Introduction
Pediatric glaucoma includes a wide variety of conditions that result in elevated intraocular pressure and optic nerve damage, ranging from primary congenital glaucoma since birth to developmental glaucoma associated with other diseases and acquired glaucoma secondary to multiple causes. Depending on the age of the patient, it presents with particular features and circumstances that need to be taken into account and frequently require surgical intervention.
Glaucoma drainage implant surgery has a definitive role in managing infants and other children with glaucoma refractory to angle surgery and trabeculectomy. GDDs are even preferred nowadays as a primary procedure in selected cases where other surgeries are contraindicated or have very few chances to succeed. Furthermore, it has been found that 20% of pediatric glaucoma patients often require 2 or more surgical procedures for adequate IOP control, and GDDs are frequently used in the second or third surgical attempt.1
Glaucoma drainage devices consist of a tube that shunts the aqueous to an endplate located in the equatorial region. The devices can be classified as valved and non-valved depending upon whether a valve mechanism is present to restrict the outflow when IOP becomes too low.
Valved devices drain aqueous only at certain IOP. The valve opens and fluid is drained into a reservoir where it is absorbed by the surrounding tissue.
Types of valved glaucoma drainage devices
- Ahmed Glaucoma valve
- Krupin disc
- Krupin band
- Optimed
- Joseph Hitchings
Out of these, the Ahmed Glaucoma valve is the most commonly used 2.
AGV: Device Description
AGV was developed by Mateen Ahmed and was approved by the Food and Drug Administration in 1993 3.
Design
It consists of 3 parts :
1) A plate, in medical-grade silicone, polypropylene, or porous polyethylene, depending on the model
2) A drainage tube in medical-grade silicone
3) A valve mechanism in medical-grade silicone
The valve mechanism of AGV consists of two thin silicone elastomer membranes, which are 8 mm long and 7 mm wide sandwiched and stretched between the plates creating a trapezoid-shaped chamber.
The silicone membranes have been pretensioned to open only when the IOP is above 8 mmHg 4. When the pressure is less than that, the valve doesn’t open so that there is no aqueous drainage and hypotony can be prevented.
Mechanics
The Venturi Effect: To reduce internal friction within the valve system, the Ahmed Glaucoma Valve utilizes a specially designed, tapered trapezoidal chamber to create a Venturi effect to help aqueous flow through the device. The inlet velocity of aqueous entering the larger port of the venturi chamber increases significantly as it exits the smaller outlet port of the tapered chamber. In an Ahmed Glaucoma Valve, this increased exit velocity greatly helps in evacuating aqueous from the valve, thereby helping to reduce valve friction.
Non-Obstructive, Self-Regulating Valve Mechanism: The Ahmed Glaucoma Valve has no obstruction in its path of fluid flow. For the flow to be non-obstructive, particles large enough to pass through the lumen of the tube will easily pass through the much larger opening of the VenturiFlow™ chamber. The elastic membranes help to regulate fluid flow at all times, consistently by changing their shape. The tension on these membranes is responsible for reducing hypotony.
Pediatric AGV models
|
TYPE |
MODEL |
PLATE SURFACE AREA(mm2) |
PLATE MATERIAL |
|
SINGLE PLATE |
FP8 |
102 |
SILICONE |
|
S3 |
85 |
POLYPROPYLENE |
|
|
PARS PLANA |
PC8 |
96 |
SILICONE |
|
PS3 |
96 |
POLYPROPYLENE |
Indications
- As a secondary procedure in previously failed trabeculectomy or angle surgery in primary congenital glaucoma.
- As a primary procedure or secondary procedure in a variety of secondary glaucomas such as glaucoma post congenital and developmental cataract surgeries, associated with aniridia, anterior segment dysgenesis, aphakia, and Sturge Weber syndrome.
Pre-operative evaluation
Examination under anesthesia (EUA) at least 2-3 weeks prior to the surgery should be preferably performed unless contraindicated.
EUA includes an assessment of the following:
- IOP
- Central corneal thickness
- Axial length
- Corneal diameter
- Retinoscopy
- Examination of the anterior segment under the microscope:
- Assess the conjunctival health and note down the areas of conjunctival scarring if any.
