Inflammation of retinal tissue causes characteristic fundus findings depending on whether it is full-thickness or partial-thickness or outer or inner retinal involvement.
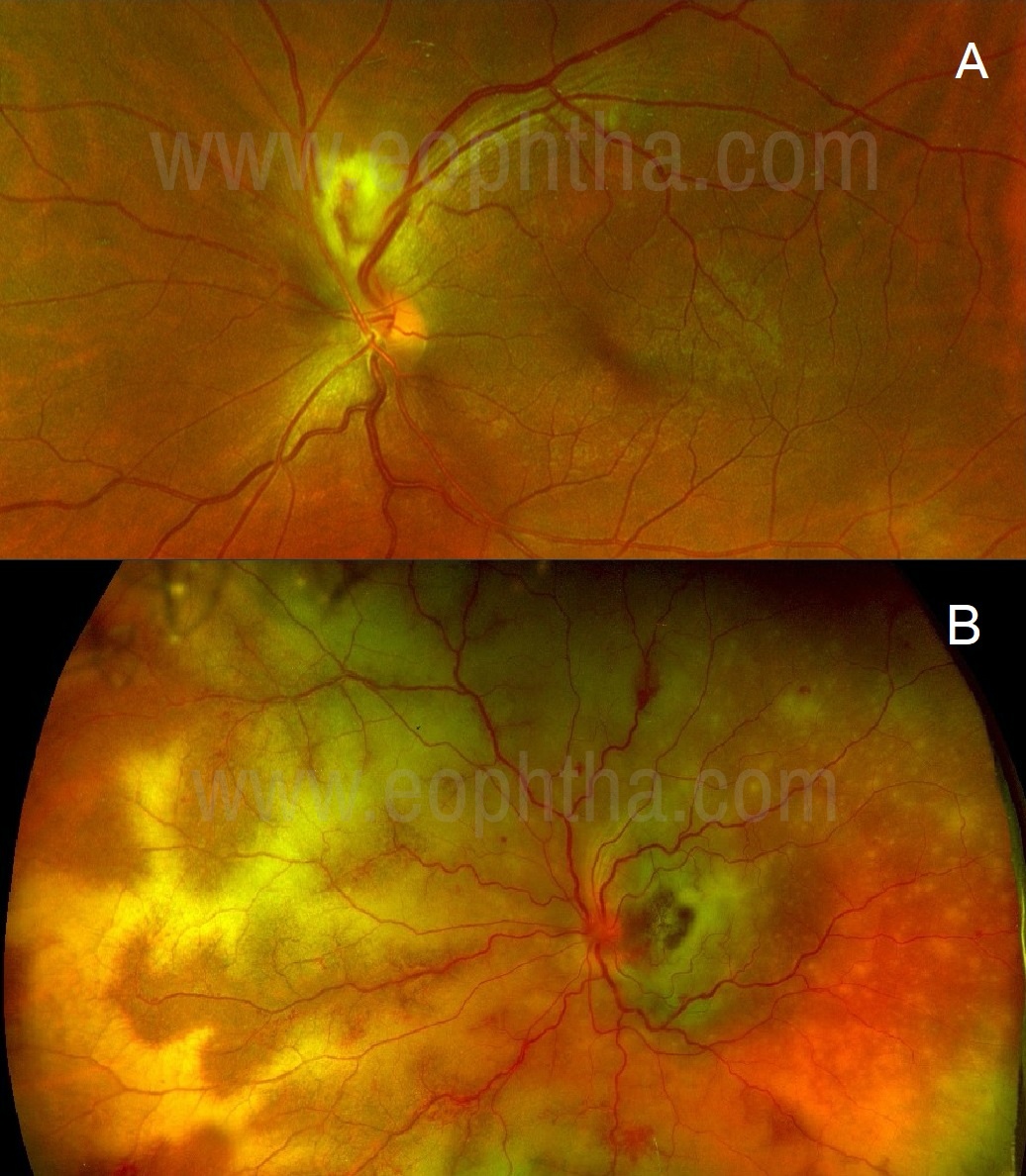
Figure 1A: A case of epidemic retinitis: Bright yellow superficial fluffy lesion superiorly to the disc obscuring retinal vessels passing through it is typical of inner or full-thickness retinitis. 1B: A case of progressive outer retinal necrosis: Yellowish lesions with clear retinal overlying vasculature suggests outer retinitis.
Full-thickness or inner retinal involvement produces bright yellow fluffy lesions with indistinct borders. (Figure 1A) Retinal vasculature passing through these lesions appears obscured. At times, such lesions can mimic cotton-wool spots and may produce moderate to severe vitritis. Outer retinal inflammatory lesions appear dull orange or yellow with indistinct borders, retinal vessels may remain clear over the lesions (Figure 1B) and vitritis could be mild-moderate.
Differentiating retinitis from choroiditis:
It is important to differentiate retinitis from choroiditis as it may narrow down the list of differential diagnoses. In contrast to the above-mentioned retinitis morphology, ophthalmoscopically choroidal lesions appear dull yellowish orange-colored, deeply situated lesions with indistinct borders (Figure 2A), and sometimes with elevated appearance. (Figure 2B) Retinal vessels will appear clear, passing over the lesion in contrast to retinitis where the vessels passing through the lesions are obscured. Vitritis in choroiditis is relatively less pronounced.
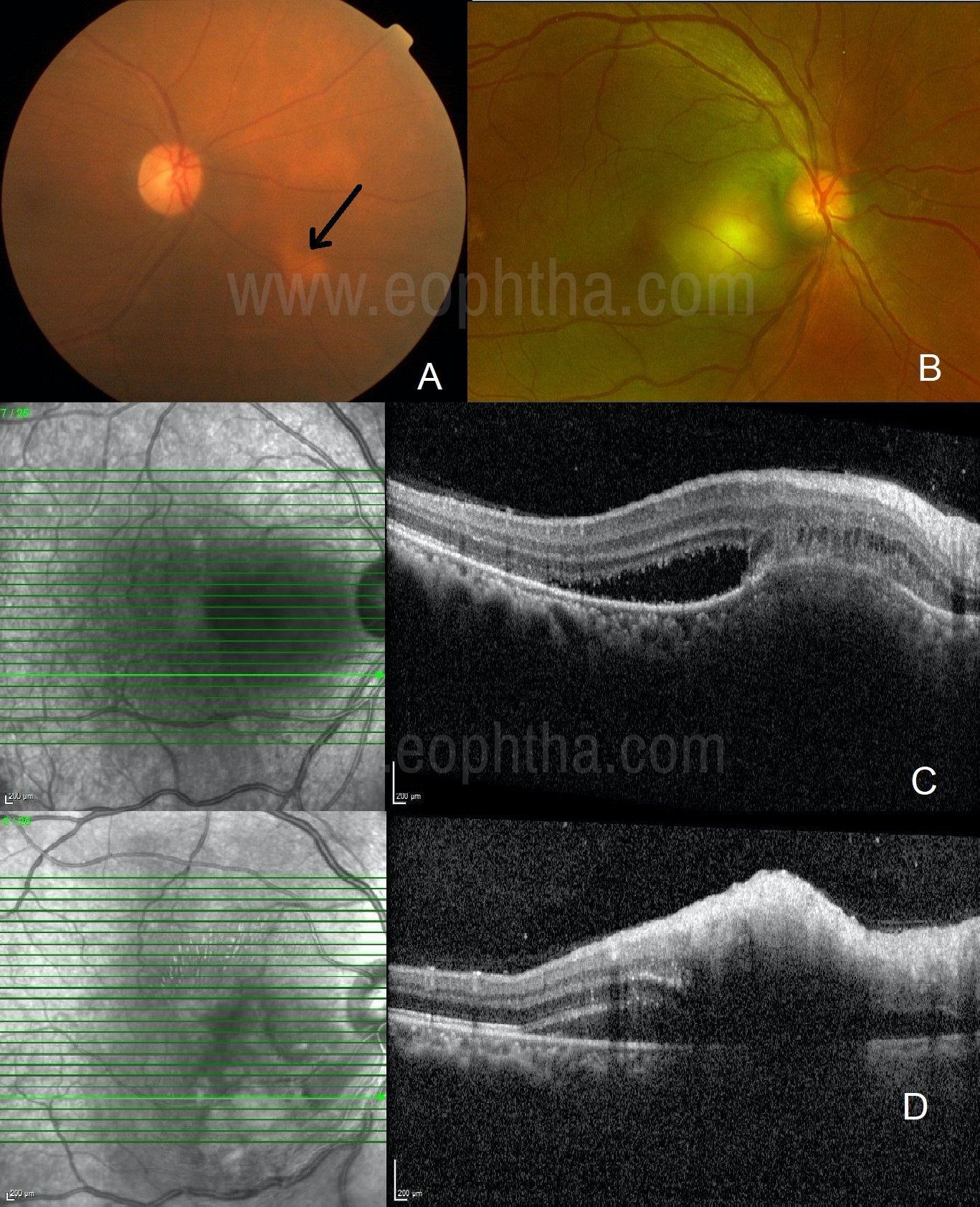
Figure 2A: A case of sarcoidosis: Dull orangish lesion in the inferonasal aspect of the disc with overlying clear vasculature is typical of choroiditis. 2B: A case of tubercular choroiditis: A granulomatous, elevated lesion with retinal vasculature clearly passing over it suggests choroidal granuloma. 2C: A case of tubercular choroiditis: OCT shows choroidal elevation, sub-retinal fibrinous deposit, mild subretinal fluid, and relatively uninvolved retina suggestive of primary choroiditis. 2D: A case of epidemic retinitis: OCT shows high inner-reflectivity of the inner retina with back-shadowing with mild subretinal fluid and flat choroid suggestive of primary retinitis.
