I. Introduction
The cornea is lined by non-keratinized, stratified squamous epithelium. At the limbus, there is a gradual transition of these cells to non-keratinized, stratified columnar epithelial cells, along with mucin-secreting goblet cells, forming the conjunctival epithelium. The corneoscleral limbus lies between these two epithelia in a healthy eye. The limbus contains palisades of Vogt, the basal layer of which function as a repository for the corneal epithelial stem cells.(1, 2)
These limbal epithelial stem cells (LESC’s) divide whenever the corneal epithelium is required to be replenished. As the stem cells divide, one of the daughter cells retains the primitive nature of the parent cell and functions to maintain the stem cell pool. The other daughter stem cell differentiates to acquire the physiological nature of a specific tissue, like the corneal epithelium. These cells are known as ‘transient amplifying cells’, which then divide and migrate centripetally, circumferentially, and vertically from the basal layer to replace the surface epithelial cells that are desquamated. This theory for corneal homeostasis was given by Thoft and is known as the XYZ hypothesis of corneal epithelial maintenance.(2)
The LESC’s act as a mechanical barrier preventing the conjunctival epithelium from growing over the corneal surface. Any insult to this barrier disrupts it, allowing the conjunctival epithelium to grow over the cornea resulting in limbal stem cell deficiency (LSCD).(3, 4)
II. Definition and pathogenesis of LSCD
LSCD is an ocular surface disease caused secondary to a decrease in the number of, or abnormal physiological functions of the LESC's. This interferes with the function of the LESC’s to regenerate and maintain a healthy corneal surface.(3) LSCD is characterized by progressive conjunctivalization (the corneal epithelium is replaced by the conjunctival epithelium) along with signs of poor corneal epithelial healing such as chronic inflammation, persistent epithelial defects, corneal neovascularization, and corneal scarring.(5)
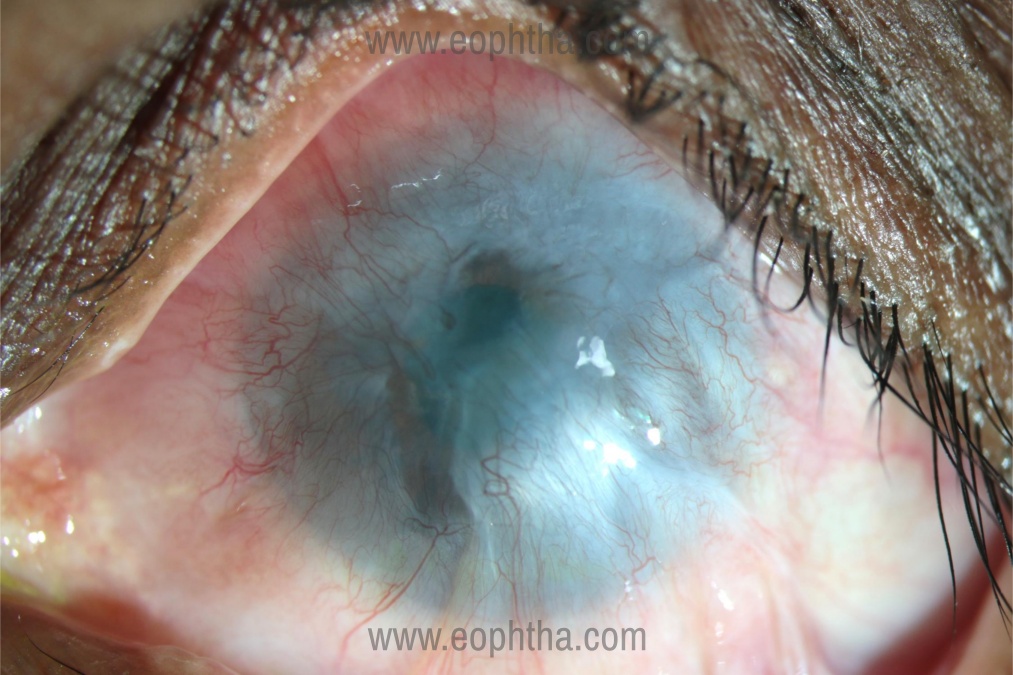
The complete loss of LESC’s and their function, results in Total LSCD, where the corneal epithelium is completely replaced by the conjunctival epithelium, thus resulting in corneal blindness. (Figure 1) Partial LSCD may occur in the presence of few functional LESCS’s, characterized by incomplete conjunctivalization of the corneal surface.(3)
Our understanding of the physiology of LESC’s, their role in maintaining the ocular surface, and that LSCD is a manifestation of their dysfunction, has fueled many surgical techniques involving transplantation of these stem cells, over the past few decades. LSCD that was once considered as an irreversible blinding disease can now be treated with various limbal stem cell transplantation (LSCT) techniques, the most recent of which is simple limbal epithelial transplantation (SLET).[3],[4],[5]
Simple limbal epithelial transplant (SLET) is a type of LSCT where one clock hour of the limbal tissue containing the LESC’s is harvested from the healthy donor eye and transferred to the eye with LSCD. This innovative surgical technique was first described by Sangwan et al in 2012.(6) Depending on the source of the donor tissue, LSCT can either be autologous (from the unaffected fellow eye of the same person) or allogeneic (from a cadaveric donor or from a live-related donor).