- Assess the chamber depth and areas of peripheral anterior synechiae (PAS) – tube insertion in AC must be avoided in the areas of extensive PAS.
- Status of the lens- In pseudophakic patients with well-positioned IOL, sulcus fixation of the tube can be planned. In aphakic patients, a vitrectomy may be required at the time of valve implantation to prevent blockage of the tube from the vitreous.
- Examination of the posterior segment:
- Assessment of optic disc.
- The detailed evaluation of the posterior pole and peripheral retina whenever feasible.
- In patients who have undergone a VR surgery earlier, make sure there is no silicone oil in situ as it may later lead to blockage of the tube.
- B-scan: Whenever fundus examination is not feasible owing to media opacities or limited dilatation.
- UBM: It helps study the anterior segment in greater detail especially in presence of hazy cornea. For pseudophakic patients when sulcus fixation is planned, UBM helps determine the IOL position precisely and helps assess whether the ciliary sulcus space is adequate for tube insertion.
Planning the surgery
- Which model to use: Based on the AXL measurement one may select a suitable model. For AXL> 21 mm, FP7 (adult model) may be considered.
- Quadrant of plate insertion: Superotemporal quadrant remains the first choice here. In the case of a second tube or extensive PAS in the superotemporal quadrant, the next best site to insert the plate would be inferotemporal.
- Site of tube insertion: For pseudophakic patients with well-positioned IOL we prefer sulcus fixation. For phakic patients with good chamber depth and good corneal health, insertion in the anterior chamber can be planned. Pars plana insertion can be planned for vitrectomized eyes.
Surgical Technique
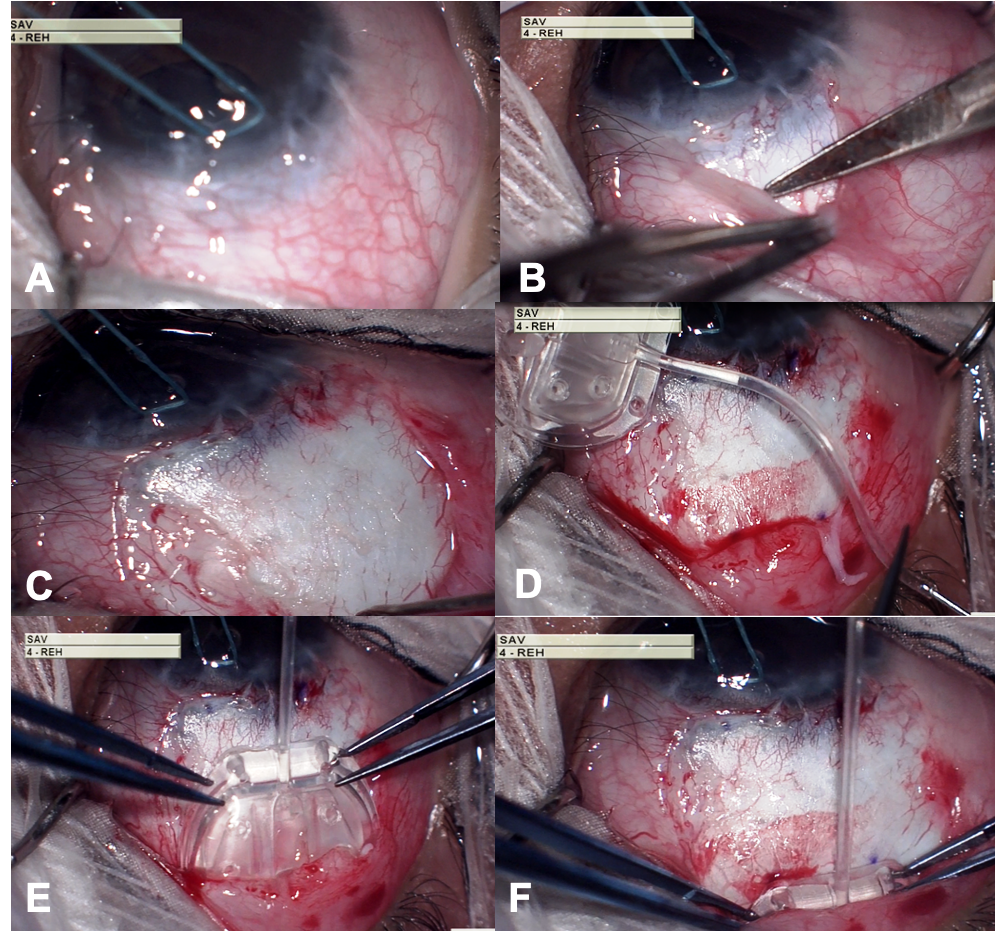
- A corneal traction suture is placed using 8-0 vicryl suture and clamped to surgical drapes to provide adequate exposure to the surgical field.