Optical Coherence Tomography (OCT) scan will ultimately differentiate choroidal lesion from retinitis. (Figure 2C, D) Indistinct hyperreflective retinal layers generally associated with dot hyper-reflective lesions in the posterior vitreous cavity, without choroidal elevation, suggests the presence of pure retinitis.
Chorio-retinitis and Retino-choroiditis:
Inflammation in ocular tissue may not remain confined to the primarily involved tissue but may spread to adjacent tissue. When the retinal tissue is primarily involved and the inflammation has spread to the choroid, the condition is called retinochoroiditis (Eg. Toxoplasma retinochoroiditis). Vice- versa when the choroid is primarily involved, and the inflammation has passed onto retinal tissue the condition is called chorioretinitis (Eg. Tubercular chorioretinitis).
Associated fundus findings:
Active retinitis can present with associated signs like vasculitis (Eg. Toxoplasma, Behcet’s disease), chorioretinal scars (Eg. Toxoplasma, Tuberculosis), disc and/or macular edema (Eg.Epidemic retinitis), rhegmatogenous retinal detachment (Eg. acute retinal necrosis) or exudative retinal detachment (g. Endogenous endophthalmitis, syphilitic retinitis).
Masquerades of retinitis:
One should be aware that certain non-inflammatory fundus lesions may delude ophthalmologists to start investigation and treatment for retinitis. One should not be mistaken by lipid exudates caused by aneurysms. (Figure 3A, B) Such aneurysms can be signs of Coat’s disease. (Figure 3C, D) The absence of vitritis should raise suspicion in such conditions. Fundus fluorescein angiography (FFA) is the choice of investigation which will reveal the culprit lesion. Emulsifying silicon oil can at times mimic retinitis lesions. (Figure 3 E) It generally appears in the superior retina and OCT scan passing over the lesion can rule out retinitis.
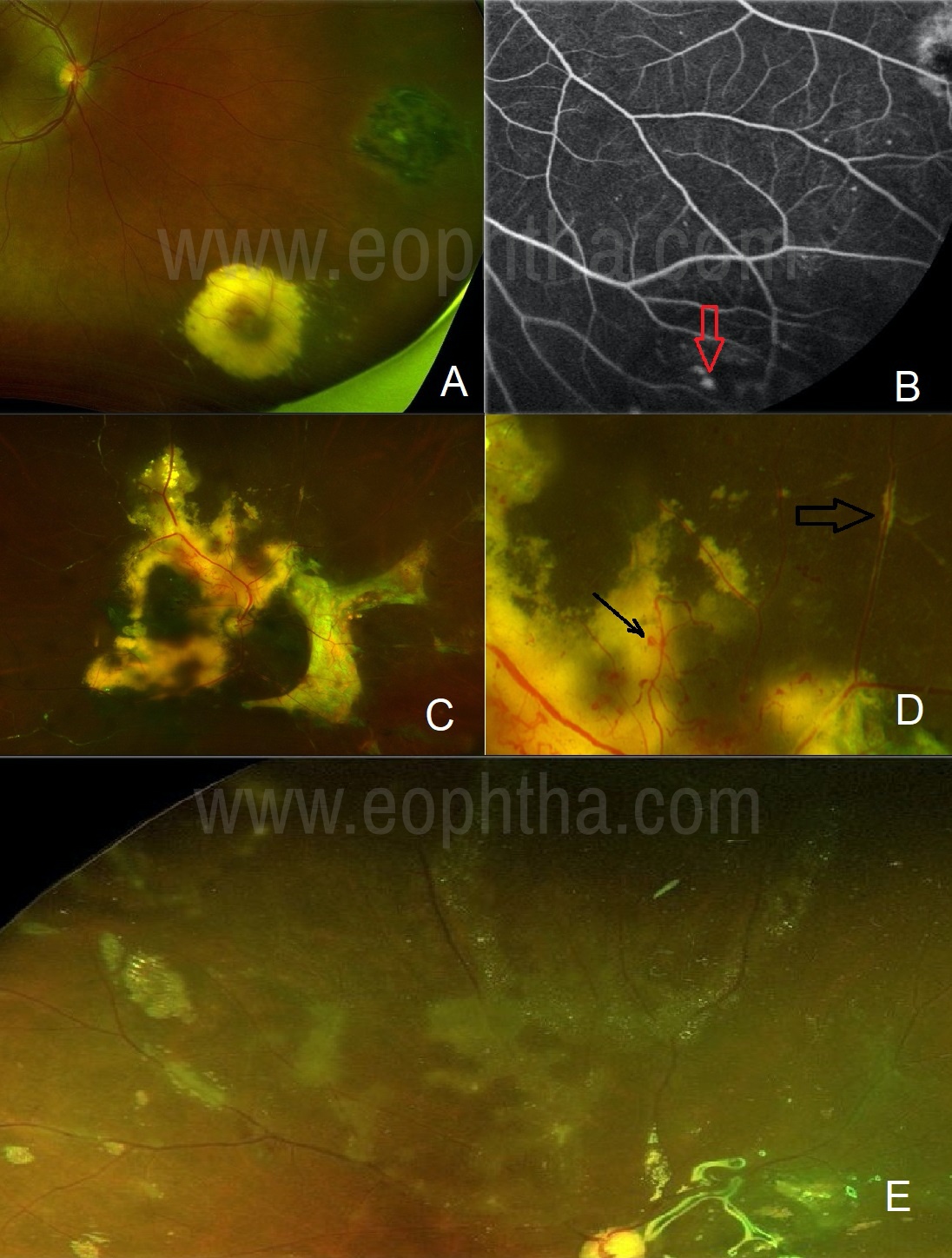
Figure 3A: A case of a peripheral aneurysm with lipid exudates: Yellow lesion with the adjacent scar in the peripheral retina mimics toxoplasma retinitis. 3B: FFA demonstrates the culprit aneurysm (arrow) in the center of the yellow lesion. 3C: A case of Coat’s disease: Subretinal lipid deposits can mimic retinitis lesion.3D: A careful observation will identify the culprit aneurysms (arrow). Also, note the pseudo-sheathing (lipids) along the vessel mimicking vasculitis (broad arrow).3E: Emulsified silicon oil can give the appearance of retinitis and vasculitis.