III. What are the other types of autologous LSCT for unilateral LSCD, other than SLET?
A. Conjunctival-limbal autograft (CLAU) described by Kenyon and Tseng, 1989(7)
Technique: Limbal tissue attached to a conjunctival carrier (of around 3 clock hours) is transplanted from the healthy donor eye to the contralateral recipient eye with LSCD, at 12 and 6’o clock positions. The transplanted tissue is hence located at the physiological position, that is, the limbus.
Indications: In cases of unilateral total LSCD with severe symblepharon which require a complex procedure to address the LSCD with symblepharon simultaneously, with or without a corneal transplant
Limitations: Iatrogenic LSCD in the donor eye.(8)
B. Cultivated limbal epithelial transplantation (CLET) described by Pellegrini et al 1997.(9)
Technique: Limbal tissue (2 × 2 mm) harvested from the contralateral healthy eye is expanded in vivo (in the laboratory), to a multilayer sheet of corneal epithelial cells on an amniotic membrane over a period of 10-14 days, this is then transferred to the recipient cornea with LSCD
Advantages: The amount of limbus harvested is small, thus no risk of iatrogenic LSCD, hence the procedure is repeatable
Limitations:
1. Considering this technique requires a laboratory for in vivo cultivation of the epithelial cells, difficult in a resource-limited setting.
2. The cultivated corneal epithelial cells may get contaminated while transporting them, strict protocols are required to be followed.
3. The cultivated cells are xenogeneic material; thus, potential safety issues arise.
4. Difficult logistically for both the patient and the surgeon as a gap of 10-14 days exists between the harvesting of limbal tissue and the transplant of the multilayer sheet of epithelial cells.
5. Expansion of the epithelial cells over the amniotic membrane might fail.
IV. Clinical presentation of LSCD (3, 4, 10, 11)
A) Symptoms:
- Conjunctival hyperemia (chronic)
- Decreased vision
- Photophobia
- Tearing
- Blepharospasm
- Foreign body sensation
- Recurrent episodes of pain (due to repeated epithelial breakdown)
B) Signs:
|
Mild stage |
Moderate stage |
Severe stage |
|
Epithelial opacity * An irregular reflex of the corneal surface (thickened and irregular corneal epithelium) |
Vortex keratopathy* The metaplastic conjunctival epithelium grows over the corneal surface in a whorled pattern This pattern of growth from the limbus to the central corneal is also called ‘whorl-like epitheliopathy’. |
Recurrent/Persistent epithelial defects A breach in the barrier function of the limbus affects the corneal epithelial healing Severe cases may develop secondary infection of these chronic non-healing epithelial defects, resulting in microbial keratitis, stromal melt, and corneal perforation. |
|
Epithelial staining – stippled pattern and late fluorescein staining* The fluorescein pools over areas of corneal epithelium that have been replaced by the conjunctival epithelium. The conjunctival epithelial cells are flatter and lack inter-cellular tight junction, allowing penetration of the fluorescein, which tends to be retained for a longer time. |
Superficial vascularization As the conjunctival epithelium does not secrete anti-angiogenic factors, peripheral superficial vascularization and pannus may be present in the moderate stage. |
Stromal neovascularization Chronic surface inflammation may cause deep stromal vascularization Not specific to LSCD, may occur in other conditions like microbial keratitis. |
|
Loss of palisades of Vogt (not a specific sign, as these changes may be age-related) These palisades are prominent along the superior and inferior limbus Their structure may get altered in the early stages of LSCD |
Stromal scarring and opacity |
*These features are better appreciated under cobalt blue filter on slit-lamp examination, after staining the corneal surface with fluorescein dye.
V. Indications of SLET:
A) Autologous SLET for unilateral LSCD (source of the donor tissue is the unaffected fellow eye of the same person)
- Ocular chemical injury (12-14)
- Iatrogenic LSCD (post topical Mitomycin C use, post excision of OSSN, post multiple surgical interventions)(15-18)
- Primary/recurrent pterygium (19-21)
- Previously failed LSCT (22)
B) Allogeneic SLET for bilateral LSCD (source of the donor tissue is the cadaveric or live-related limbus) (23)
Inflammatory causes such as
- Stevens-Johnson Syndrome (SJS),
- Mucous membrane pemphigoid (MMP)
- Chronic ocular allergy – Vernal Keratoconjunctivitis (VKC)
- Graft versus host disease
V. Pre-operative Considerations (23)
A. Evaluation of the donor cornea
1. Most common site for harvesting the donor tissue is the superior limbus as the limbal palisades of Vogt are maximum in this area.