- A fornix-based incision is made through the conjunctiva and Tenon’s capsule
- A pocket is formed in the superior quadrant between the lateral rectus and superior muscles by blunt dissection of Tenon’s capsule from the episclera. Radial relaxing incisions are made to improve surgical exposure.
- The AGV valve is primed by injecting 1 cc BSS using a 26G cannula. The valve is ready for use once BSS is seen coming out of the valve in a steady stream. The forceful injection must be avoided as it can damage the valve mechanism housed in the scleral plate.
- The implant is introduced into the subconjunctival pocket.
- The proximal end of the plate is positioned 8- 10 mm away from the limbus.
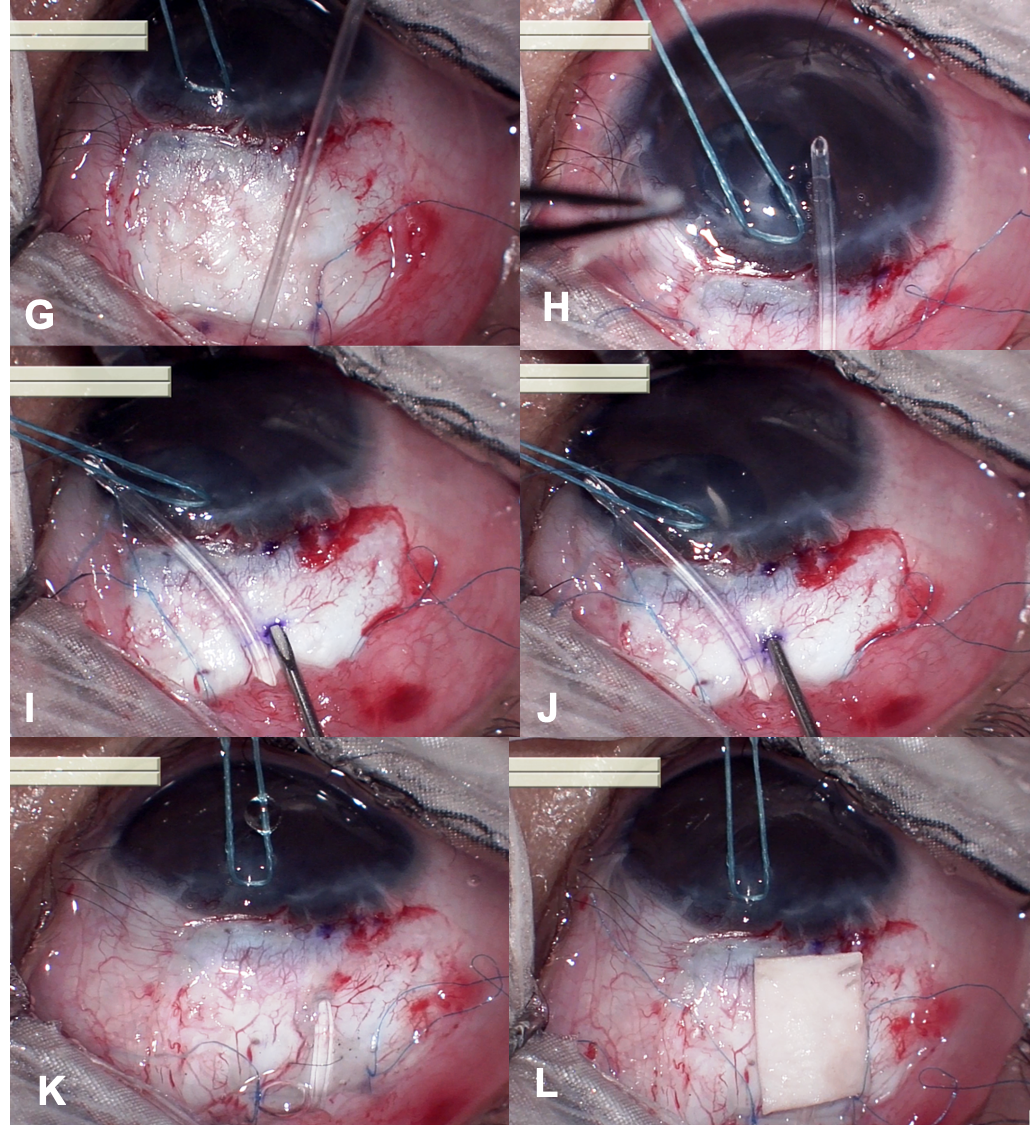
G. The plate is sutured to the limbus using 8-0 prolene suture.
H. The drainage tube is trimmed to permit a 2-3 mm insertion of the tube into the anterior chamber (AC). The tube should be cut with the bevel-up at an angle of 30 0.
I. A paracentesis is performed, and the AC is entered at 1-2 mm away from the limbus with a sharp 23 gauge needle.
J. A needle track is created parallel to the plane of the iris to facilitate the tube entry into the anterior chamber.
K. Tube is inserted through the needle track with the help of a tube inserter instrument and after ensuring that it is well placed, the tube is anchored to the sclera using 10-0 monofilament nylon suture.