Masquerades such as lymphoma and leukemia can mimic retinitis lesions. Certain OCT signs may differentiate lymphoma from retinitis,1, 2 but confirmatory diagnosis can be achieved by means of vitreous or chorioretinal biopsy in cases of lymphoma. In the case of leukemia, associated findings such as pseudo-sheathing, Roth’s spots, retinal hemorrhages may point towards masquerades.3 Rarely large choroidal infiltration can be seen on multimodal imaging and FFA may reveal miliary microaneurysms.4, 5 Blood workup including peripheral blood smear, and/or bone marrow biopsy will ultimately prove the diagnosis. Purtscher retinopathy,6 or Putscher-like retinopathy of SLE should be distinguished from true retinitis.7
Etiology of retinitis / retinochoroiditis
Infectious:
Parasites: Toxoplasmosis, Toxocariasis, ruptured cysticercosis, Diffuse Unilateral Subacute Neuroretinitis (DUSN)
Bacteria: Epidemic retinitis (Rickettsiosis), Typhoid, Bartonellosis, Brucellosis, Borreliosis, Syphilis, other bacteria causing endogenous endophthalmitis.
Viruses: Acute Retinal Necrosis (HSV, VZV, CMV, Epstein Barr Virus), Epidemic retinitis (Chikungunya, Dengue, West Nile Virus (WNV), Subacute Sclerosing Panencephalitis (Measles), Retinopathy (HIV, Rubella), Congenital retinochoroiditis (Zika, Ebola)
Non-infectious / Syndromic:
Behcet’s disease, Sarcoidosis (rare), Cancer-associated retinopathy (CAR), Acute Zonal Occult Outer Retinopathy (AZOOR), Multiple Evanescent White Dot Syndrome (MEWDS), Acute Macular Neuroretinopathy (AMN)
Classification:
Necrotizing: HSV, VZV, CMV, Measles, Toxoplasma, Syphilis, Endogenous Endophthalmitis
Non-necrotizing: Epidemic retinitis, Syphilis, Behcet’s, White dot syndromes, DUSN
Common retinitis entities:
Toxoplasma Retinochoroiditis:
Toxoplasma gondii is one of the most common causes of retinochoroiditis worldwide. The infection is acquired from not only cats but also from dogs, birds, contaminated food, and water.8 Humans contract the disease by ingestion of toxoplasma oocyst. The cyst remains dormant in the tissue like the brain, muscles, and retina. Slow growing bradyzoites still within the cyst, when acquires the form of tachyzoites, this rapidly replicating organism causes the manifestation of the disease.
Acute onset of painless blurring of vision without any systemic association is a common presenting complaint. Toxoplasma generally causes posterior uveitis but panuveitis is also not rare. When the anterior segment is involved, mild circumcilliary congestion, granulomatous KPs, mild-moderate anterior chamber reaction can be seen on slit-lamp evaluation. Vitritis may vary depending on the patient’s immune status and stage of presentation. Early presentation in an immunocompetent patient may not show the classical “headlights in fog” appearance. (Figure 4A) Bright yellow retinitis lesion with a fuzzy border may or may not be associated with chorioretinal scars. If the individual has newly acquired the infection, the chorioretinal scar will be absent. Exudative vasculitis (periphlebitis) is generally present along with retinitis. Vascular cuffing which simulates vascular emboli has been described as “Kyrieleis' Arteriolitis” in toxoplasmosis.9
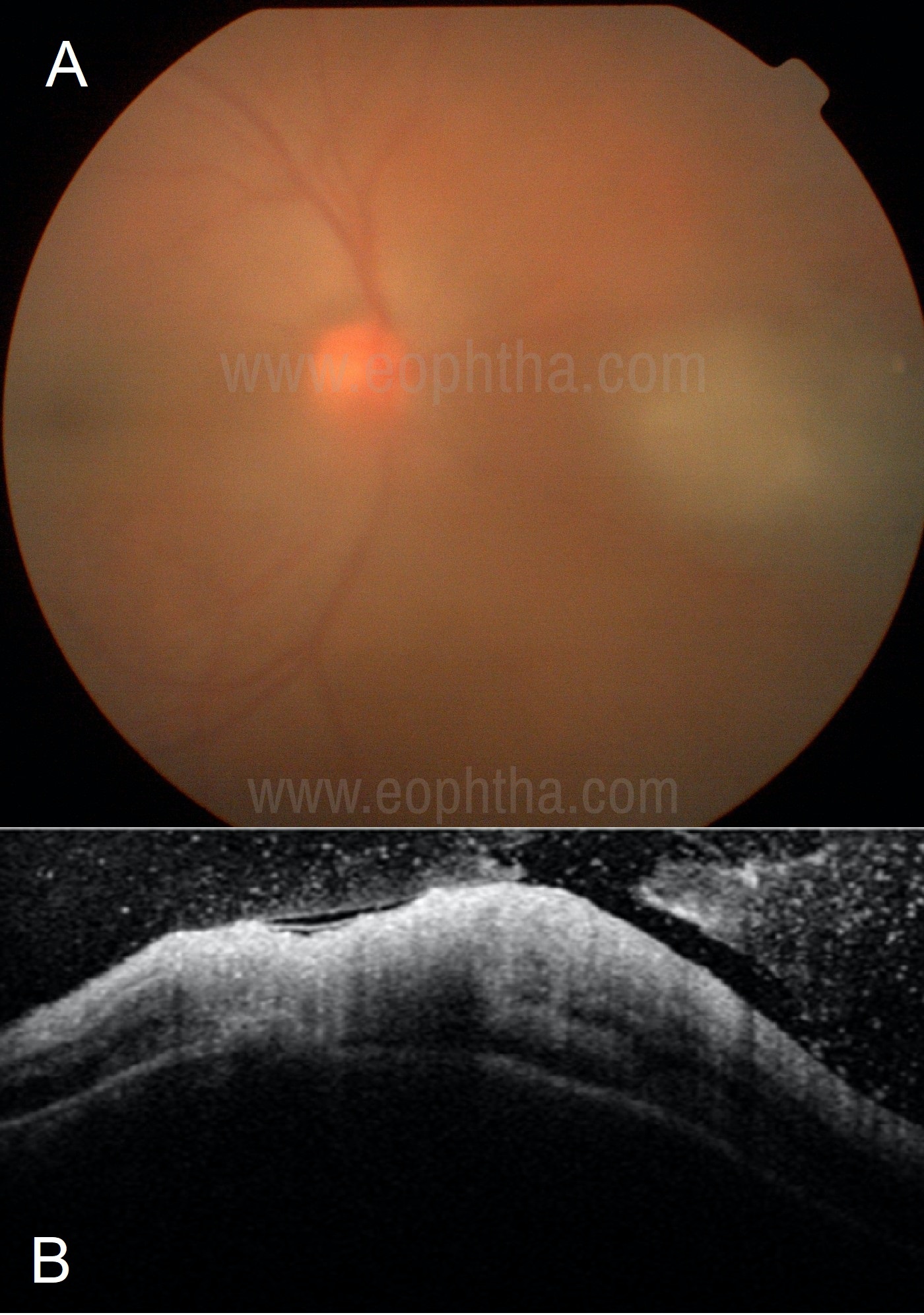
Figure 4A: A dull yellow fluffy lesion nasal to the disc seen through moderate vitreous haze giving the appearance of “headlights in the fog” suggests toxoplasma retinitis. 4B: OCT in a case of toxoplasma retinochoroiditis shows full-thickness retinal involvement with a choroidal elevation and vitritis.
