Introduction:
Sarcoidosis is a chronic multisystemic granulomatous disorder caused by an exaggerated cellular immune response to a variety of self or non-self-antigens in a genetically predisposed individual resulting in non-caseating granulomas. Ocular involvement occurs in 25-60% of systemic sarcoidosis at some point of time1. In this article, we look at the emerging literature on epidemiology, pathogenesis, clinical profile, and management in ocular sarcoidosis.
Epidemiology:
Sarcoidosis has an overall incidence of 6-10 per 100,000. Although sarcoidosis occurs worldwide, it is predominant in certain ethnic and racial groups like the Afro-Caribbean, Scandinavian, and Irish populations2-4. Highest incidence of ocular sarcoidosis is in the 20-40-year age group. Some studies show two peaks of incidence for ocular sarcoidosis the first at ages 20-30 and the second at ages 50-605. There is also a higher incidence of ocular involvement in women4. The reported incidence of ocular sarcoidosis varies according to the geography, patient population, diagnostic criteria employed, and referral patterns of the reporting ophthalmologists. Earlier considered a disease of the developed world, in recent years, ocular sarcoidosis has increasingly been recognized from developing countries like India, Singapore, Thailand, Taiwan, Malaysia, Kuwait, Lebanon, and Turkey1,6,7. This may be due to the increasing awareness of this disease and the availability of improved diagnostic modalities like computed tomography and trans-bronchial lymph node biopsies. Ocular involvement in ocular sarcoidosis can be as high as 90% and ocular involvement can precede systemic manifestations of sarcoidosis1.
Pathogenesis:
Sarcoidosis is caused by antigenic stimulation. In genetically susceptible individuals, antigenic stimulation activates a cascade of immune reaction leading to sarcoidosis. Disease-modifying genes and the interplay of regulatory immune response mediated by regulatory cells and cytokines determine the severity of the disease. Presentation of antigen by the antigen-presenting cells leads to activation of CD4+ cells resulting in the release of cytokines and recruitment of immunocompetent cells and, compartmentalization at the site of resulting in the production of granuloma8. Sarcoidosis is a multisystem granulomatous disease of unknown etiology. Various factors implicated in the pathogenesis of sarcoidosis have been summarized in table 1 8-31.
|
Mechanism |
Description |
|
|
1 |
Dysregulation of the IL-23/IL-17 Pathway and Role of T Helper Cells |
IL23R pleomorphism has been reported to be associated with increased susceptibility to sarcoidosis, more so ocular sarcoidosis |
|
2 |
Microorganisms |
Mycobacteria, Propionibacterium, Atopobium, Fusobacterium, Hepatitis C, Barr virus, herpes virus, and helicobacter pylori |
|
3 |
Drug-induced sarcoidosis-like reaction |
Ipilimumab, Nivolumab, Pembrolizumab, highly active antiretroviral therapy, Interferon-alpha, Interferon-beta, Etanercept, Adalimumab, and Infliximab BRAF inhibitors.[21] Vemurafenib.[25] |
|
4 |
Genetic predisposition |
Monozygotic twins are more often concordant for the disease than dizygotic twins Familial clustering occurs in in 5-16% of the patients ACCESS study has revealed a familial risk of sarcoidosis to have an estimated odds ratio of 5.8 for siblings and 3.8 for first degree relative parents. [29] HLA-DRB1 and HLA-DQB1 have been linked to sarcoidosis.[30,31] Non-HLA genes associated with sarcoidosis are BTNL2, NOD2, NOTCH 4, RAS23 ANXA11. Polymorphisms in genes encoding cytokines like transforming growth factor-beta, interleukin-8, and tumor necrosis factor-alpha and Toll-like receptor-4 have also been reported [16,17]. [38,39] Genetic analyses also established that IL23R polymorphisms were associated with the susceptibility to sarcoidosis, especially ocular sarcoidosis.[11] RAB23 and ANXA11 genes were associated with an increased risk of sarcoidosis- associated uveitis.[40] |
|
5 |
Vascular Endothelial Damage |
Uveitis in sarcoidosis patients has been hypothesized to be secondary to vascular dysfunction. ocular involvement can be a biomarker of vascular damage, and choroidal involvement can predict an increased risk of cardiac disease |
Histopathology:
The classic sarcoid granuloma is made up of modified macrophages or epithelioid cells surrounded by a rim of lymphocytes and fibroblasts. Necrosis or caseation, which is usually seen with tuberculosis and fungal infections, is absent in sarcoid granulomas. Occasionally, small foci of fibrinoid necrosis may be present in sarcoidosis. Immunohistochemical studies demonstrate the presence of CD4+Tcells admixed with the epithelioid cells in the center of the cellular infiltrate. The compartmentalization of CD4+Tcells at sites of the disease leads to a markedly high CD4/CD8 ratio of more than 10. An elevated bronchoalveolar lavage (BAL) CD4/CD8 T-cell ratio greater than 3.5 predicts the diagnosis of sarcoidosis with 94% specificity and 53% sensitivity. This is also associated with a favorable prognosis. Histologic observations suggested that granuloma and fibrosis were associated with the activation of Th1 cells, monocytes, and macrophages, as well as the overproduction of IFN-γ, IL-2, TNF-α, IL-15, and CCL2032-34.