L. The exposed drainage tube is then covered with a piece of suitable patch graft material which is sutured into place using 10-0 monofilament nylon suture. We prefer using the Tutoplast® processed human pericardium graft to cover the tube. The Tutoplast® Process is a scientific and technology-based method of virally inactivating, preserving, and sterilizing human tissue. The chances of infection are nullified. It is immunologically inert, hence doesn’t trigger inflammation. It provides excellent cosmesis by not adding any additional bulk to the tissue.
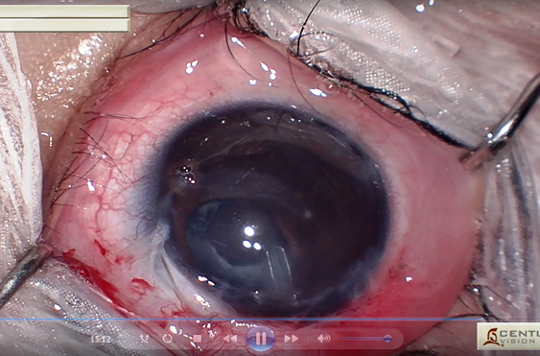
M. The conjunctiva is closed with vicryl 8-0 suture.
Particular challenges regarding glaucoma surgery in infants are as follows:
- Small orbit large globe-One might have to struggle placing the implant due to lack of adequate space.
- Distorted anatomical landmarks-Buphthalmic eyes have stretched limbus and requires more posterior insertion of the plate (10-11 mm from the apparent position of the limbus ) and also a longer scleral tunnel ( initiating it at 5 mm from the apparent position of limbus).
- Low scleral rigidity and thin sclera-Utmost care has to be taken while securing the plate, tube and the graft material to the sclera to avoid perforation.
- Growing eyeball-Pediatric eyes continue to grow. There are higher chances of tube retraction and plate extrusion compared to adults and the parents must be thoroughly counseled regarding the same.
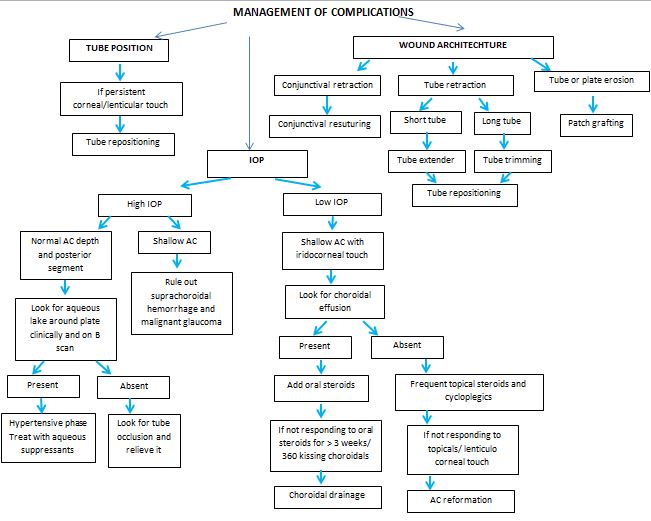
Post-operative evaluation
Post-operative evaluation for pediatric groups unlike the adults is a tricky task except for a few older children who might co-operate for the slit lamp evaluation. For all other cases, a thorough evaluation would be possible only upon examination under anesthesia which is planned as per the anesthetist’s advice usually within 6 weeks.