2. The donor limbus should be healthy. Ensure there is no previous history of trauma (mechanical/chemical), inflammation, or surgery performed at the site from where the tissue is to be harvested.
3. If cadaveric limbal tissue is used (cadaveric allogeneic SLET) (24)
- It should be utilized within 48 hours of harvesting the tissue
- Have clinically visible limbal palisades of Vogt
- No associated epithelial sloughing
- Donor age less than 60 years
These criteria ensure that the proliferative potential of the cadaveric tissue is as good as tissue from a live-related donor.(24)
B. Evaluation of the Recipient Cornea (23)
1.Ensure that the recipient corneal surface is wet
- Repeated Schirmer’s score value of more than 10 mm after instilling topical anesthetic drops
- There should be no evidence of conjunctival/corneal surface keratinization
2. No existing adnexal pathologies (lagophthalmos, entropion/ectropion, trichiasis/distichiasis)
- If adnexal pathologies are present, they need to be corrected first before performing SLET
3. Look for location and extent of symblepharon
- A symblepharon is a pathological condition where the palpebral conjunctiva partially or completely adheres to the bulbar conjunctiva
- Cases that need symblepharon release with conjunctival autograft along with SLET are cases where:
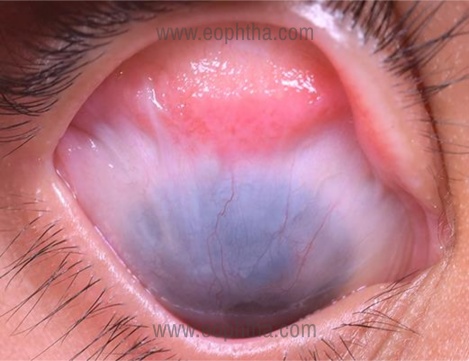
- The symblepharon extends till the limbus (Figure 2)- this may interfere with placement of eye speculum intra-operatively and placement of bandage contact lens (BCL) or scleral lenses in the post-operative period
- The symblepharon limits the extraocular movement of the eye causing diplopia.
4. Presence of underlying stromal scarring and corneal thickness
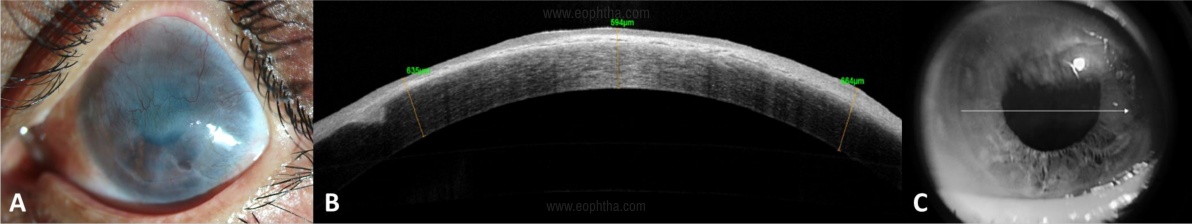
- An ideal case would be an eye with clear to translucent underlying stroma of the cornea (Figure 3) – this can be delineated on an anterior segment optical coherence tomography (AS-OCT)
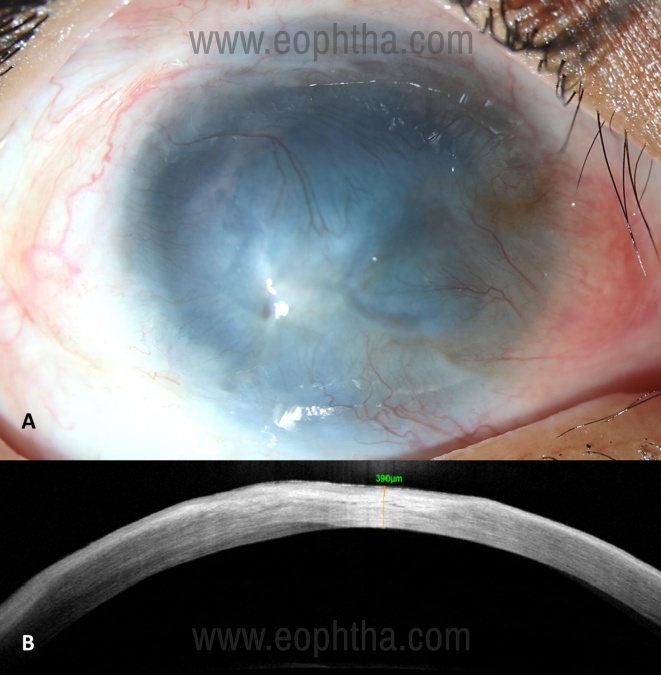
- In cases that have dense stromal opacification (Figure 4A), an AS-OCT can provide additional information like:
a. Extent of underlying stromal scarring (Figure 4B) - These cases may need additional surgery like a full-thickness penetrating keratoplasty or anterior lamellar keratoplasty in addition to SLET to address the scarring, either as a single surgery or a staged procedure later.
