Angioid streaks are visible irregular crack-like dehiscences in Bruch’s membrane that are associated with atrophic degeneration of the overlying retinal pigmented epithelium. Despite 50 % of cases being idiopathic, this condition grabs attention as it requires good history taking and workup to determine the cause.
Historical background
The first description of angioid streaks was provided in 1889 by Dr. Robert W. Doyne, an English ophthalmologist. He noticed irregular, jagged deeply pigmented lines especially around the discs of both eyes in a patient who sustained trauma in both eyes and attributed it to be due to the rupture of pigment layer of retina. [1] Because of their resemblance to blood vessels, Herman J. Knapp, a German-born American ophthalmologist coined and introduced the term "angioid streaks" in 1892. [2] They are also known as Knapp’s striae. Clinical examination with histopathological findings by Bock in 1938 in two patients with pseudoxanthoma elasticum confirmed that the underlying abnormality was not vascular in nature but rather a structural alteration in Bruch’s membrane. [3] Subsequently similar histopathological results were found in patients suffering from Paget’s disease and several systemic diseases as well. [4]
Etiopathogenesis
The exact pathogenesis and pathophysiology of angioid streaks is not known. A dual mechanism suggests a primary abnormality of fibers of the Bruch membrane, and an increase in availability of metal salts or a tendency for their deposition, resulting in a secondary brittleness of the membranes. [5] This abnormal structural composition of the Bruch’s membrane predisposes it to localized areas of rupture. The lines of force within the eye resulting from the pull of intrinsic and extrinsic ocular muscles on the relatively fixed site of the optic nerve account for the configuration of the breaks. This process may occur spontaneously or could be secondary to blunt trauma, even very minor. Hence scleral depression should generally be withheld as this could theoretically contribute to formation of additional breaks in Bruch's membrane.
Systemic associations ( Table1 )
Up to 50 % of angioid streak are idiopathic. They can be associated with numerous systemic diseases, the most common being pseudoxanthoma elasticum. Other systemic associations are enlisted in table 1. The presence of angioid streaks should be considered indicative of an underlying systemic illness, even if the specific disorder cannot be identified or defined accurately at the time that the angioid changes are first noted. The patient must undergo a systemic evaluation and remain under continuing surveillance.
|
Pseudoxanthoma elasticum (PXE) PXE is a connective tissue disorder that affects the elastin fibrils in the dermis, arterial walls, heart, gastrointestinal tract, and Bruch's membrane, resulting in mineralization and deposition of phosphorus. It is the most common systemic disorder associated with angioid streaks. Angioid streaks are seen in 80–87% of all patients who have pseudoxanthoma elasticum. [6] The combination of PXE and ocular involvement is referred to as Grönblad-Strandberg syndrome.[7] Systemic manifestations include Dermatologic findings-
|
|
Paget's disease Paget’s disease is a chronic, progressive disease that gives rise to enlargement of the skull, kyphoscoliosis, deafness, and deformities of long bones. However, angioid streaks occur in fewer than 2% of these patients. |