Until a detailed EUA is possible, assess the following:
- Conjunctival closure is watertight
- The anterior chamber is well-formed
- Fundal glow is unaltered
- Rule out severe hypotony
Once the child is ready to be assessed under anesthesia, a detailed evaluation as mentioned ahead is performed with special attention to the following:
- Tube position– The tube is well positioned and there are no areas of tube endothelial touch.
- Tube patency- Rule out occlusion (blood clot, fibrin, vitreous, iris tissue) at the proximal end as well as across the length of the tube.
- Tube and plate covering- The tube and the plate are well covered with overlying conjunctiva with no areas of thinning or exposure.
- Bleb functioning- Rule out bleb fibrosis (flat bleb with no aqueous lake surrounding the plate) and bleb encystment (tense, elevated bleb with thick overlying Tenon’s capsule) whenever the IOP is high.
Whenever possible, document the tube position and the bleb functioning using UBM
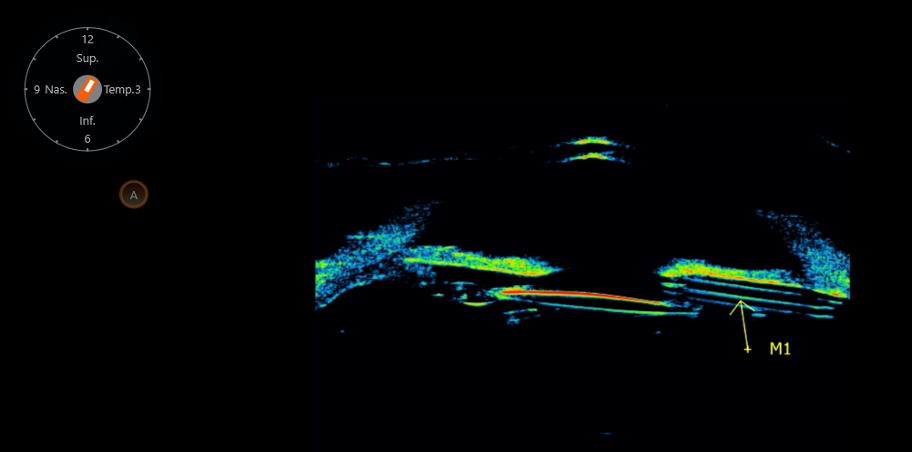
Figure 1: Axial scan showing AGV tube in the ciliary sulcus along the superotemporal quadrant.
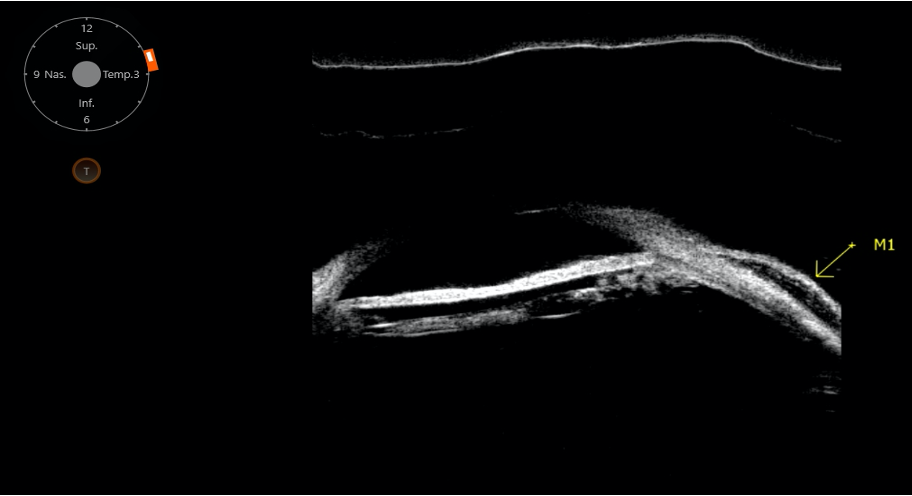
Figure 2: Transverse scan showing good functioning bleb characterized by hypoechoic pattern within the intrableb area and good bleb height.