b. Extent of corneal thickness (linear scan) - Areas that are thin need careful dissection of pannus over them, as there is a risk of perforation in these areas
|
Pre-operative assessment of recipient cornea |
|
|
S |
Surface wetness |
|
L |
Lid and adnexal pathologies |
|
E |
Extent of Symblepharon |
|
T |
Thickness of the pannus and opacification of the underlying cornea |
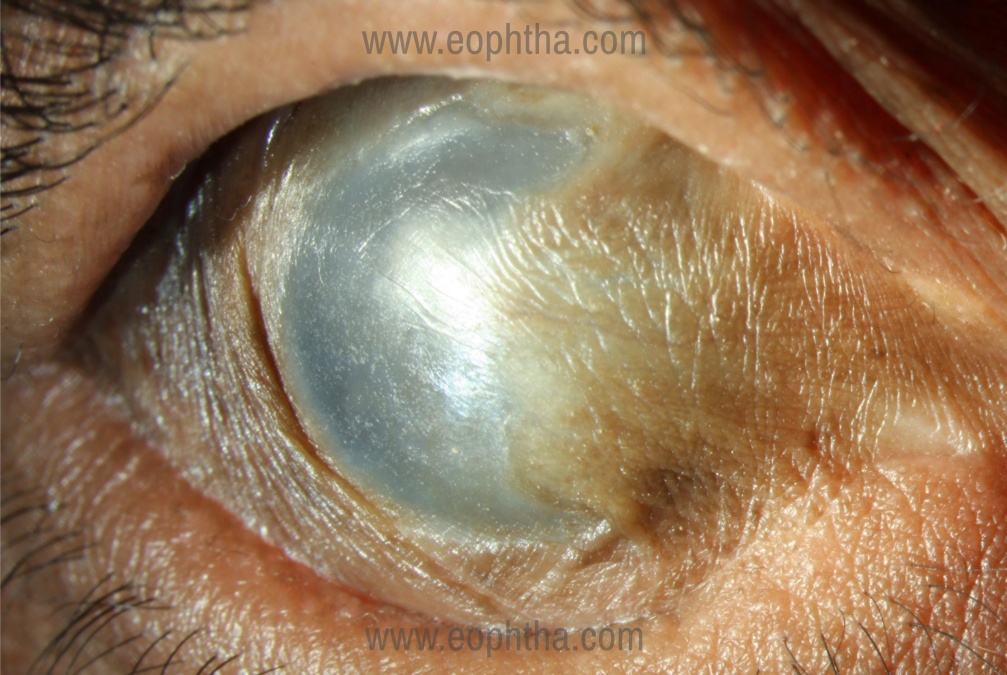
Absolute contraindications for SLET (23)
- Dry ocular surface (defined as repeated Schirmer's I score with anaesthesia of less than 10 mm or presence of corneal or bulbar conjunctival keratinization) (Figure 5)
- blind eye with no visual potential
- disorganized anterior segment (adherent leukoma, anterior staphyloma, or extensive peripheral anterior synechiae)
- presence of uncorrected adnexal pathologies like lagophthalmos, ectropion, entropion, trichiasis, and dacryocystitis
C. Pre-operative counseling:
- Since harvesting the limbal graft involves operating on the unaffected eye, it is important to explain to the patient and relatives/guardians clearly about the procedure and address all their queries.
- The goal of surgery should be explained:
- The cosmesis of the affected eye is likely to improve after surgery, however SLET being an epithelial regenerative procedure has limited impact on corneal stromal opacification. The patient should be counselled that additional surgeries may be required later to improve vision.
- In long-standing cases, especially when initial insult has occurred in childhood, certain coexisting pathologies like ptosis, squint, or poor vision due to amblyopia may not improve after surgery. For better cosmesis, conditions like ptosis and squint may need additional surgical procedures.
- Consent should be taken for surgical intervention in both the eyes, after explaining the procedure and associated risks in a language the patient/relative/guardian best understands.
VI. Surgical technique(23)
The surgical steps are sequentially depicted in the following videos:
A. Anesthesia
Surgery in children should always be performed under general anesthesia. For adults, harvesting the limbal graft can be done under topical/subtenon’s/peribulbar anesthesia. Surgery on the affected eye should be performed under peribulbar anesthesia.
Pre-operative vasoconstrictors
- Brimonidine tartrate (0.15%) and phenylephrine (5%) eye drops can be used 2-3 times alternatively for 5-10 minutes before starting the surgery
- This reduces the intra-operative bleeding in the donor and recipient eye at the time of surgical dissection.