Investigations: OCT scan passing over the retinitis lesion will show full-thickness retinal involvement. Rarely mild-moderate choroidal elevation can also be seen. (Figure 4B) Thick ERM is also typically seen in toxoplasma retinitis.
In most cases the diagnosis is clinical. Positive toxoplasma IgM and IgG levels confirm the diagnosis. But in clinical practice generally, IgM remains negative and, in such cases, high titers of IgG are also helpful to consider the diagnosis of toxoplasmosis. PCR studies from aqueous or from the vitreous fluid may confirm the diagnosis in challenging cases. Yet another useful but rarely used test is Goldmann-Witmer coefficient (GWC) which compares the levels of intraocular antibody (IgG) production to that serum, as measured by ELISA. (GWC = Level of specific IgG in aqueous humour/level of specific IgG in serum)/(Total IgG in aqueous humour/Total IgG in serum. A value of 2 or above is generally taken as evidence of the intraocular synthesis of Toxoplasma specific IgG).10
Treatment: Systemic anti-toxoplasma medications along with steroids titrated to the inflammation is recommended in immunocompetent patients. Among various anti-toxoplasma drugs, Trimethoprim (160mg)/Sulfamethoxazole (800mg) (Bactrim DS twice daily), Azithromycin (500mg OD) and clindamycin (300mg QID) are commonly used. A combination of 2 anti-toxoplasma drugs along systemic steroids is a common practice. In the case of systemic contraindications (Eg. Pregnancy, hypersensitivity) one can opt for intravitreal clindamycin (1 mg/ 0.1 mL) with dexamethasone therapy. In pregnant ladies, oral spiramycin can be used to prevent transmission to the child.
Acute retinal necrosis (ARN):
ARN is a morphological diagnosis, which denotes necrotizing retinitis starting from the peripheral retina and rapidly progressing in circumferential fashion then towards the center. As a rule, causative agents are herpetic viruses, commonly HSV, VZV, CMV, and rarely Epstein-Barr virus, Toxoplasma, and syphilis.
Similar to toxoplasma retinitis patients present with complaints of acute onset of blurring of vision. Pain is generally absent or minimal. ARN is commonly posterior or panuveitis. When the anterior segment is involved, IOP spike can be seen, centrally located or diffuse pigmented granulomatous KPs will be evident. Moderate anterior chamber reaction and moderate-severe vitritis can be seen in immunocompetent patients. Plaque-like yellow multiple retinitis lesions in peripheral retina progressing in a circumferential manner is a typical picture. Occlusive vasculitis, frequently arteriolitis, and rarely Kyrieleis vasculitis has also been described in ARN.11, 12 Optic neuritis, scleritis, and uveal effusion are rare associations.13, 14, 15
Investigations: OCT passing over the retinitis lesions can show full-thickness retinal involvement, tissue loss, so-called “cavernous retinitis”,16 and choroid remains uninvolved unless the patient also develops uveal effusion.
Ocular fluid analysis for PCR may help to find the causative organism and it should be considered where possible as isolation of CMV or other rare organisms warrants changes of conventional anti-viral treatment. Systemic investigations to rule out immunosuppression and other infectious etiologies like toxoplasmosis and syphilis should be considered especially in atypical cases.
Treatment: Intravenous acyclovir 10-15mg/kg TID for 14 days followed by oral acyclovir 800mg 5 times/day until resolution of retinitis lesions is a standard of care treatment for ARN caused by HSV and VZV. Few authors found oral valacyclovir as effective as intravenous acyclovir.17 Although standard valacyclovir dose is 1gm TID, higher doses may require in non-responding cases.18 Acyclovir resistance is rare but should be considered if retinitis is worsening with standard treatment in PCR proven cases. In the case of systemic contraindications, antiviral therapy can be administered intravitreally with ganciclovir 2mg/0.4ml twice a week, especially if CMV is isolated from ocular fluid.
Complications: Retinal detachment is the most common complication seen within weeks or months of the onset of the disease. Controversies exist whether prophylactic barrage laser prevents retinal detachment or not. ARN with sclerokeratouveitis, optic neuritis, posterior scleritis, or uveal effusion, frozen globe syndrome are rare manifestations.
CMV retinitis:
CMV retinitis is the most common cause of retinitis in immunocompromised patients, especially in HIV patients with low CD4 counts (50-100 cells/mm3) but rarely can also occur in immunocompetent patients.19Frequently it is bilateral. The anterior chamber may or may not be involved. Diffuse small granulomatous KPs can be seen. The anterior chamber may show a 1+ or 2+ cellular reaction. Vitritis is mild or may remain absent in severely immunocompromised patients. The posterior pole is commonly involved. Retinitis lesions are generally associated with hemorrhages, classically described as “pizza pie or tomato ketchup appearance”. Peripheral retinitis lesions may show granularity (a granular form of CMV retinitis). The fulminant form presents with hemorrhagic necrosis with cloudy retinitis lesions. Rarely perivascular form may present with “frosted branch” angiitis.
Investigations: OCT may show full-thickness retinal involvement with tissue loss. Choroid remains uninvolved. HIV screening with CD 4 counts should be done in uninvestigated cases. If CD4 counts are less than 50cells/mm3 patient should be screened frequently, as active retinitis not involving macula may remain asymptomatic.20
Ocular fluid analysis for PCR is needed in atypical cases to differentiate from other herpetic viruses and to make choice between acyclovir and ganciclovir. In small children isolation of CMV from blood can also aid in the diagnosis when the ocular fluid analysis is not possible,21 or else one may rely on positive IgM and IgG titers in such cases.
Treatment: Ganciclovir is the drug of choice for the treatment of CMV retinitis. Acyclovir is not as effective as the virus lacks thymidine kinase. In immunocompromised patients, I.V. ganciclovir is given at 5 mg/kg twice daily for 2-3 weeks followed by daily 5 mg/kg infusions. Oral valganciclovir 900mg BD for 3 weeks followed by a maintenance dose of 900mg OD can be considered. Advocating intravitreal ganciclovir therapy, 2mg/0.05ml twice weekly can be considered in case of systemic contraindications or to aim for faster recovery.
Complications: Immune recovery uveitis (IRU) is a common complication after commencing HAART. Such patients may develop CME, vitreous haze, and anterior uveitis while retinitis is resolving and require steroid therapy.22 In the case of contraindication for systemic steroids, one may opt for intravitreal dexamethasone therapy which can be combined with ganciclovir if retinitis is still resolving. Peribulbar and intravitreal triamcinolone for IRU has also been reported.23 Incidence of RD is rare and commonly seen in peripheral form or in case of frank ARN caused by CMV.
Progressive outer retinal necrosis (PORN):
PORN is a rare but more severe form of retinitis in immunocompromised patients. CD4 counts can range from 1-130.24 PORN is notorious for its rapid progression leading to blindness. VZV is the common causative agent followed by HSV and CMV.25
Yellowish large placoid lesion at the posterior pole with clear retinal superficial vasculature and the clear vitreous and anterior chamber is a typical presentation. (Figure 1b) Such lesions can be confused with syphilitic placoid retinitis. Peripheral progression of the lesion may develop a retinal detachment.
Treatment: PORN requires aggressive management with antiviral therapy. Both intravenous (acyclovir/gancyclovir) as well as intravitreal antiviral agents (ganciclovir) are needed to halt the progression of the disease. When systemic ganciclovir is not indicted in severely immunocompromised patients due to low blood counts, intravitreal antiviral therapy remains the only option.26
Epidemic retinitis:
Epidemic retinitis (ER) is an umbrella term in which clubs together retinitis post community outbreaks like, Rickettsiosis, Dengue, Chikungunya, West-Nile virus, and several other yet unknown organisms.27 It is commonly seen in tropical countries frequently after the rainy season. In south India, each year the epidemic starts in months of July-August, gradually picks up with spikes in November-February, then the tide recedes from April onwards and the pattern repeats each year suggesting a seasonal variation.