|
COMPLICATIONS |
|
|
EARLY |
LATE |
|
|
Summary
Traditionally, GDDs were used as the last resort for control of IOP. Over the past few years, the popularity of GDDs has increased and they are being used as a viable alternative to trabeculectomy in most of the secondary and complicated glaucomas commonly seen in the pediatric age group. Considerable volume and experience of the center in dealing not only with skillful glaucoma surgery but also with safe anesthesia are very important. Proper patient selection, preoperative assessment, meticulous technique and anticipation, and timely management of complications are the key to improve the success of GDDs.
Glaucoma secondary to childhood cataract surgery – How we combat this devil
One of the most severe complications after surgery for congenital and developmental cataracts is glaucoma. The incidence of glaucoma after childhood cataract surgery has been reported to be between 8% and 59%.5–15. This variation in the reporting of the incidence can be attributed to the differences in the study population, criteria for the diagnosis of glaucoma, and length of the follow-up period.
The most common type of glaucoma to develop following congenital cataract surgery is open-angle glaucoma16, 17. The risk factors include age at surgery, preexisting ocular abnormalities, type of cataract, and the effect of lens particles, lens proteins, inflammatory cells, and retained lens material. In addition, microcornea, secondary surgery, chronic postoperative inflammation, the type of lensectomy procedure or instrumentation, pupillary block, and the duration of postoperative observation have been found to influence the likelihood of glaucoma after pediatric cataract surgery 16. Undergoing lensectomy at a young age, especially in the first year of life, maybe a risk factor for the development of glaucoma 17. It has been suggested that the immaturity of the developing infant’s angle leads to increased susceptibility to secondary surgical trauma 17.
In the majority of the cases, glaucoma appears to be the consequence of surgery rather than the cataract itself. Hence it becomes extremely important to discuss this possibility with the parents. During our pre-operative counseling session with the parents and family, we discuss at length the chances of developing glaucoma in the post-operative period and also years later. The parents need to understand that cataract surgery is not a one-stop solution to the existing problem and apart from many other challenges, glaucoma is one particular challenge they must be ready to face as long as their child grows up and probably also beyond that.
The next crucial aspect in this battle against glaucoma is its timely diagnosis and management. Unless the examining ophthalmologist is specifically looking for glaucoma, the diagnosis is likely to be missed. Here comes the role of EUA. A comprehensive examination that includes refraction, measurement of IOP, corneal diameter, axial length, assessment of the anterior, and posterior segments is essential to arrive at a diagnosis and this explains why subjecting the child to that extra shot of anesthetic agent is inevitable.
Whenever feasible, we try to go a step ahead in our examination and perform UBM. UBM enables us to precisely document important details such as the presence of anterior and posterior synechiae, study the anterior chamber angle, and most importantly the IOL position in pseudophakic patients. An IOL imaged to be lying well inside the bag or a well-captured optic offers a big sigh of relief against one with an asymmetric sulcus fixation which alarms us to be extra cautious as it has the potential to trigger uveal inflammation and subsequent glaucoma. Sometimes, the IOL can be the main culprit and in absence of an imaging technique such as this, we are likely to miss the real cause. Thus we strongly recommend making UBM an integral part of your armamentarium to combat glaucoma.