Clinical Manifestations:
Patients may be asymptomatic or present with blurred vision, floaters, redness, or discomfort. Ocular involvement is usually a chronic disease and has an insidious onset. It can involve the lacrimal glands, orbit, eyelids, conjunctiva, uveal tract, and the optic nerve (Figure 1-3). The ocular findings in sarcoidosis are summarized in table 21-4, 6,7
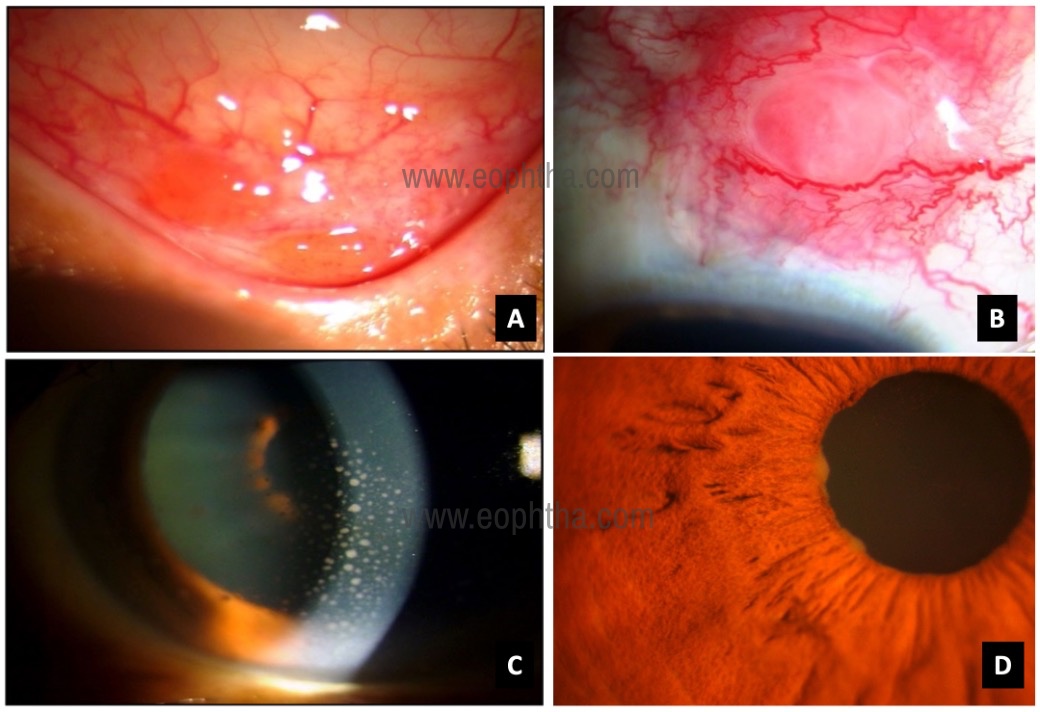
patient with pulmonary sarcoid B: Scleral nodule C: Keratic precipitates
in eyes granulomatous anterior uveitis D: large Koeppe nodules
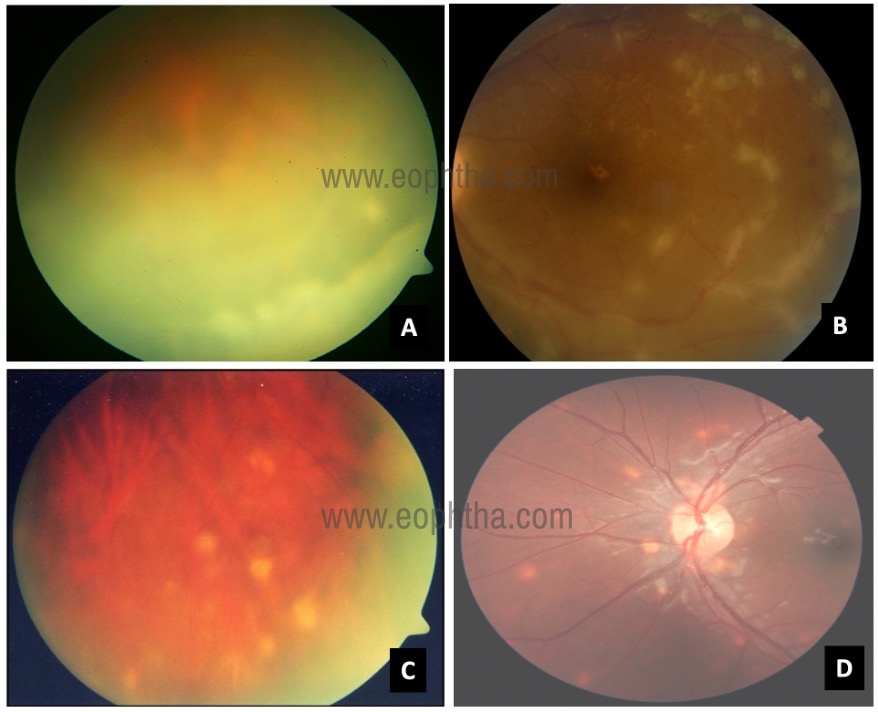
B: perivasculitis C: Peripheral chorioretinal punched out scars
D: multiple choroidal granuloma
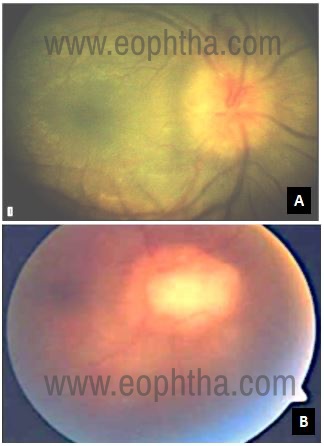
A: disc edema B: Optic nerve granuloma
|
Eye lid |
Granuloma, lagophthalmos (if associated with facial palsy) |
|
Lacrimal gland |
Keratoconjunctivitis sicca |
|
Sclera |
Scleritis/episcleritis |
|
Conjunctiva |
Conjunctival granuloma and nodules |
|
Cornea |
Band shape keratopathy |
|
Pupillary abnormality |
Horner syndrome, tonic pupils, and Argyll Robertson pupils |
|
Anterior uveitis |
Nongranulomatous anterior uveitis Mutton Fat keratic precipitates Iris and pupillary nodules Increase in IOP Tent shaped peripheral anterior synechiae Nodules in trabecular meshwork (Berlin nodules) |
|
Lens |
cataract |
|
Intermediate uveitis |
Snowballs/strings of pearls vitreous opacities Multifocal peripheral chorioretinal lesions (active and atrophic) |
|
Retinal and choroidal lesions |
Nodular and /or segmental periphlebitis (with or without candle wax exudate) Choroidal granuloma Hemorrhagic retinopathy with a branch or central retinal venous occlusions |
|
Optic nerve |
Optic disc nodules/ granuloma /optociliary shunts/ dilated collateral veins on the optic nerve head |
|
Neurological manifestations |
Cranial nerve palsies Encephalopathy Chiasmal syndromes Motility disorders Optic atrophy either due to direct sarcoid tissue infiltration or compression by cerebral mass. |
|
Nasolacrimal drainage system |
Nasolacrimal duct obstruction |
Orbits, lacrimal gland and lacrimal sac:
Orbital involvement is usually primary, but rarely, sarcoidosis primarily affecting the paranasal sinuses can invade the orbit36. Sarcoidosis coexisting with Graves’ disease has been reported2,3. Sarcoidosis can involve the orbital adipose tissue, extraocular muscles, and optic nerve sheath, mimicking inflammatory disorders affecting the orbit36,37.