|
Ehlers-Danlos syndrome It is a rare collagen disorder resulting from a deficiency of hydroxylysine. Ocular findings include epicanthal folds, keratoconus, high myopia, retinal detachment, blue sclera, ectopia lentis and angioid streaks. Systemic associations include the following
|
|
Hemoglobinopathies Hemoglobinopathies that are occasionally associated with angioid streaks include homozygous sickle cell disease (Hb-SS), sickle cell trait (Hb-AS), sickle cell thalassemia (Hbthal), sickle cell hemoglobin (Hb-SC), hemoglobin H (Hb-H), homozygous B-thalassemia major, intermedia, and minor and hereditary spherocytosis. |
Table 1. Major systemic associations of Angioid streaks
- Pseudoxanthoma elasticum
- Paget’s disease
- Ehler-Danlos syndrome
- Sickle cell hemoglobinopathies
- Hemochromatosis
- Acromegaly
- Diabetes mellitus
- Acquired hemolytic anemia
- Hereditary spherocytosis
- Myopia
- Neurofibromatosis
- Sturge- Weber syndrome
- Hyperphosphatemia
Histopathology
Histopathologic studies have shown extensive calcification and thickening of Bruch's membrane and the intervening interstitial space between capillaries. In angioid streaks the elastic lamina that occupies the midsegment of Bruch’s membrane is affected, resulting in the disintegration of the elastic fibers. The streaks present as ruptures in this elastic lamina of Bruch's membrane. In pseudoxanthoma elasticum, the primary lesion is the degeneration of elastic fibers of the connective tissue, while the calcium deposition represents a secondary disorder.[8] In Paget’s disease the bone deformities lead to calcium binding by the elastic fibers. [9] The absence of angioid streaks in anemias other than sickle cell hemoglobinopathy has changed the earlier hypothesis of iron deposition [5,10][ leading to Bruch’s membrane fragility. Later studies proved that angioid streaks in patients with sickle-cell hemoglobinopathies are correlated to calcium deposition at Bruch’s membrane. [11] Other histopathological findings include the break or absence of choriocapillaris beneath angioid streaks and thinning or decoloration of RPE. [12]
Clinical features
The initial diagnosis of an abnormal macula or of optic disc changes may be made during a routine ophthalmoscopic examination of the eye or based on patient complaints of metamorphopsia/ reduced vision. In addition, in certain cases in which the family history is positive for angioid streaks, a screening examination will reveal the presence of the characteristic signs of the disease. Angioid streaks can be difficult to identify unless a careful examination of the posterior pole is performed, especially the peripapillary area. They usually present as linear gray or dark red lines, with irregularly serrated edges lying beneath normal retinal blood vessels. The distribution of streaks can be peripapillary in 27% of cases when it is confined to 2 disc diameters from the optic nerve or more widespread in 73% of cases where the streak radiate for varying distances in the fundus, however not extending past the equator.[13] The color of angioid streaks depends on the background coloration of the fundus and the degree of atrophy of the overlying retinal pigment epithelium (RPE). In lightly colored fundi, angioid streaks are red, reflecting the pigmentation of the underlying choroid. In patients who have darker background pigmentation, angioid streaks are usually a medium to dark brown. [9, 14, 15]