By default, patients have a history of a recent fever. In many cases, the diagnosis remains pyrexia of unknown origin. The patient may give a history of joint pain, and/or skin rash. Ocular symptoms typically start after 1-4 weeks of the fever. The patient comes to ophthalmologist with fading skin rashes and a history of a resolved fever. The presentation is acute with rapid onset of loss of vision, generally painless.
Depending on macular involvement and disc status vision may vary from 6/6 to Hand Motions. IOP is generally within normal limits. The ocular examination may reveal mild-moderate circumciliary congestion, non-granulomatous KPs, mild-moderate AC reaction, and rarely posterior synechiae. Anterior segment may remain uninvolved. Posterior segment examination can show mild-moderate vitreous haze. Fundus typically shows multiple yellowish cotton wool spot-like retinitis lesions mainly at the posterior pole and around the disc with or without macular edema and/or disc edema. Vascular sheathing is an uncommon finding. The condition is frequently bilateral and can be asymmetric.
Investigations: OCT scan can demonstrate the severity of macular edema. Mild-Moderate subretinal fluid with cystoid macular edema which can be diffuse of sectoral is a common finding.28 OCT scan passing over retinitis lesions may reveal inner retinal hyper-reflectivity with after shadowing.
FFA can pick up subclinical vasculitis and may demonstrate vasculitic leakage in the area of CWS-like retinitis lesions. Early hypofluorescence and late staining of borders of retinitis lesions can be seen on FFA.
In an attempt to find the cause, one may advocate a battery of investigations: readily available and cheaper Weil Felix test, or costly but gold standard tests like immunofluorescence assay (IFA) for the diagnosis of rickettsial disease; serology (IgG, IgM) for Chikungunya, Dengue, and WNV. PCR from ocular fluid for the above organisms can be considered if available in challenging cases but can be avoided if the presentation is typical, as regardless etiology this condition is self-resolving in most of the cases.
Treatment: Being a self-limiting condition, one can opt for observation. But due to the aggressive presentation of the disease with macular edema, most of the ophthalmologist prefers to commence the treatment. In case of rickettsial as well as in the case of dengue-related ER, oral doxycycline with or without systemic steroids can be given.28, 29
In the presence of optic neuritis or clinically visible vasculitis, or severe macular ischemia patients may require systemic, intraocular, or peri-ocular steroids.
Complications: ER generally has a good visual prognosis. Mild retinal thinning and loss of small vessel architecture at the site of retinitis lesions can be seen on OCT, OCT-A, or on FFA scan years after resolution of retinitis. Occlusive vasculitis with neovascularization and vitreous hemorrhages are rare complications.
Behcet’s disease:
Uveitis with associated oro-genital ulcers is a well-known clinical correlation of Behcet’s disease (BD). The International Criteria for Behcet’s Disease (ICBD), created in 2006, includes oral aphthosis, genital aphthosis, skin lesions (Pseudo Folliculitis, Erythema Nodosum), uveitis, systemic vascular manifestations (superficial phlebitis, deep vein thrombosis), and a positive Pathergy test. Genital aphthous lesions and uveitis get each 2 points and other manifestations at one point each. A score ≥3 is diagnostic of BD. Uveitis can be presenting features of BD or else can be the only manifestation of BD precluding the diagnosis.
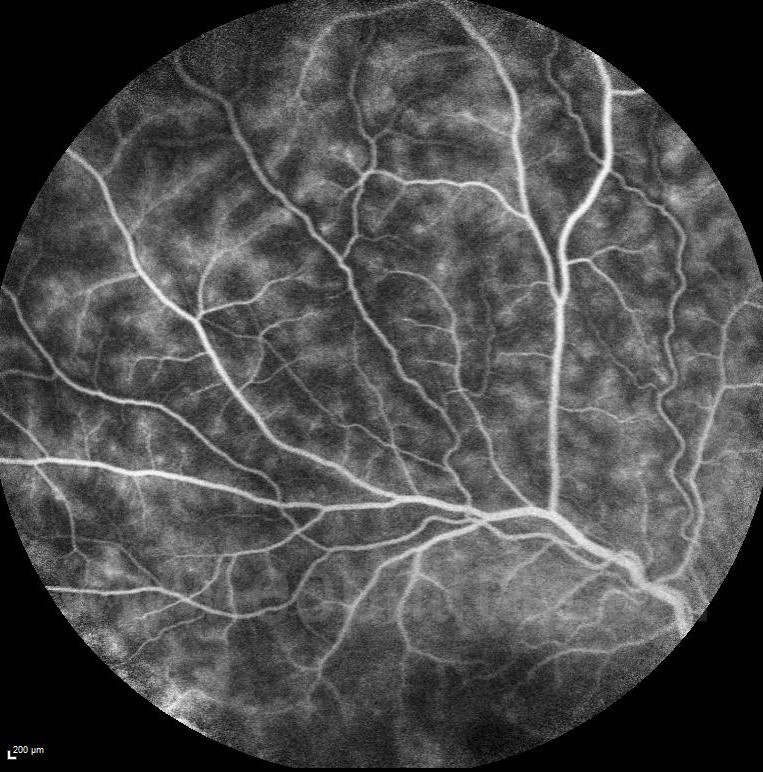
Figure 5: A case of Behcet’s disease, shows perivascular “fern-like leakage” on FFA in active inflammation.
Bilateral non-granulomatous mobile hypopyon uveitis is a classical presentation. Intermediate uveitis and posterior uveitis in the form of occlusive vasculitis and transient retinitis lesions are common manifestations of the disease. Retinitis lesions can range from small yellowish-white dots to larger multiple spots or even plaques, involving superficial and deep retinal layers.30 They may resolve spontaneously in weeks without scarring. Exudative retinal detachment due to massive infiltrates is a rare manifestation.31 Small hemorrhages can be seen abutting those lesions.
Investigations: OCT scan passing through these lesions can show partial-thickness or full-thickness involvement of retina. Choroid remained uninvolved.
FFA can show vasculitic leakage from major vessels or fern-like perivascular leakage in intermediate uveitis. (Figure 5)
In most cases, diagnosis is clinical, based on the diagnostic criteria (vide supra). When there is no clue from the systemic examination, pathergy test, and HLA B51 typing can be done. HLA-B51 positivity along with a hypoactive ERAP1 (Endoplasmic reticulum aminopeptidase-1) allotype contributes to Behçet's disease risk.32
Treatment: Most of the patient requires immunosuppressive therapy to avoid relapses. Apart from conventional immunomodulatory therapy (IMT), biologics, Interferon, and colchicine are popular treatment choices in BD. Intravitreal steroids as well as anti-VEGF have been used for CME. Being a systemic disease and bilateral disease, it is recommended to rely on systemic therapy than local.
Complications: Occlusive vasculitis can be the root cause of several ocular complications like neo-vascularization, vitreous hemorrhage, neovascular glaucoma, ischemic maculopathy. Severe chronic uncontrolled inflammation can ultimately lead to a phthisis bulbi.
Syphilis:
A sexually transmitted disease caused by spirochete-Treponema pallidum, popularly also known as “the great imitator” can present with necrotizing or non-necrotizing retinitis of varied morphology, multifocal spot-like infiltrative lesions to large placoid lesions, partial thickness (superficial retinal precipitates) or full-thickness retinal involvement, with or without vasculitis or retinal detachment. Multifocal spot-like retinitis can mimic Behcet’s retinitis, while placoid lesion at the posterior pole (acute syphilitic placoid posterior chorioretinopathy) can be mistaken for PORN, AZOOR or Serpiginous choroiditis. Placoid lesions starting from the peripheral retina may give the appearance of ARN of viral etiology.33 A combination of these lesions in both the eyes especially in the immunocompromised individuals should raise suspicion for syphilis. A “ground glass” with multifocal spot-like retinitis associated with vasculitis said to be characteristic of syphilis.34 These precipitates may represent a collection of the organism along with inflammatory cells. Vasculitis is frequently associated with retinitis lesions and can affect both arteries and/or veins. Both rhegmatogenous or exudative retinal detachment can be seen in syphilitic retinitis.