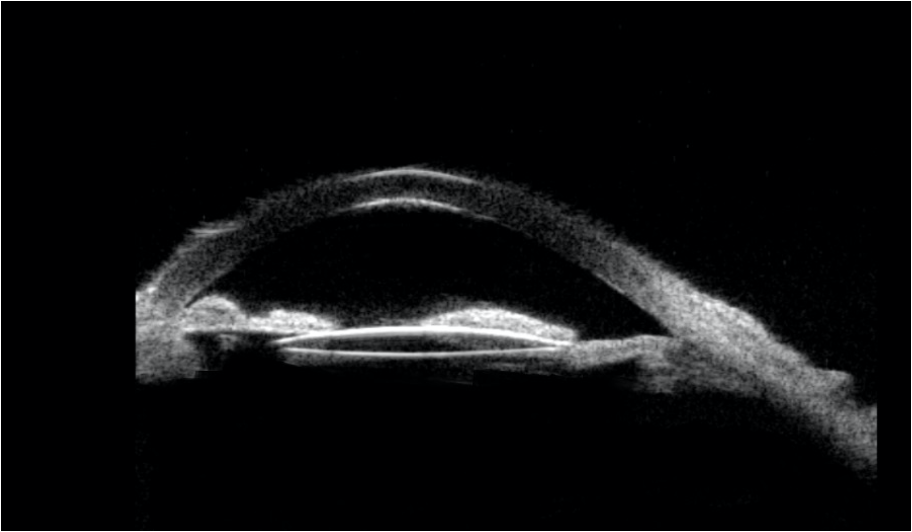
Figure 3: Post-operative UBM scan of a 4-month-old child operated for bilateral congenital cataract showing the IOL in the sulcus with peripheral anterior synechiae and extensive posterior synechiae.
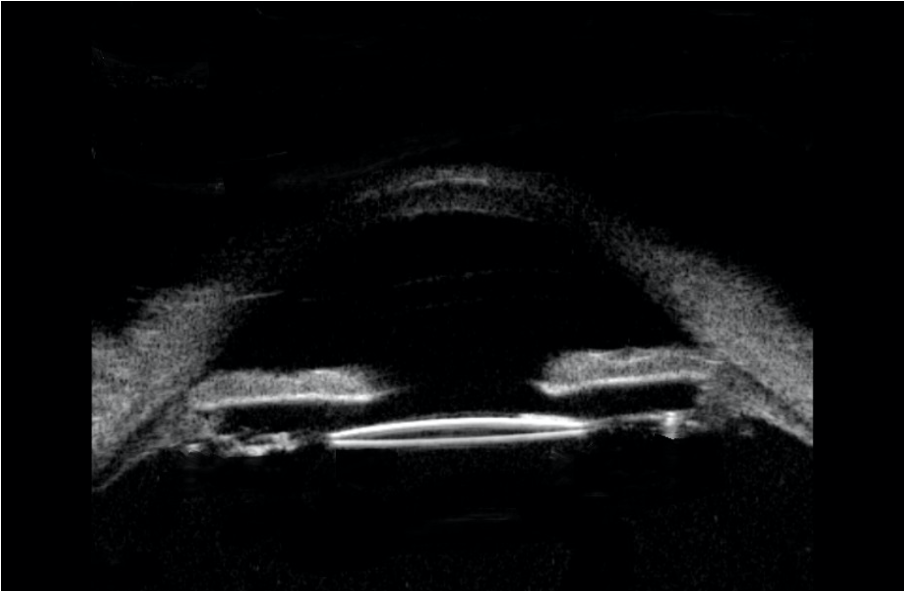
Figure 4: Post-operative UBM scan of a 9-month-old child operated for unilateral congenital cataract showing well-positioned IOL in the bag with adequate space between the iris and the IOL and wide-open angles.
After a detailed EUA, we label the eye as safe, at-risk, or established glaucoma and plan our management accordingly. For the eyes tagged as safe, 4 to 6 monthly assessment is scheduled. The eyes at risk need to be scrutinized more often. For eyes with established disease, the first step is to start medical therapy and assess the response within 6 weeks. In our experience, the role of medical therapy in managing glaucoma secondary to pediatric cataract surgery is limited, and sooner or later surgical intervention is warranted.
Regarding the choice of surgery, conventional trabeculectomy remains the first choice and GDD is reserved for eyes where trabeculectomy fails. While both are likely to fail, in our experience GDD offers an edge over trabeculectomy in terms of longevity if the device is well placed. For glaucoma surgery, it is said that the first chance is the best chance and we would not hesitate to offer GDD as the primary option rather than wait for the trabeculectomy to fail and then put in the tube. Besides, pseudophakia gives us the freedom to insert the tube in the ciliary sulcus and safeguard the cornea from tube related decompensation. For eyes where IOL is the prime culprit, along with glaucoma surgery we would like to explore the possibility of getting rid of the triggering factor as well. Traditionally IOL explantation has not been favored by many owing to the excessive surgical manipulation involved. However, with the refinement in the surgical technique and surgical expertise, IOL explantation is no more as traumatic as it used to be earlier and should be definitely considered whenever warranted.