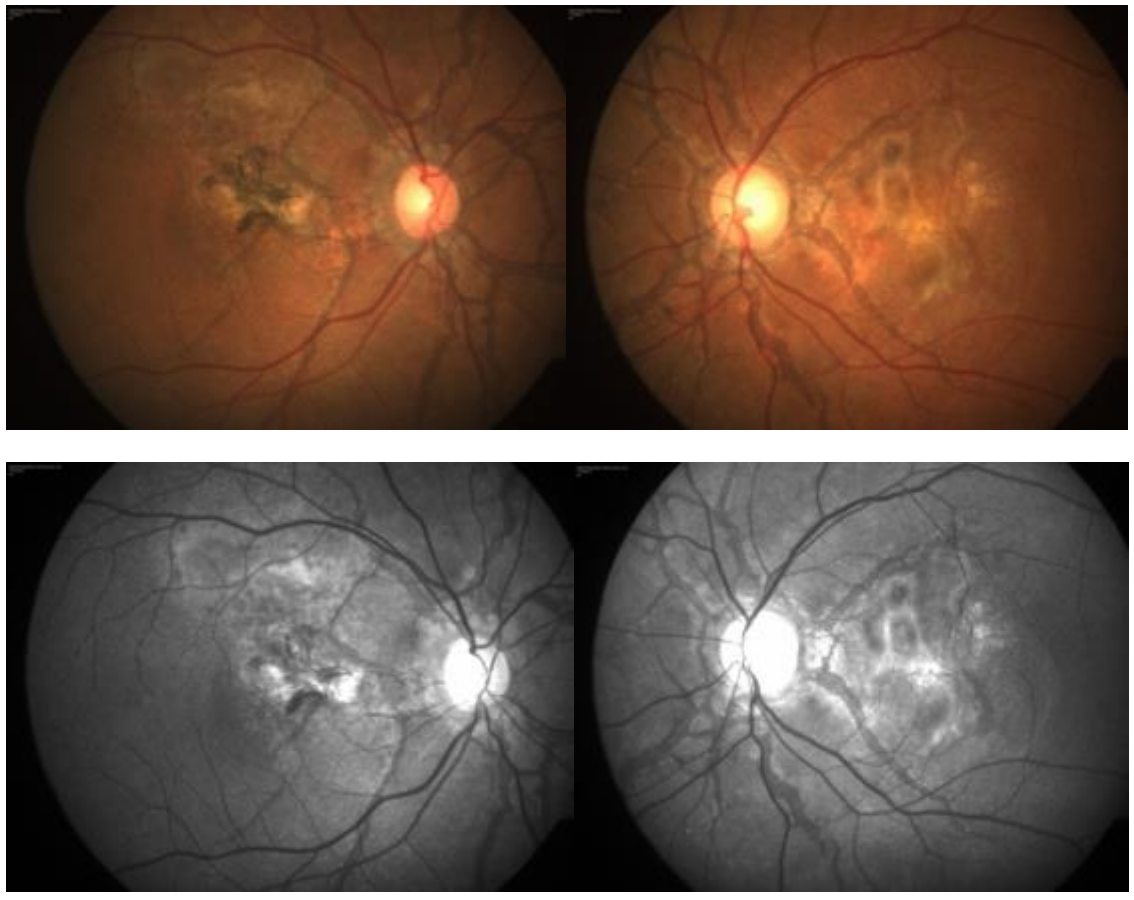
Fig: Fundus photos of a 54 years old male with angioid streaks. Below- Red free images delineating the streaks better.
Other ocular features of angioid streaks include
Peau d'orange or leopard skin spotting It consists of speckled yellowish mottling of the posterior pole mostly apparent in the temporal macula. This typical appearance is caused due to alterations at the level of the Bruch membrane. Although most commonly seen with angioid streaks related to pseudoxanthoma elasticum, the peau d’orange fundus is also seen in patients who have other underlying systemic diseases. [9] Peau d'orange does not affect visual acuity, even when the fovea is involved. However, when readily identifiable in one individual, siblings and parents should be investigated for the presence of peau d'orange even in the absence of angioid streaks.
Optic nerve head drusen (hyaline bodies) They may precede the appearance of angioid streaks. As many as 10% of patients with angioid streaks have clinical evidence of disc drusen. [16] The usual complications of intrapapillary drusen including neovascularization in the peripapillary area, visual field deficits, and even acute visual loss due to the pressure effect on the optic nerve head can occur in this association.
Salmon spots These are punched out focal yellowish chorioretinal atrophic lesions which appear as peripapillary chorioretinal atrophy, focal peripheral chorioretinal scars and/ or reticular pigment dystrophy of the macula. o In older patients, peripheral retinal scars and calcifications can be seen.[17]
Clinical course and complications
Patients with angioid streaks are generally asymptomatic unless the patients develop some secondary complications. The major causes of visual loss in angioid streaks include
Choroidal Neovascularisation [14, 18, 19, 20] The incidence of CNV in patients with angioid streaks varies between 72%–86% in numerous studies. Commonly, CNV does not occur simultaneously in both eyes; there is an interval of roughly 18 months with the subsequent serous and hemorrhagic detachment of the fovea. They are bilateral in 50% of patients. While type 2 NV is the most common presenting anatomical subtype of NV to occur in the setting of angioid streaks, type 1 and mixed (type 1 and type 2) lesions occur in up to 16% of eyes. [21] Patients with angioid streaks who develop CNV are symptomatic and their main symptoms are metamorphopsia and reduction of vision. The risk of developing CNV increases with age. Other risk factors comprise the width, length, and location of the angioid streaks. The wider and longer are the angioid streaks the higher the risk for CNV and especially if the lesions are located in a distance less than one optic disc diameter from the foveola. Interruptions in the mechanical integrity of Bruch’s membrane can result in dysregulation of regional growth factor gradients that together with a potential communication between retinal and choroidal compartments can culminate in CNV. Angioid streaks associated with pseudoxanthoma elasticum have a relatively high probability of developing macular CNV than other systemic disorders.