B. Harvesting the limbal graft from the donor's eye
Marking the biopsy site is not mandatory, but it is advisable to do so. In live donors (autologous/allogeneic), the marking is done using a skin marking pen, slightly beyond the limbus on the conjunctival surface. The limbus should not be marked directly as the alcohol in the marking ink may damage the LESC’s.
Do not use limbal pigmentation as a guide for taking the limbal biopsy
- The amount of limbal pigmentation is not uniform in all patients
- Sometimes the pigmentation may extend beyond the limbus
- When the conjunctival flap is lifted, the limbal pigmentation is not visible to the operating surgeon
The limbal graft of around one clock hour/3.5-4mm limbal tissue is sufficient. Conjunctival bleb is created using fluid, slightly behind the marked area for biopsy. A limbus-based conjunctival flap is lifted till the insertion of Tenon’s capsule at the limbus. This area is marked by blood vessels and defines the posterior limit of the limbus. Dissection is carried out using a 15 number blade on a Bard-Parker handle.
The blade is kept parallel to the dissecting plane to avoid superficial dissection, which can cause a buttonhole in the graft or a deep dissection which may include the corneal stroma.
The flap is reposited, and the conjunctival part is separated from the limbus by excising the grey demarcation line (anterior to this line is the limbus and posterior to the line is the white conjunctival tissue)
The conjunctival part is preserved or discarded depending upon whether the conjunctival graft needs to be performed with SLET. For example, in cases with extensive symblepharon, the limbal tissue is excised separately using Vannas or Wescott scissors and preserved in a balanced salt solution (BSS)
In cadaveric donors, it is not possible to make a conjunctival flap, as the corneoscleral rim may not have any conjunctival tissue. ‘Snip biopsy’ technique has been described to harvest the limbal tissue from cadaveric donors. In this technique, the limbal tissue is pinched using a Lim’s forceps and one clock hour is cut using a Vannas or Wescott’s scissors. This tissue is bulky as it contains more stromal tissue as compared to a graft harvested from a live donor and requires stromal trimming before transplantation.
In allogeneic SLET, it is important not to take a lengthier biopsy or put more pieces on the cornea because the risk of immunological rejection increases with the increase in the amount of transplanted allogeneic tissue.
C. Dissection in the recipient eye
Any symblepharon interfering with insertion of eye speculum should be excised first. Conjunctival peritomy is performed 360 degrees around the presumed limbus using Vannas scissor. This provides a good amount of tissue to hold on to, while dissecting the pannus over the cornea. Careful dissection of the pannus is performed over the corneal surface using an appropriate techniquelike peeling, and blunt and sharp dissection. Dissection over the central cornea is performed last, as the cornea is thinnest in this area and there is a higher risk of inadvertent corneal perforation. Excessive manipulation of the corneal stroma to achieve a clear corneal surface is best avoided.
Removing a 3-4 mm frill of tenon’s capsule all around the limbus offers the following advantages:
- The resected conjunctiva adheres better to the underlying sclera over the hAM
- It is easier to tuck the hAM under the conjunctiva thus preventing bunching up and further dislocation of the hAM
The hAM is placed epithelium side up and secured over the recipient corneal surface with fibrin glue (TISSEEL kit from Baxter AG, Austria). The hAM is ironed out with a blunt spatula and tucked all around the recessed conjunctival margins. Folds in the hAM over the cornea should be avoided.
The limbal tissue is then removed from the BSS and divided into 6-10 pieces using a Vannas scissor. These grafts are then placed in a concentric fashion in the midperiphery of the cornea, outside the pupillary area, over the hAM.
Always keep the instrument wet while arranging the transplants in the mid-peripheral cornea.
Ensure that the grafts are placed in the correct orientation with the epithelial side up.
- The correct orientation can be identified from the pigmentation and smooth surface of the epithelial side and the thick white fibrous surface of the stromal side
- The orientation is important so that the growth of the epithelial cells from the grafts is not inhibited by the stromal tissue
- Reverse orientation may delay epithelization of the cornea
A drop of fibrin glue is placed over each piece to ensure that they adhere to the underlying hAM. Allow sufficient time for the glue to polymerize (seen as white lines on the surface) over the transplants, before placing the BCL on the corneal surface.
- The thick component of the fibrin glue (fibrinogen) is first placed over the transplants and spread evenly with a blunt spatula
- The thin component (thrombin) is then placed over the transplants.
The speculum should be removed carefully by simultaneously cutting the fibrin glue strands attached to the speculum with Vannas scissor. If adequate care is not taken while removing the speculum, the sheet of fibrin glue holding the transplants may be pulled and dislodged, and hAM may also be dislodged simultaneously.