Despite the advancements in surgical technology and instrumentation, secondary glaucoma remains a major challenge to pediatric ophthalmologists. All the surgeons dealing with pediatric cataracts have to be ready to face the viciousness of glaucoma. It is the most commonly missed, neglected or mismanaged entity after pediatric cataract surgery. Sound counseling of the parents along with timely intervention can save vision for a lifetime. And the joy of these patients when they walk into your OPD for follow up when they grow up is something that is inexpressible and beyond words!
References
- Hill R, Ohanesian R, Voskanyan L, Malayan A. The Armenian Eye Care Project: surgical outcomes of complicated paediatric glaucoma.Br J Ophthalmol. 2003; 87(6):673-676. doi:10.1136/bjo.87.6.673
- E. O’Malley Schotthoefer, T. L. Yanovitch, and S. F. Freedman, “Aqueous drainage device surgery in refractory pediatric glau-comas: I. Long-term outcomes,”Journal of AAPOS, vol.12, no.1, pp.33–39, 2008.
- Coleman AL, Hill R, Wilson MR, et al. Initial clinical experience with the Ahmed glaucoma valve implant. Am J Ophthalmol.1995; 120(1):23–31.
- B. A. Francis, A. Cortes, J. Chen, and J. A. Alvarado, “Char-acteristics of glaucoma drainage implants during dynamic andsteady- state flow conditions,”Ophthalmology,vol.105,no.9,pp.1708–1714, 1998.
- Egbert JE, Wright MM, Dahlhauser KF, Keithahn MA, Letson RD, Summers CG. A prospective study of ocular hypertension and glaucoma after pediatric cataract surgery. Ophthalmology. 1995; 102:1098 –1101.
- Keech RV, Tongue AC, Scott WE. Complications after surgery for congenital and infantile cataracts. Am J Ophthalmol. 1989;108: 136 –141.
- Robb RM, Petersen RA. Outcome of treatment for bilateral congenital cataracts. Ophthalmic Surg. 1992;23:650 – 656.
- Mills MD, Robb RM. Glaucoma following childhood cataract surgery. J Pediatr Ophthalmol Strabismus. 1994;31:355–360.
- Miyahara S, Amino K, Tanihara H. Glaucoma secondary to pars plana lensectomy for congenital cataract. Graefes Arch Clin Exp Ophthalmol. 2002;240:176 –179.
- Rabiah PK. Frequency and predictors of glaucoma after pediatric cataract surgery. Am J Ophthalmol. 2004;137:30 –37.
- Parks MM, Johnson DA, Reed GW. Long-term visual results and complications in children with aphakia: a function of cataract type. Ophthalmology. 1993;100:826 – 840.
- Johnson CP, Keech RV. Prevalence of glaucoma after surgery for PHPV and infantile cataracts. J Pediatr Ophthalmol Strabismus. 1996;33:14 –17.
- Ariturk N, Oge I, Mohajery F, Erkan D, Turkoglu S. Secondary glaucoma after congenital cataract surgery. Int Ophthalmol. 1998; 22:175–180.
- Simon JW, Mehta N, Simmons ST, Catalano RA, Lininger LL. Glaucoma after pediatric lensectomy/vitrectomy. Ophthalmology. 1991;98:670 – 674.
- Chen TC, Bhatia LS, Halpern EF, Walton DS. Risk factors for the development of aphakic glaucoma after congenital cataract surgery. Trans Am Ophthalmol Soc. 2006;104:241–251.
- Mandal AK, Netland PA. Glaucoma in aphakia and pseudophakia after congenital cataract surgery. Indian J Ophthalmol 2004; 52:185–198. An interesting article describing glaucoma associated with aphakia and pseudophakia after congenital cataract surgery.
- Chen TC, Walton DS, Bhatia LS. Aphakic glaucoma after congenital cataract surgery. Arch Ophthalmol 2004; 122:1819–1825.