Choroidal rupture secondary to trivial trauma [22] Owing to the brittle nature of Bruch’s membrane in patients with angioid streaks, even trivial trauma can result in rupture of these membranes. Breaks of Bruch’s membrane are frequently followed by subretinal hemorrhages which can be easily misinterpreted as CNV. A retrospective study on patients with angioid streaks mentioned that 15% of those who suffered a head injury developed significant visual impairment.
Foveal involvement of a streak with damage to the RPE and choriocapillaris The increase in length and width of streaks is considered an expected feature of the disease. When the streak passes through fovea, it causes permanent damage to the RPE and choriocapillaries resulting in a significant drop in vision. RPE tears have also been reported in angioid streaks.[23]
Multimodal imaging in angioid streaks
The diagnosis is mainly clinical. In early cases where the findings are subtle, the imaging modalities have a major role to play.
Fundus autofluorescence (FAF)
Autofluorescence is a noninvasive imaging modality for mapping the naturally or pathologically appearing fluorophores of the ocular fundus. It detects the integrity of RPE cells and reflects its metabolic activity. Angioid streaks on FAF appear as radial fissures with central hypoautofluorescence and with a variable amount of hyperautofluorescent focal spots alongside the angioid streaks at the borders of the cracks. [Fig- ] The presence of optic nerve head drusen can be seen as hyperautofluorescent bodies. RPE changes detected by FAF are more widespread and extensive than seen on fundus photography or fluorescein angiography. [24]
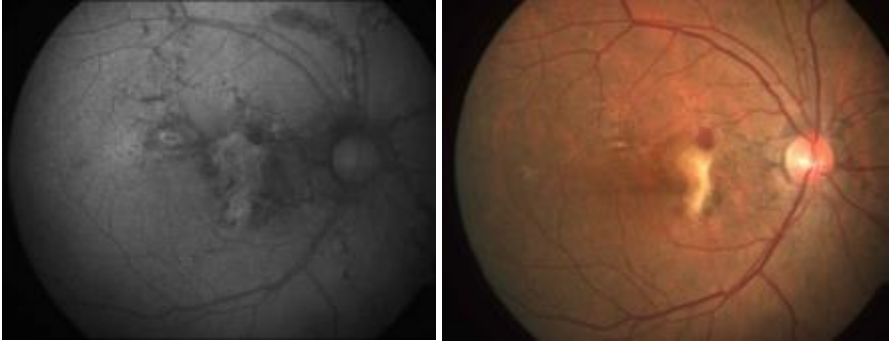
Figure: Fundus autofluorescence imaging shows areas of hypoautofluorescence in the peripapillary area that is greater in size than the corresponding AS seen on color photography.
Confocal Near-Infrared imaging (NIR)
Confocal NIR reflectance imaging at 820 nm is an excellent noninvasive method for imaging abnormalities in the deep retinal and subretinal structures. [25]The absorbance of shorter wavelength monochromatic light by melanin and lipofuscin granules in the RPE, limits the capability of FAF (488 nm) and FA (490 nm) in detecting these deeper pathologies. The absorption of near IR light by melanin is minimal leading to good visibility of structures below the RPE. [26] Additionally, confocality enables visualization of alterations of a given plane with improved contrast and detail. In angioid streaks, breaks in Bruch's membrane appear darker compared with the surrounding fundus reflex on confocal NIR.