Investigations: OCT can show inner retinal hyper-reflectivity in case of superficial retinal precipitates, indistinct retinal layers in case of full-thickness involvement, retinal thinning at the resolution of retinitis. Loss of the external limiting membrane, accumulation of subretinal fluid, and punctate hyperreflectivity in the choroid can be seen on the placoid posterior chorioretinitis form. Multimodal imaging to identify patterns of pre-retinal, retinal, retinochoroidal and optic nerve involvement can aid to the diagnosis.35
A nontreponemal tests, such as the Venereal Disease Research Laboratory (VDRL) and Rapid Plasma Reagin (RPR), and treponemal tests, such as the fluorescent treponemal antibody absorbed (FTA-ABS), and treponema pallidum haemagglutination (TPHA) tests are used in the diagnosis of syphilis. TPHA is a reliable (sensitivity >95%, specificity >99%) 36 and cheaper test routinely used by uveitis specialists in India. Isolation of the organism from ocular fluid on culture is not possible but PCR can be a sensitive and quick test to confirm the diagnosis.37
Treatment: IV crystalline penicillin G 18–24MU per day (4 MU every 4 hours) for 10 to 14 days or IM procaine penicillin G 2.4 MU once a day with oral probenecid 500mg QID, for 10 to 14 days is a recommended treatment for ocular syphilis.38 Alternatively, especially in patients with penicillin allergy, ceftriaxone or doxycycline can be considered for the treatment. Intravitreal ceftazidime has been used in rare occasions.39 Pretreatment with steroids before or during the penicillin therapy40 is not only useful to avoid Jarish–Herxheimer's reaction but also to avoid collateral damage to uveal tissue due to inflammation.
Candida:
In contrast to endophthalmitis pictures, candida can also present as focal or multifocal retinitis without vitreous fluff balls or significant vitritis.41 Candida albicans followed by Candida parapsilosis and Candida tropicalis are common culprit organisms.42 Long-term intravenous therapy, indwelling bladder catheters, urinary tract infections, gastrointestinal surgery, immunosuppression, malignancy, debilitation, and intravenous drug abuse are common risk factors.
Focal or multifocal deep yellow-white retinal infiltrate approximately 250µm diameter, are typical lesions seen at the posterior pole or in the periphery. Nerve fiber layer infarcts, hemorrhages, Roth spots without chorioretinal infiltrates are other non-specific signs. Vitreous haze can be mild-moderate.
Investigations: FFA can show early hyperfluorescence and late staining of the lesion. OCT may demonstrate inner retinal, full-thickness retina, or choriocapillaris involvement. A peculiar hyper-reflective preretinal aggregate obscuring the underlying retina due to a shadowing effect has been described as a "rain-cloud" sign.43
Treatment: Candida retinitis readily responds to amphotericin or voriconazole or fluconazole. Intravenous Voriconazole 6 mg/kg every 12 hours for 24 hours, then at 3 mg/kg every 12 hours followed by oral voriconazole at 200 mg twice a day or amphotericin 0.7–1.0 mg/kg per day, for 3–7 days, followed by oral fluconazole (400 mg/d) or voriconazole can be given.44 Intravitreal voriconazole 100 micrograms /0.1 mL or amphotericin 5 micrograms/0.1 mL is also a better treatment option in the treatment of candida retinitis.
Complication: Unless identified and treated promptly, candida chorioretinitis can quickly progress to full-fledged endophthalmitis. Choroidal neovascular membrane secondary to candida chorioretinitis is a rare complication.45
Toxocara:
Similar to toxoplasma, Toxocara is also a parasitic infection acquired from cats and dogs. Toxocara cati and Toxocara canis are the nematodes implicated in the formation of granulomas in the ocular tissue. Children are the most affected.
A white triangular lesion in the peripheral fundus with a vitreous veil extending towards the optic disc is a typical presentation of ocular toxoplasma. Rarely when toxocara involves posterior pole or mid periphery the lesion can present as retinitis. White superficial granulomatous lesion of various sizes can be seen at the posterior pole or in the mid periphery. (Figure 6) Toxocara lesions occasionally can show vasculitis, and macular edema. Intraocular inflammation is significant when the parasite is dead. This can be grade 3-4 vitritis along with hypopyon, confusing clinical picture with endogenous endophthalmitis, especially in children.
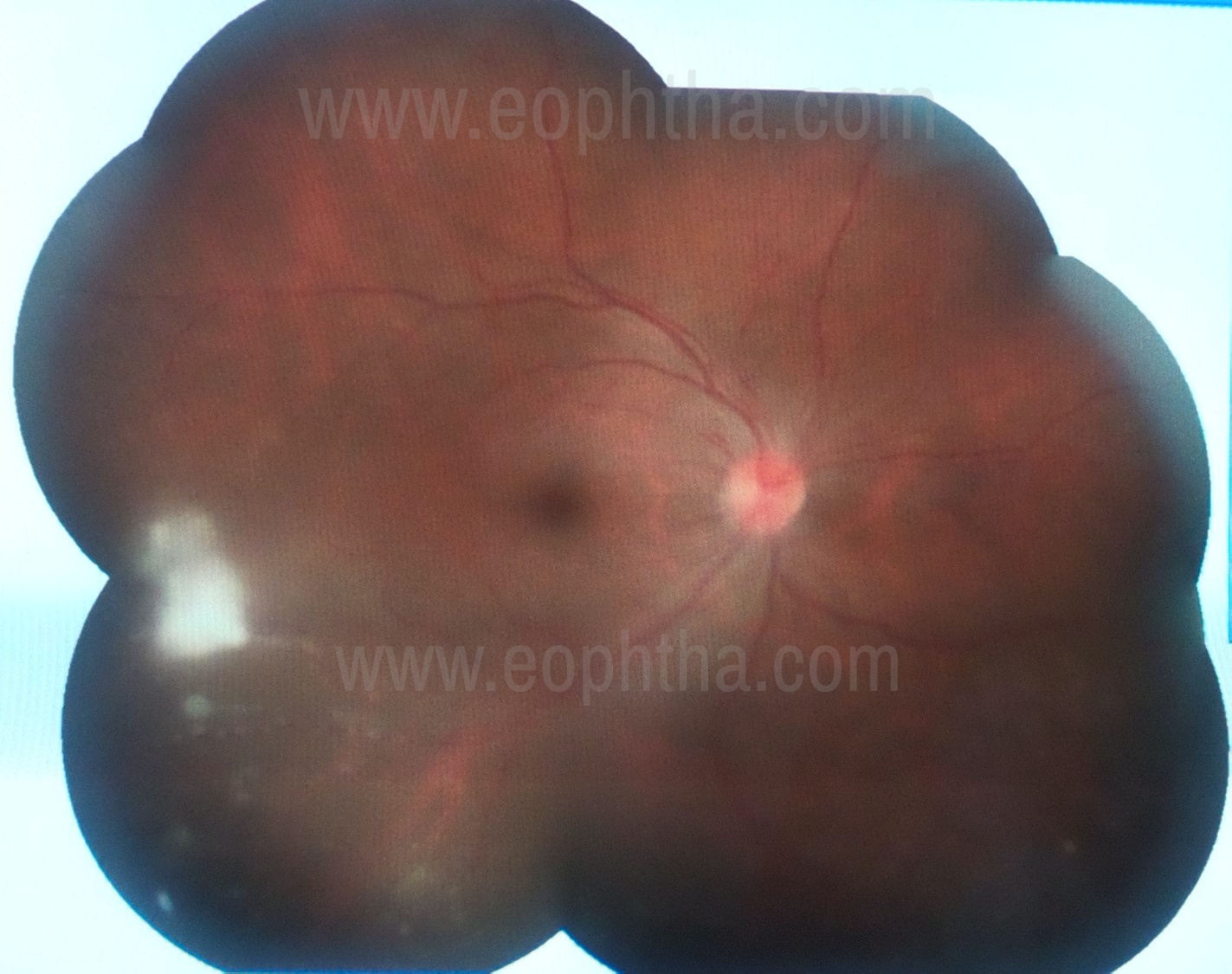
Figure 6: Whitish exudative retinal lesion seen temporally suggestive of toxocara retinitis.