In children, temporary lateral suture tarsorrhaphy is recommended postoperatively, to avoid accidental loss of BCL/hAM with transplants as children often tend to rub their eyes.
VII. Post-operative regimen(23)
|
Autologous transplant |
|
|
Topical steroids (Prednisolone acetate 1%) 6 times /day, then tapered weekly and stopped after six weeks, in both the recipient and the donor eyes |
The BCL may be removed at 1 week. It is advisable to check the epithelium healing using fluorescein stain, and if the epithelial defect has not healed, BCL should be replaced Temporary suture tarsorrhaphy may be released at 1-2 weeks post operatively |
|
Topical antibiotics (Moxifloxacin 0.5%) eye drops 4 times/day in both the recipient and the donor eyes till the corneal epithelium has completely healed |
|
|
Topical cyclosporin 0.05% eyedrops may be used for several months in the presence of superficial corneal haze |
|
|
Oral steroids (prednisolone,1 mg/kg ) and antibiotic-steroid eye ointment may be used to reduce periocular tissue edema . |
|
Allogeneic transplant |
|
|
Topical steroids (Prednisolone acetate 1%) 6 times/day, then tapered weekly and continued at once or twice daily dose (never to be stopped completely) |
Since the transplanted allogeneic limbal tissue is placed on the mid-peripheral avascular corneal stroma, the risk of immunological rejection is theoretically less compared to conjunctival/keratolimbal allografts. Irrespective of the regimen, haematological investigations and hepatic and renal parameters need to be reassessed every 4–6 weeks. Patients can be prescribed scleral contact lenses for optimal visual recovery after 6–8 weeks of the procedure |
|
Topical antibiotics (Moxifloxacin 0.5%) eye drops 4times/day till epithelial defect heals |
|
|
Intravenous methylprednisolone 500mg bolus on the day of transplant. Repeat at one week, 6 weeks, every 6 weeks thereafter until 6 months, every 2 months thereafter until 1-year; every 3 months thereafter until 2 years, and every 6 months thereafter. (in patients who can follow-up, and in patients who may not be compliant to oral medications) |
|
|
Alternate oral regime: Oral cyclosporine is started in a dosage of 5 to 7 mg/kg, 48-hours before surgery, along with methylprednisolone 1g intravenously, for the first 3 consecutive postoperative days. During the postoperative period, cyclosporine is tapered to the maintenance dosage of 1.5 to 2 mg/kg over 4 to 8 weeks, with diltiazem hydrochloride, 90 mg, added as an adjunct to cyclosporine to reduce the cost and increase the serum levels of cyclosporine. |
|
|
Oral prednisolone, 1 mg/kg tapered on a weekly basis to the maintenance dosage of 5 mg/day. |
Allograft Rejection
Patients may present with a sudden drop of vision, epithelial haze, positive fluorescein staining, and superficial vascularization approaching the limbal transplants.
Management: Additional dose of 500mg IVMP and stepping up topical steroids
VIII. Mechanism of action of SLET
The epithelial cells grow from all directions, usually from each transplant, until they form a uniform sheet of confluent epithelial cells covering the surface of the cornea. This has been demonstrated by Mittal et al in study to serially demonstrate this process using fluorescein staining.(25) Other conclusions drawn from the study were:
- It takes around 14 days for complete epithelization of the ocular surface.
- All the transplants usually do not assume activity at the same time or at the same rate.
- Inactive transplants were overrun by epithelial cells growing from the adjoining transplant.
- The hAM provides a substrate for these cells to proliferate, migrate and adhere better to the ocular surface.
In another study, sequential corneal imaging (High-resolution anterior segment optical coherence tomography, HR-ASOCT) was performed post SLET, for total LSCD after ocular chemical burn.(26) The observations made during the 3 year follow up were:
- The corneal epithelization started on the 3rd day post-operatively and was completed on the 14th day.
- The thickness of the corneal transplants gradually reduced as they integrated in the corneal epithelium and could not be clinically appreciated on slit-lamp examination after 2 years.
- The hAM was seen as a hyper-reflective layer below the corneal epithelium that could be recognized for the entire follow-up duration of 3 years, however, the thickness gradually reduced over the period of time.