Spectral Domain OCT
With the improved resolution of spectral-domain OCT (SD-OCT), the pathology of angioid streaks was localized to the Bruch's membrane-retinal pigment epithelium (RPE) complex. The width of the gap in Bruch's membrane is variable and overlying RPE can remain unaffected, but often appeared altered, focally detached, or absent. [27] In many advanced cases, fibrovascular tissue extended from the choroidal into the subneurosensory space through the gaps in Bruch's membrane. [Fig ] Fundus fluorescein angiography (FA) The diagnosis of angioid streaks is usually made on the basis of fundoscopy, but intravenous fluorescein angiography can help to delineate the presence of the disease when the ophthalmoscopic appearance is subtle. FA detects angioid streaks only when there is a concomitant loss of overlying RPE. Typically, angioid streaks have a ‘window defect’ in fluorescein angiography due to atrophy of RPE adjacent to them. FA also helps to confirm the presence of classic CNV and delineate the margins of the neovascular membrane, which is typically above or adjacent to the angioid streaks. [Fig ]
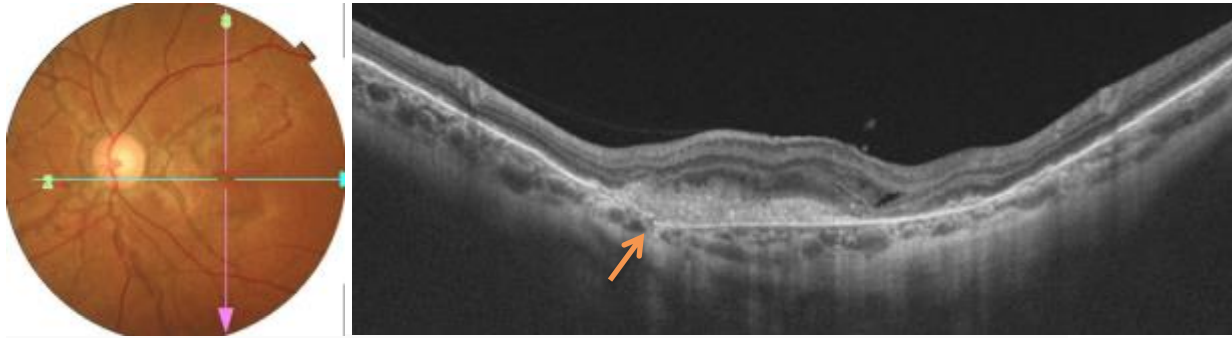
Figure: Optical coherence tomography shows the angioid streaks as disruptions in Bruch’s membrane shown in arrow. Note the fibrovascular tissue extending from the choroid into the subneurosensory space through the gap in Bruch's membrane
Indocyanine green angiography (ICG)
ICG angiography shows hyperfluorescent lines corresponding to the streaks with ‘pinpoints’ over their whole length that are larger and more numerous than those on fluorescein angiography or red-free photography. It is helpful especially in cases where it is difficult to determine the borders of the neovascular membrane in fluorescein angiography due to adjacent hemorrhages or RPE lesions.
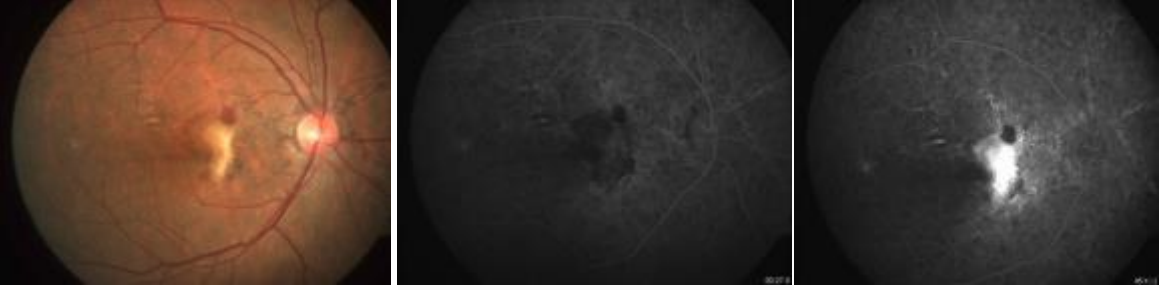
Figure: “ Window defect” in FA due to atrophy of RPE adjacent to angioid streaks. Note the leak at the macula due to active CNV with blocked fluorescence corresponding to the subretinal hemorrhage.
OCT angiography (OCT)
Optical coherence tomography angiography (OCTA) is a relatively newer imaging modality that exploits flow-characteristics to construct non-invasive images of the vascular network.[28] On OCTA the morphology of CNV is characterized by a tangled network of vessels. The configuration of CNV closely follows the trajectory of angioid streaks. [Fig- ]
Figure: En Face OCTA (c) demonstrates two areas of CNV that demonstrate a tangled morphology of vascular networks. Sites of CNV closely correlate to sites of angioid streaks
Management options
The current treatments for patients with angioid streaks can be separated into those treating the systemic disease and those treating the ocular findings.
