tomography-guided management algorithm for descemet membrane detachment after intraocular surgery. Cornea 2015;34:1170-4
- Kumar DA, Agarwal A, Sivanganam S, Chandrasekar R. Height-, extent-, length-, and pupil-based (HELP) algorithm to manage post-phacoemulsification Descemet membrane detachment. J Cataract Refract Surg 2015;41:1945-53
- Arslan OS, Unal M, Arici C, Görgün E, Yenerel M, Cicik E,et al. Descemet-stripping automated endothelial keratoplasty in eyes with toxic anterior segment syndrome after cataract surgery. J Cataract Refract Surg 2010;36:965-9
- Juan G. Gaviria, MD, Luis Escaf, MD, Juanita Londoño, MD, and Luz M. Melo, MD,. Managing Endothelial Risk - A stepwise approach to avoiding damage to the endothelium during cataract surgery.Review of Ophthalmology. Published9 June 2017
- Soler N, Romero-Aroca P, Gris O, et al. Corneal endothelial changes in patients with chronic obstructive pulmonary disease and corneal vulnerability to cataract surgery. J Cataract Refract Surg 2015;41:313-319
- Nida Wongchaisuwat, Ankana Metheetrairat, Pratuangsri Chonpimai, Waree Nujoi, Pinnita Prabhasawat.Comparison of central corneal thickness measurements in corneal edema using ultrasound pachymetry, Visante anterior-segment optical coherence tomography, Cirrus optical coherence tomography, and Pentacam Scheimpflug camera tomography. Clinical Ophthalmology 2018 Volume 12:1865-1873.