The epithelial cells grown from each transplant not only have cells of corneal phenotype (CK3+, CK12+, CK19-, MUC5AC-) but also there is focal retention of stem cells (ABCG2+, ΔP63α+) in the basal epithelial layer of this newly regenerated epithelium. This was confirmed by the histopathology, immunohistochemistry, and cytokeratin studies of the excised corneal buttons undergoing PK after SLET.(12)
IX. Outcomes of SLET
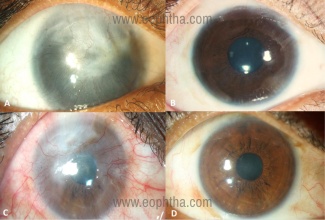
- Promising results post SLET have been published in large studies with a success rate of 70-84%, over a mean follow-up period of 1-3 years.(12-14) In all these three studies, in a majority of cases SLET was performed for the primary indication of ocular chemical burns. Successful outcome was labelled in eyes that maintained a well epithelized and avascular corneal surface without progressive conjunctivalization.(Figure 6)
- A few risk factors contributing to the failure of SLET were the presence of symblepharon and cases where simultaneous keratoplasty was performed. (12-14) Better outcomes were seen where keratoplasty was performed (DALK or PK) as a sequential surgery in cases where the ocular surface inflammation was well-controlled and stable after autologous SLET, as compared to combined surgery (SLET with PK).(27, 28)
- Outcomes of SLET in children has not been extensively published. However, successful results were seen in all 4 eyes that underwent SLET six months after AMT was done for acute ocular burns, either as a primary procedure or repeat SLET with CLAG for a failed SLET.(29)
- Exceptional outcomes were also seen where simultaneous SLET was performed with OSSN excision,(15) and recurrent pterygium excision. (19)
X. Complications (12, 23)
|
In the donor eye |
In the recipient eye |
|
Focal non-progressive LSCD |
Conjunctivalization |
|
Pyogenic granuloma |
Symblepharon formation |
|
Subconjunctival hemorrhage |
Hemorrhage under hAM |
|
Detached hAM |
|
|
Loss of corneal transplants |
|
|
Microbial keratitis |
XI. Limitations and advantages of SLET (6, 23)
- Advantages:
- Single staged procedure
- Requires a small amount of healthy limbus to regenerate the entire corneal surface in the affected eye – hence it is a repeatable procedure and minimizes chances of iatrogenic LSCD in the donor eye
- Can be performed in a resource-limited setting - Does not require a lab for the cultivation of the epithelial cells, hence is also cost-effective
- Limitations:
- Cases with bilateral LSCD, need allogeneic SLET which needs long-term systemic immunosuppression
- Cases with bilateral total LSCD with extensive symblepharon, CAG cannot be performed. Hence in these cases an allogeneic SLET needs to be combined with oral mucous membrane graft taken from the labial mucosa, or a CLAU is an alternative procedure that may be performed, although CLAU may not be able to address eyes with multiple symblephara
XII. Conclusions
SLET has shown promising results in eyes with unilateral LSCD. This surgery has been commonly performed in eyes with LSCD secondary to ocular surface burns. This procedure helps in epithelial regeneration and maintaining a healthy stable ocular surface. The success rate of this technique is high, especially when the case selection is appropriate. This technique does not have a steep learning curve and has been adopted by corneal surgeons worldwide. This surgical procedure can also be performed easily in a resource-limited setting without the need for an expensive laboratory set-up.
References
1. Thoft RA, Wiley LA, Sundarraj N. The multipotential cells of the limbus. Eye (London, England). 1989;3 ( Pt 2):109-13. 2. Thoft RA, Friend J. The X, Y, Z hypothesis of corneal epithelial maintenance. Investigative ophthalmology & visual science. 1983;24(10):1442-3. 3. Deng SX, Borderie V, Chan CC, Dana R, Figueiredo FC, Gomes JAP, et al. Global Consensus on Definition, Classification, Diagnosis, and Staging of Limbal Stem Cell Deficiency. Cornea. 2019;38(3):364-75. 4.Le Q, Xu J, Deng SX. The diagnosis of limbal stem cell deficiency. The ocular surface. 2018;16(1):58-69. 5. Shukla S, Shanbhag SS, Tavakkoli F, Varma S, Singh V, Basu S. Limbal Epithelial and Mesenchymal Stem Cell Therapy for Corneal Regeneration. Current eye research. 2020;45(3):265-77. 6. Sangwan VS, Basu S, MacNeil S, Balasubramanian D. Simple limbal epithelial transplantation (SLET): a novel surgical technique for the treatment of unilateral limbal stem cell deficiency. The British journal of ophthalmology. 2012;96(7):931-4. 7. Kenyon KR, Tseng SC. Limbal autograft transplantation for ocular surface disorders. Ophthalmology. 1989;96(5):709-22; discussion 22-3. 8. Basti S, Mathur U. Unusual intermediate-term outcome in three cases of limbal autograft transplantation. Ophthalmology. 