The lacrimal gland is the most common organ in the orbit to be affected. Histopathologic studies of biopsy-proven orbital sarcoidosis revealed the main lacrimal gland involvement in 42-63% cases38-40. Usually, patients with lacrimal gland involvement are asymptomatic, but if there is extensive involvement, it can lead to severe keratoconjunctivitis sicca. An enlarged lacrimal can present as a palpable mass, ptosis, diplopia, or proptosis (Figure 4). Nasolacrimal duct obstruction can occur as a consequence of granulomatous inflammation and lead to watery eyes41.
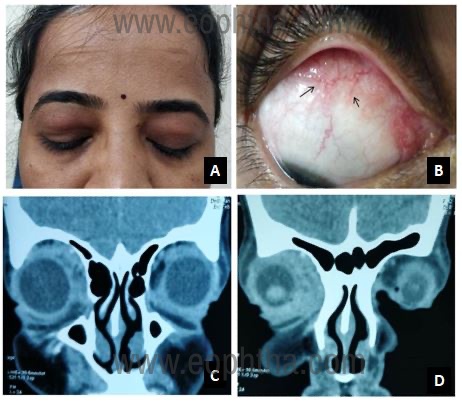
superonasally to the right upper eyelid (a), conjunctival
granulomas (black arrows) (b), and CT orbits
showing ill-defined, heterogenous lesion in the superior
and nasal orbit near the lacrimal sac regions (c,d)
Eyelids and Conjunctiva:
Conjunctival granulomas are millet shaped to large cream to brown nodular lesions, which can occur in sarcoidosis6,42. They are usually asymptomatic. Large conjunctival granulomas can rarely cause diplopia. conjunctival granulomas generally respond to topical steroids. Eyelid granulomas are also seen in sarcoidosis43. Patients can also present with fullness of the lids.
Sclera
Sarcoidosis can rarely present as scleritis and episcleritis. It may present as anterior diffuse, anterior nodular, or posterior scleritis44-47.
Optic Nerve Involvement
Optic nerve involvement is very rare in sarcoidosis. The optic nerve can be affected by various mechanisms which include inflammation of the optic nerve, compression or infiltration of the anterior visual pathway, secondary to retinal ischemia and choroidal inflammation, glaucoma, granuloma, or consequence of hydrocephalus. Patients with sarcoidosis may develop a unilateral or, occasionally, a bilateral retrobulbar optic neuropathy44,49.
Pupillary involvement sarcoidosis
Patients with sarcoidosis can have the involvement of the pupil. Pupillary manifestations can be in the form of Horner syndrome, tonic pupils, or Argyll Robertson pupils50-55.
Uveitis:
It is typically chronic bilateral uveitis and characterized by a granulomatous inflammatory reaction in the anterior segment which includes mutton fat keratic precipitates, iris nodules, trabecular meshwork nodules, and tent-shaped peripheral anterior synechiae; snowball and/or “string of pearls” opacities in the vitreous; and in the posterior segment, nodular periphlebitis, multiple peripheral active, or atrophic chorioretinal lesions, optic disc nodules/granuloma, and choroidal nodules56. In acute onset sarcoidosis, non-granulomatous anterior uveitis may be seen.
International workshop on ocular sarcoidosis (IWOS) criteria laid down criteria for the diagnosis of ocular sarcoidosis in 200957. Validation studies of IWOS criteria revealed that the criteria except liver enzymes had high diagnostic value only in Japan, while a study with an international cohort involving 14 centers across the world had many limitations58,59. Also, the international validation study revealed a low sensitivity of the category ‘possible ocular sarcoidosis’. The criteria of a negative tuberculin test in a BCG vaccinated patient or having had a positive purified protein derivative (PPD) skin test done previously was not applicable in all countries, as the BCG vaccination was not performed in many countries and many patients had not received tuberculin test previously. The international study also suggested the need for more than five systemic investigations in suspected patients with ocular sarcoidosis59.