Systemic evaluation and Management
There is a high association between patients with angioid streaks and other systemic diseases. It is therefore important that patients who have angioid streaks to be further investigated for their systemic condition by a physician. The three most common systemic associations are pseudoxanthoma elasticum, Paget's disease of bone, and sickle cell hemoglobinopathies, and therefore any screening workup should focus on these entities. A complete physical examination by an internist, a skin biopsy, and serologic testing to include serum alkaline phosphatase, serum calcium and phosphate, and hemoglobin electrophoresis is recommended. All individuals with disorders like PXE, should undergo a diligent, complete ophthalmologic examination and annual surveillance for ocular complications of this disorder.
Ocular management
Observation Angioid streaks are usually asymptomatic requiring observation only. Prophylactic photocoagulation of angioid streaks may stimulate CNV formation and is contraindicated.
Secondary prevention Patients with angioid streaks are at higher risk of choroidal rupture and subretinal hemorrhage secondary to mild blunt trauma. [29] They are advised to wear protective goggles and sports glasses when playing sports and during work.
Indications and options for treatment.
Treatment is indicated only whenever a choroidal neovascularization has developed. Untreated CNV associated with angioid streaks generally have poor visual prognosis. [30] Treating CNVM associated with angioid streaks is sometimes challenging. Both occult and classic CNV can occur in the same eye and usually are located very close to the foveal avascular zone. Many of these membranes grow fast once they break through the Bruch membrane. Treatment options include laser photocoagulation, anti-vascular endothelial growth factor (AntiVEGF), Photodynamic therapy (PDT), and surgical removal of CNVM under the fovea.
Laser photocoagulation The reported incidence of CNV in angioid streaks is the quite high, however prophylactic laser is no longer advocated and many researchers strongly believe that this kind of intervention can actually induce CNV. [31] Laser is recommended to treat well-defined juxtafoveal or extrafoveal CNV. The results were not encouraging in macular CNV due to the higher chance of recurrence and the laser-induced scar progression resulting in further deterioration of visual acuity.[22, 32] Photodynamic therapy (PDT) Results of previous studies on the efficacy of photodynamic therapy (PDT) with verteporfin for CNV secondary to age-related macular degeneration (AMD) and pathologic myopia [ 33-34] were encouraging. Hence clinicians tried to use PDT in other pathological entities causing CNV. However when applied to CNV in angioid streaks, it failed to affect the natural course of the disease unless the repetition of treatment was applied in shorter intervals. The long term results of PDT alone are disappointing and include enlargement of the CNV lesion to a disciform scar and associated visual loss, ruptures of already brittle Bruch’s membrane, and damage to collateral choriocapillaries.
Anti-Vascular endothelial growth factor (anti-VEGF) With the introduction of anti- VEGF agents namely bevacizumab and ranibizumab, the treatment of choroidal neovascularization secondary to angioid streak has taken a great positive turn. Treatment with anti- VEGF agents has demonstrated a marked reduction in the rate of visual acuity loss in treated eyes compared with untreated eyes. In contrast to previous therapeutic regimens which resulted in disciform scarring and irreversible loss of overlying RPE, intravitreal anti- VEGFs have shown improvement in visual acuity and anatomic outcomes. [35]
Combined therapy(PDT+ anti- VEGF) Combination therapy like PDT with intravitreal Triamcinolone Acetonide (IVTA) or PDT with intravitreal Bevacizumab has been found to cause regression of CNVM and visual improvement. Additionally the mean number of injections required reduced significantly with combination therapy.[36, 37 ]
Surgical option – Macular translocation In the pre- anti-VEGF era macular translocation was considered as an option for CNV. Macular translocation is a surgical technique wherein the macular neuroretina is moved to lie on top of an area of RPE without previous choroidal neovascularization. The technique involves either a limited translocation or 360 degrees translocation where the entire retina is rotated.[37, 38]
Conclusion
A primary abnormality of fibers of the Bruch membrane combined with an increased brittleness due to metallic salt deposition has been implicated in the pathogenesis of angioid streaks. The three most common systemic associations are pseudoxanthoma elasticum, Paget's disease of bone, and sickle cell hemoglobinopathies. Usually they are bilateral and asymptomatic, however, the majority of cases develop CNV which is the most serious vision-threatening complication in these eyes. Treatments with laser, PDT alone and macular translocation have not resulted in significant visual improvement. Anti- VEGF treatment has shown promising results in the treatment of CNV associated with angioid streaks. Long term studies are warranted in this regard and on the combination therapy.