Investigations: OCT passing through the posterior pole lesions may demonstrate the involvement of inner retina with a dome-shaped elevation or protruding into the vitreous cavity.
Diagnosis is generally clinical based on the classical appearance of fundus lesions and a history of contact with pets. ELISA for Toxocara is a reliable test with good sensitivity (around 78%) but not readily available in India. Alternatively, one can evaluate blood and/or ocular fluid for high counts of eosinophils. Surgical excision and histopathology of the granuloma may show remnants of the parasite.
Treatment: Few authors recommend treating Toxocara lesions with anti-helminthes along with steroids while others prefer steroid only therapy or surgery to excise the granuloma. The commonly used drug is albendazole 400mg OD for 5 days- 2 weeks along with steroids titrated to the grade of inflammation.
Complications are the formation of retinal bands and traction ultimately leading to retinal detachment and rarely vitreous hemorrhages. Chronic inflammation may develop macular edema.
Rare causes:
Retinitis rarely can be seen in sarcoidosis in the form of retinal granuloma.46 Pre-retinal multiple small granulomas located inferior and anterior to the equator are described as 'lander sign'.
Punctate deep retinitis like lesions in rhegmatogenous RD,47 and autoimmune retinopathy in SLE are a rare presentation.48
Graphic Courtesy: Dr.Aayesha Khanum
References:
1. Barry RJ, Tasiopoulou A, Murray PI, et al. Characteristic optical coherence tomography findings in patients with primary vitreoretinal lymphoma: a novel aid to early diagnosis. Br J Ophthalmol. 2018;102(10):1362‐1366. doi:10.1136/bjophthalmol-2017-311612 2. Deák GG, Goldstein DA, Zhou M, Fawzi AA, Jampol LM. Vertical Hyperreflective Lesions on Optical Coherence Tomography in Vitreoretinal Lymphoma. JAMA Ophthalmol. 2019;137(2):194‐198. doi:10.1001/jamaophthalmol.2018.5835 3. Huang PK, Sanjay S. Visual Disturbance as the first Symptom of Chronic Myeloid Leukemia. Middle East Afr J Ophthalmol. 2011;18(4):336‐338. doi:10.4103/0974-9233.90143 4. Kawali A, Mahendradas P, Hazarika D, Shetty R. Multimodal imaging in a rare case of leukaemic masquerade. Can J Ophthalmol. 2018;53(5):e176‐e179. doi:10.1016/j.jcjo.2018.01.004 5. Kawali A, Mahendradas P, Sanjay S, Shetty R. Miliary microaneurysms: An angiographic biomarker of leukemic retinopathy? Indian J Ophthalmol 2020;XX:XX-XX. 6. Hamoudi H, Nielsen MK, Sørensen TL. Optical Coherence Tomography Angiography of Purtscher Retinopathy after Severe Traffic Accident in 16-Year-Old Boy. Case Rep Ophthalmol Med. 2018;2018:4318354. Published 2018 Mar 1. doi:10.1155/2018/4318354 7. Dai Ye, Chi Wei. Purtscher-like retinopathy as a first manifestation of systemic lupus erythematosus BMJ 2018; 360 :j5551 8. Palanisamy M, Madhavan B, Balasundaram MB, Andavar R, Venkatapathy N. Outbreak of ocular toxoplasmosis in Coimbatore, India. Indian J Ophthalmol2006;54:129-31 9. Pichi F, Veronese C, Lembo A, et al. New appraisals of Kyrieleis plaques: a multimodal imaging study. Br J Ophthalmol. 2017;101(3):316‐321. doi:10.1136/bjophthalmol-2015-308246 10. Garweg JG, de Groot-Mijnes JD, Montoya JG. Diagnostic approach to ocular toxoplasmosis [published correction appears in Ocul Immunol Inflamm. 2011 Dec;19(6):456]. Ocul Immunol Inflamm. 2011;19(4):255-261. doi:10.3109/09273948.2011.595872 11. Francés-Muñoz E, Gallego-Pinazo R, López-Lizcano R, García-Delpech S, Mullor JL, Díaz-Llopis M. Kyrieleis' vasculitis in acute retinal necrosis. Clin Ophthalmol. 2010;4:837-838. Published 2010 Jul 30. doi:10.2147/opth.s11960 12. Chawla R, Tripathy K, Sharma YR, Venkatesh P, Vohra R. Periarterial Plaques (Kyrieleis' Arteriolitis) in a Case of Bilateral Acute Retinal Necrosis. Semin Ophthalmol. 2017;32(2):251‐252. doi:10.3109/08820538.2015.1045153 13. Murugan SB, Velis GB, Sindal MD. Bilateral acute retinal necrosis associated with bilateral uveal effusion in an immunocompetent patient: A challenging association. Indian J Ophthalmol. 2018;66(6):866‐868. doi:10.4103/ijo.IJO_954_17 14. RAMSAY A, CUNNINGHAM E, PAVESIO CAcute retinal necrosis presenting with scleritis and raised intraocular pressureBritish Journal of Ophthalmology 2000;84:1203 15. Witmer MT, Pavan PR, Fouraker BD, Levy-Clarke GA. Acute retinal necrosis associated optic neuropathy. Acta Ophthalmol. 2011;89(7):599‐607. doi:10.1111/j.1755-3768.2010.01911.x 16. Invernizzi A, Agarwal A, Ravera V, Oldani M, Staurenghi G, Viola F. OPTICAL COHERENCE TOMOGRAPHY FINDINGS IN CYTOMEGALOVIRUS RETINITIS: A Longitudinal Study. Retina. 2018;38(1):108‐117. doi:10.1097/IAE.0000000000001503 17. Baltinas J, Lightman S, Tomkins-Netzer O. Comparing Treatment of Acute Retinal Necrosis With Either Oral Valacyclovir or Intravenous Acyclovir. Am J Ophthalmol. 2018;188:173‐180. doi:10.1016/j.ajo.2018.02.001 18. Senter G, Nascimento HMD, Belfort R Junior. Acute Retinal Necrosis: Is The Current Valacyclovir Regimen Adequate?.Ocul Immunol Inflamm. 2020;28(4):665‐666. doi:10.1080/09273948.2019.1616771 19.Karkhaneh R, Lashay A, Ahmadraji A. Cytomegalovirus retinitis in an immunocompetent patient: A case report. J CurrOphthalmol. 2016;28(2):93-95. Published 2016 Jan 11. doi:10.1016/j.joco.2015.12.003 20. Tun N, London N, Kyaw MK, et al. CMV retinitis screening and treatment in a resource-poor setting: three-year experience from a primary care HIV/AIDS programme in Myanmar. J Int AIDS Soc. 2011;14:41. Published 2011 Aug 15. doi:10.1186/1758-2652-14-41 21.Mahendradas P, Sinha S, Vinekar A, Pradeep M, Shetty BK. Bilateral acute necrotizing retinitis due to cytomegalovirus infection in an infant: Challenging case report. Am J Ophthalmol Case Rep. 2019;16:100553. Published 2019 Sep 10. doi:10.1016/j.ajoc.2019.100553 22. Yeo TH, Yeo TK, Wong EP, Agrawal R, Teoh SC. Immune recovery uveitis in HIV patients with cytomegalovirus retinitis in the era of HAART therapy-a 5-year study from Singapore. J Ophthalmic Inflamm Infect. 