1999;106(5):958-63. 9. Pellegrini G, Traverso CE, Franzi AT, Zingirian M, Cancedda R, De Luca M. Long-term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet (London, England). 1997;349(9057):990-3. 10. Le Q, Chauhan T, Deng SX. Diagnostic criteria for limbal stem cell deficiency before surgical intervention-A systematic literature review and analysis. Survey of ophthalmology. 2020;65(1):32-40. 11. Jawaheer L, Anijeet D, Ramaesh K. Diagnostic criteria for limbal stem cell deficiency-a systematic literature review. Survey of ophthalmology. 2017;62(4):522-32. 12. Basu S, Sureka SP, Shanbhag SS, Kethiri AR, Singh V, Sangwan VS. Simple Limbal Epithelial Transplantation: Long-Term Clinical Outcomes in 125 Cases of Unilateral Chronic Ocular Surface Burns. Ophthalmology. 2016;123(5):1000-10. 13. Vazirani J, Ali MH, Sharma N, Gupta N, Mittal V, Atallah M, et al. Autologous simple limbal epithelial transplantation for unilateral limbal stem cell deficiency: multicentre results. The British journal of ophthalmology. 2016;100(10):1416-20. 14. Gupta N, Joshi J, Farooqui JH, Mathur U. Results of simple limbal epithelial transplantation in unilateral ocular surface burn. Indian journal of ophthalmology. 2018;66(1):45-52. 15. Kaliki S, Mohammad FA, Tahiliani P, Sangwan VS. Concomitant Simple Limbal Epithelial Transplantation After Surgical Excision of Ocular Surface Squamous Neoplasia. American journal of ophthalmology. 2017;174:68-75. 16. Narang P, Mittal V, Menon V, Bhaduri A, Chaudhuri BR, Honavar SG. Primary limbal stem cell transplantation in the surgical management of extensive ocular surface squamous neoplasia involving the limbus. Indian journal of ophthalmology. 2018;66(11):1569-73. 17. Mittal V, Narang P, Menon V, Mittal R, Honavar S. Primary Simple Limbal Epithelial Transplantation Along With Excisional Biopsy in the Management of Extensive Ocular Surface Squamous Neoplasia. Cornea. 2016;35(12):1650-2. 18.Amescua G, Atallah M, Nikpoor N, Galor A, Perez VL. Modified simple limbal epithelial transplantation using cryopreserved amniotic membrane for unilateral limbal stem cell deficiency. American journal of ophthalmology. 2014;158(3):469-75.e2. 19. Mednick Z, Boutin T, Einan-Lifshitz A, Sorkin N, Slomovic A. Simple limbal epithelial transplantation for recurrent pterygium: A case series. American journal of ophthalmology case reports. 2018;12:5-8. 20. Trinh T, Mimouni M, Mednick Z, Einan-Lifshitz A, Cohen E, Santaella G, et al. Outcomes of Ipsilateral Simple Limbal Epithelial Transplantation, Tenonectomy, Mitomycin and Amniotic Membrane Transplantation for Treatment of Recurrent Pterygium. Cornea. 2021;40(1):43-7. 21. Hernández-Bogantes E, Amescua G, Navas A, Garfias Y, Ramirez-Miranda A, Lichtinger A, et al. Minor ipsilateral simple limbal epithelial transplantation (mini-SLET) for pterygium treatment. The British journal of ophthalmology. 2015;99(12):1598-600. 22. Basu S, Mohan S, Bhalekar S, Singh V, Sangwan V. Simple limbal epithelial transplantation (SLET) in failed cultivated limbal epithelial transplantation (CLET) for unilateral chronic ocular burns. The British journal of ophthalmology. 2018;102(12):1640-5. 23. Shanbhag SS, Patel CN, Goyal R, Donthineni PR, Singh V, Basu S. Simple limbal epithelial transplantation (SLET): Review of indications, surgical technique, mechanism, outcomes, limitations, and impact. Indian journal of ophthalmology. 2019;67(8):1265-77. 24. Notara M, Shortt AJ, O'Callaghan AR, Daniels JT. The impact of age on the physical and cellular properties of the human limbal stem cell niche. Age (Dordrecht, Netherlands). 2013;35(2):289-300. 25.Mittal V, Jain R, Mittal R. Ocular Surface Epithelialization Pattern After Simple Limbal Epithelial Transplantation: An In Vivo Observational Study. Cornea. 2015;34(10):1227-32. 26.Kate A, Shanbhag SS, Goyal R, Basu S. Serial anterior segment optical coherence tomography post autologous simple limbal epithelial transplantation. BMJ case reports. 2020;13(12). 27.Singh D, Vanathi M, Gupta C, Gupta N, Tandon R. Outcomes of deep anterior lamellar keratoplasty following autologous simple limbal epithelial transplant in pediatric unilateral severe chemical injury. Indian journal of ophthalmology. 2017;65(3):217-22. 28. Gupta N, Farooqui JH, Patel N, Mathur U. Early Results of Penetrating Keratoplasty in Patients With Unilateral Chemical Injury After Simple Limbal Epithelial Transplantation. Cornea. 2018;37(10):1249-54. 29. Mittal V, Jain R, Mittal R, Vashist U, Narang P. Successful management of severe unilateral chemical burns in children using simple limbal epithelial transplantation (SLET). The British journal of ophthalmology. 2016;100(8):1102-8.