Clinical pearls on Angioid Streaks
The Most common systemic associations of angioid streaks can be remembered with pnemonic “PEPSI”.
Ocular signs - angioid streaks, peau d’orange, optic disc drusen, and peripheral fundus salmon spots.
Causes of vision impairment in angioid streaks- CNV, choroidal rupture due to trauma, and extension of the streaks to involve the fovea.
Eyes with angioid streaks are more susceptible to damage following blunt ocular trauma. Protective goggles are recommended during contact sports.
References
1. Doyne RW. Choroidal and retinal changes. The result of blows on the eyes. Trans Ophthalmol Soc UK. 1889;9:128.
2. Knapp H. On the formation of dark angioid steaks as an unusual metamorphosis of retinal haemorrhage. Arch Ophthalmol. 1892;21:289-92.
3. Bock Z. KIinik und Anatomie der gefassahnlichen Streifen im Augenhintergrund. Z Augenheilkd.1938;95:1–50.
4. Hagedoorn A. Angioid streaks. Arch Ophthalmol. 1939;21:746–774. 935–965.
5. Klien BA. Angioid Streaks: A clinical and histopathologic study. American Journal of Ophthalmology. 1947. 30:955-68.
6. Clarkson JG, Altman RD. Angioid streaks. Surv Ophthalmol. 1982;26:235–46.
7. Gass JDM. Stereoscopic Atlas of Macular Diseases: Diagnosis and Treatment. Vol. 1. 4th ed. St. Louis, MO; Mosby; 1997:120.
8. Gass JD. Stereoscopic Atlas of Macular Diseases. 3rd edition. St Louis: CV Mosby Co; 1987. pp. 102–109.
9. Clarkson JG, Altman RD. Angioid streaks. Surv Ophthalmol. 1982;26:235–246.
10. Paton D. Angioid streaks and sickle cell anemia. Arch Ophthalmol. 1959;62:852–858.
11. Jampol LM, Acheson R, Eagle RC, Jr, Serjeant G, O’Grady R. Calcification of Bruch’s membrane in angioid streaks with homozygous sickle cell disease. Arch Ophthalmol. 1987;105:93–98.
12. Federman JL, Tomer TL, Annesley WH. The macula A Comprehensive Text and Atlas. Baltimore: Williams and Wilkins; 1978. pp. 218–231.
13. Shields JA, Federman JL, Tomer TL, Annesley WH Jr. Angioid streaks Ophthalmoscopic variations and diagnostic problems. Br J Ophthalmol. 1975; 59:257-65.
14. Terry TL. Angioid streaks and osteitis deformans. Trans Am Ophthalmol Soc. 1934;32:555–73.
15. Schatz H. Other retinal pigment epithelial diseases. Int Ophthalmol Clin. 1975; 15:181–97.
16. Shields JA, Federman JL, Tomer TL, Annesley WH Jr. Angioid streaks. Ophthalmoscopic variations and diagnostic problems. Br J Ophthalmol.1975;59:257-65.
17. Dhermy P. Histologie Angioid Streaks. Cosca G, Soubane G, eds. Neovasseaux Sous-Retiniens et al Laser. Paris, France: 1987. 210-1.
18. Shilling JS, Blach RK. Prognosis and therapy of angioid streaks. Trans Ophthalmol Soc U K.1975;95:301–306.
19. Mansour AM, Ansari NH, Shields JA, Annesley WH, Jr, Cronin CM, Stock EL. Evolution of angioid streaks. Ophthalmologica. 1993;207:57–61.
20. Mansour AM, Shields JA, Annesley WH, Jr, el-Baba F, Tasman W, Tomer TL. Macular degeneration in angioid streaks. Ophthalmologica. 1988;197:36–41.