2016;6(1):41. doi:10.1186/s12348-016-0110-3 23. SIRIMAHARAJ, MAYTINEE MD*; ROBINSON, MICHAEL R. MD†; ZHU, MEIDONG PHD*; CSAKY, KARL G. MD†; DONOVAN, BASIL MD‡§; SUTTER, FLORIAN MD*; GILLIES, MARK C. MD, PHD*‡ INTRAVITREAL INJECTION OF TRIAMCINOLONE ACETONIDE FOR IMMUNE RECOVERY UVEITIS, Retina: May 2006 - Volume 26 - Issue 5 - p 578-580 24. Ormerod LD, Larkin JA, Margo CA, Pavan PR, Menosky MM, Haight DO, Nadler JP, Yangco BG, Friedman SM, Schwartz R, Sinnott JT. Rapidly progressive herpetic retinal necrosis: a blinding disease characteristic of advanced AIDS. Clin Infect Dis. 1998 Jan;26(1):34-45; discussion 46-7. PubMed PMID: 9455507 25. Sfeir M. Cytomegalovirus implicated in a case of progressive outer retinal necrosis (PORN). J Clin Virol. 2015;69:86-90. doi:10.1016/j.jcv.2015.04.006 26. Gore DM, Gore SK, Visser L. Progressive Outer Retinal Necrosis: Outcomes in the Intravitreal Era. Arch Ophthalmol. 2012;130(6):700–706. doi:10.1001/archophthalmol.2011.2622 27. Kawali A, Mahendradas P, Mohan A, Mallavarapu M, Shetty B. Epidemic Retinitis. Ocul Immunol Inflamm. 2019;27(4):571‐577. doi:10.1080/09273948.2017.1421670 28. Kawali A, Srinivasan S, Mohan A, Bavaharan B, Mahendradas P, Shetty B. Epidemic Retinitis with Macular Edema -Treatment Outcome with and without Steroids [published online ahead of print, 2020 Jan 21]. Ocul Immunol Inflamm. 2020;1‐5. doi:10.1080/09273948.2019.1704792 29. Bhattacharjee B, Bhattacharya S, Sardar B, et al. Dengue and doxycycline-experience in a tertiary care hospital in eastern India in the year 2017 - An initial report. J Pharmacol Ther Res.2018;2:14–17 30. Kötter I, Eckstein AK, Stübiger N, et al. Treatment of ocular symptoms of Behçet’s disease with interferon α2a: a pilot study. British Journal of Ophthalmology 1998;82:488-494. 31. Vrabec TR. Exudative retinal detachment in Behçet disease. Arch Ophthalmol. 2001;119(9):1383-1386. 32. Takeuchi M, Ombrello MJ, Kirino Y, et al. A single endoplasmic reticulum aminopeptidase-1 protein allotype is a strong risk factor for Behçet's disease in HLA-B*51 carriers. Ann Rheum Dis. 2016;75(12):2208-2211. doi:10.1136/annrheumdis-2015-209059 33. Agarwal, M., Ranjan, R., Paul, L. et al. Syphilitic uveitis misdiagnosed as viral retinitis—a misleading history. J OphthalInflamm Infect 8, 22 (2018). https://doi.org/10.1186/s12348-018-0164-5 34. Browning DJ. Posterior segment manifestations of active ocular syphilis, their response to a neurosyphilis regimen of penicillin therapy, and the influence of human immunodeficiency virus status on response. Ophthalmology 2000;107:2015-23. 35. Pichi F, Neri P. Multimodal imaging patterns of posterior syphilitic uveitis: a review of the literature, laboratory evaluation and treatment. Int Ophthalmol. 2020;40(5):1319‐1329. doi:10.1007/s10792-020-01285-9 36. Wiwanitkit V. A cost-utility analysis of Treponema pallidum haemagglutination (TPHA) testing for syphilis screening of blood donors: is the TPHA test useful for syphilis screening in a blood centre? Blood Transfus. 2009;7(1):65‐66. doi:10.2450/2008.0024-08 37. Müller M, Ewert I, Hansmann F, et al. Detection of Treponema pallidum in the vitreous by PCR. Br J Ophthalmol. 2007;91(5):592‐595. doi:10.1136/bjo.2006.110288 38. Dutta Majumder P, Chen EJ, Shah J, et al. Ocular Syphilis: An Update. Ocul Immunol Inflamm. 2019;27(1):117‐125. doi:10.1080/09273948.2017.1371765 39. Sood AB, Pearce WA, Workowski KA, Lockwood J, Yeh S. Combined Intravitreal and Systemic Antibiotic Therapy in a Patient with Syphilitic Uveitis. Ocul Immunol Inflamm. 2019;27(1):131‐133. doi:10.1080/09273948.2017.1385817 40. Butler T. The Jarisch-Herxheimer Reaction After Antibiotic Treatment of Spirochetal Infections: A Review of Recent Cases and Our Understanding of Pathogenesis. Am J Trop Med Hyg. 2017;96(1):46-52. doi:10.4269/ajtmh.16-0434 41. Astrid M. L. Oude Lashof, AnikiRothova, Jack D. Sobel, Markus Ruhnke, Peter G. Pappas, Claudio Viscoli, Haran T. Schlamm, Iwona T. Oborska, John H. Rex, Bart Jan Kullberg, Ocular Manifestations of Candidemia, Clinical Infectious Diseases, Volume 53, Issue 3, 1 August 2011, Pages 262–268, https://doi.org/10.1093/cid/cir355 42. Donahue SP, Greven CM, Zuravleff JJ, et al. Intraocular candidiasis in patients with candidemia. Clinical implications derived from a prospective multicenter study. Ophthalmology. 1994;101(7):1302‐1309. doi:10.1016/s0161-6420(94)31175-4 43. Invernizzi A, Symes R, Miserocchi E, et al. SPECTRAL DOMAIN OPTICAL COHERENCE TOMOGRAPHY FINDINGS IN ENDOGENOUS CANDIDA ENDOPHTHALMITIS AND THEIR CLINICAL RELEVANCE. Retina. 2018;38(5):1011‐1018. doi:10.1097/IAE.0000000000001630 44. Astrid M. L. Oude Lashof, AnikiRothova, Jack D. Sobel, Markus Ruhnke, Peter G. Pappas, Claudio Viscoli, Haran T. Schlamm, Iwona T. Oborska, John H. Rex, Bart Jan Kullberg, Ocular Manifestations of Candidemia, Clinical Infectious Diseases, Volume 53, Issue 3, 1 August 2011, Pages 262–268, https://doi.org/10.1093/cid/cir355 45. Jampol LM, Sung J, Walker JD, et al. Choroidal neovascularization secondary to Candida albicans chorioretinitis. Am J Ophthalmol. 1996;121(6):643-649. doi:10.1016/s0002-9394(14)70630-0 46. McMahon JF, Adam MK, Dunn JP. Optical Coherence Tomography and Management of a Retinal Granuloma in Presumed Ocular Sarcoidosis. Ocul Immunol Inflamm. 2019;27(7):1052‐1056. doi:10.1080/09273948.2018.1497662 47. Russell JF, Russell SR. Whitish Outer Retinal Spots in Rhegmatogenous Retinal Detachment. Ophthalmol Retina. 2018;2(5):389‐395. doi:10.1016/j.oret.2017.10.020 48. Wuthisiri W, Lai YH, Capasso J, et al. Autoimmune retinopathy associated with systemic lupus erythematosus: A diagnostic dilemma. Taiwan J Ophthalmol. 2017;7(3):172-176. doi:10.4103/tjo.tjo_40_17
.jpg)