21. Nakagawa, Satoko, et al. "The time course changes of choroidal neovascularization in angioid streaks." Retina 33.4 (2013): 825-833.
22. Lim JI, Lam S. A retinal pigment epithelial tear in a patient with angioid streaks. Arch Ophthalmol. 1990;108:1672-74.
23. Piro P, Scheraga D, Fine S. Angioid streaks: natural history and visual prognosis. In: Fine SL, Owens SL. Management of Retinal Vascular and Macular Disorders. Baltimore, MD: Williams and Wilkins; 1983. p. 136–139.
24. Finger RP, Issa PC,n Ladewig MS et al, Pseudoxanthoma elasticum. Surv Ophthalmol 2009; 54:272-85.
25. Elsner AE Burns SA Weiter JJ Delori FC . Infrared imaging of sub-retinal structures in the human ocular fundus. Vision Res. 1996;36:191–205.
26. Issac PC, Finger RP, Holz FG, Scholl HPN. Multimodal Imaging for characterization of Outer Retinal Pathology in Pseudoxanthoma Elasticum. Invest Ophthalmol Vis Sci. 2009;50:5913-8.
27, Peter Charbel Issa, Robert P. Finger, Frank G. Holz, Hendrik P. N. Scholl. Multimodal Imaging Including Spectral Domain OCT and Confocal Near Infrared Reflectance for Characterization of Outer Retinal Pathology in Pseudoxanthoma Elasticum. Investigative Ophthalmology & Visual Science December 2009, Vol.50, 5913-5918. doi:10.1167/iovs.09-3541
28. Dansingani KK, Naysan J, Freund KB (2015) En face OCT angiography demonstrates flow in early type 3 neovascularization (retinal angiomatous proliferation). Eye 29:703–706.
29. Kubota M, Hayashi T, Arai K, Tsuneoka H. Choroidal neovascularization after blunt ocular trauma in angioid streaks. Clin Ophthalmol. 2013. 7:1347-51.
30. Pece A, Avanza P, Galli L, Brancato R. Laser photocoagulation of choroidal neovascularization in angioid streaks. Retina. 1997; 17(1):12-6.
31. Verhoeff F. Histological findings in a case with angioid streaks. Br J Ophthalmol. 1948;32:531–544.
32. TAP Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: one-year results of 2 randomized clinical trials – TAP report. Treatment of agerelated macular degeneration with photodynamic therapy (TAP) Study Group. Arch Ophthalmol. 1999;117:1329– 1345.
33. TAP Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age related macular degeneration with verteporfin: Two-years results of 2 randomized clinical trials -TAP report 2. Arch Ophthalmol. 2001;119:198–207.
34. VIP Study Group. Verteporfi n therapy of subfoveal choroidal neovascularization in age-related macular degeneration: Two-years results of a randomized clinical trial including lesions with occult with no classic choroidal neovascularization – report 2. Am J Ophthalmol. 2001;131:541–560.
35. Shaikh S, Ruby AJ, Williams GA. Photodynamic therapy using verteporfin for choroidal neovascularization in angioid streaks. Am J Ophthalmol. 2003;135:1–6.
36. Schmidt-Erfurth U, Schlotzer-Schrehard U, Cursiefen C, Michel S,Influence of PDT on expression of vascular endothelial growth factor (VEGF), VEGF Receptor 3, and pigment epithelium-derived factor. Invest Ophthalmol Vis Sci. 2003;44:4473-80.
37. Prabhu VV, Morries RJ, Shah PK, Narendran V. Combination treatment of low fluence PDT and intravitreal ranibizumab for choroidal neovascular membrane secondary to angioid streaks in Paget’s disease. Indian J Ophthalmol 2011;59:306-8. 38. Lee JW, Shin JP, Kim SY. A case of Intravitreal Bevacizumab Injection for the treatment of Choroidal Neovascularization in Angioid Streaks. Korean J Ophthalmol. 2011;25:218-21. 39. Thomas MA, Dickinson JD, Melberg NS et al. Visual results after removal of subfoveal choroidal neovascular membrane. Ophthalmology 1994; 101:1384-96.
1.jpeg)