To overcome these limitations, the IWOS criteria was revised recently in 2019 which arrived at the following consensus:(1) Other causes of granulomatous uveitis must be ruled out (2) seven intraocular clinical signs suggestive of ocular sarcoidosis (3) eight results of systemic investigations in suspected ocular sarcoidosis, and (4) three categories of diagnostic criteria depending on biopsy results and combination of intraocular signs and results of systemic investigations60.(Table 3)
|
I Other causes of granulomatous uveitis must be ruled out |
|
II. Intraocular signs suggestive of ocular sarcoidosis |
|
1. Mutton-fat keratic precipitates (large or small) and/or iris nodules at pupillary margin (Koeppe) or in stroma (Busacca) |
|
2. Trabecular meshwork nodules and/or tent-shaped peripheral anterior synechia |
|
3. Snowballs/strings of pearls vitreous opacities |
|
4. Multiple chorioretinal peripheral lesions (active and/or atrophic) |
|
5. Nodular and/or segmental periphlebitis (candle-wax drippings) and/or macroaneurysm in an inflamed eye |
|
6. Optic-disc nodule(s)/granuloma(s) and/or solitary choroidal nodule |
|
7. Bilaterality (assessed by ophthalmological examination including ocular imaging showing subclinical inflammation) |
|
III. Systemic investigations result in suspected ocular sarcoidosis |
|
1. Bilateral hilar lymphadenopathy by chest X-ray and/or chest computed CT scan |
|
2. Negative tuberculin test in a BCG-vaccinated patient or interferon-gamma releasing assays |
|
3. Elevated serum angiotensin converting-enzyme |
|
4. Elevated serum lysozyme |
|
5. Elevated CD4/CD8 ratio (>3.5) in bronchoalveolar lavage fluid |
|
6. Abnormal accumulation of 67-gallium scintigraphy or 18F-fluorodesoxyglucose positron emission tomography imaging |
|
7. Lymphopenia |
|
8. Parenchymal lung changes consistent with sarcoidosis, as determined by pneumologists or radiologists |
|
Diagnostic criteria of ocular sarcoidosis |
|
• Definite ocular sarcoidosis: diagnosis supported by biopsy with compatible uveitis |
|
• Presumed ocular sarcoidosis: diagnosis not supported by biopsy, but bilateral hilar lymphadenopathy present with two intraocular signs |
|
• Probable ocular sarcoidosis: diagnosis not supported by biopsy and bilateral hilar lymphadenopathy absent, but three intraocular signs and two systemic investigations selected from two to eight are present |
Complications:
The most frequent complications include cystoid macular edema (76%), cataract (49%), glaucoma (36%), retinal ischemia (16%), and neovascularization (11%)61. Corneal band-shaped keratopathy occurs in longstanding cases of uveitis. Reports of inflammatory choroidal neovascular membranes (CNVM) have been described in sarcoidosis. Rarely, can CNVM be an initial manifestation of ocular sarcoidosis.
Investigations/Laboratory Tests
Laboratory, radiological and ocular investigations play a pivotal role in narrowing down to diagnosis of ocular sarcoidosis (figure 5-9). The investigative tools used in the diagnosis of ocular sarcoidosis have been summarized in table 4.
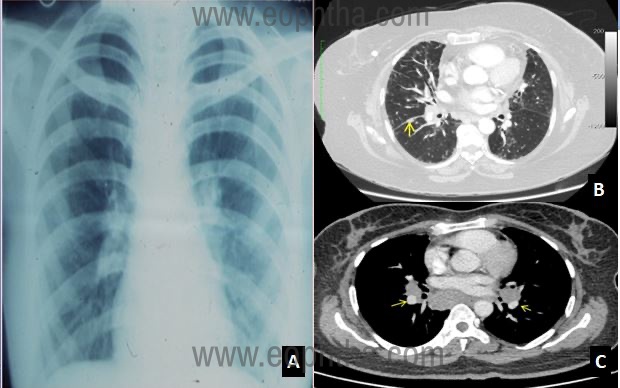
in a patient with sarcoidosis B: Coronal high resolution computerized
tomography (HRCT) scan showing fissural nodules in ocular
sarcoidosis (yellow arrow) C: Coronal HRCT scans showing symmetrical
hilar lymph node enlargement in ocular sarcoidosis (yellow arrows)
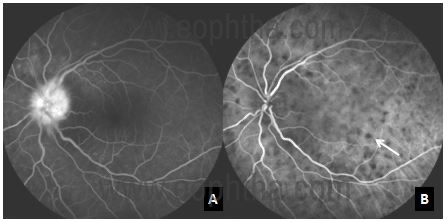
a eyes with posterior uveitis with multiple sarcoid choroidal
granulomas B: Indocyanine green angiography showing multiple
hypocyanascent choroidal granulomas which is appreciated better than
Fluorescein angiography
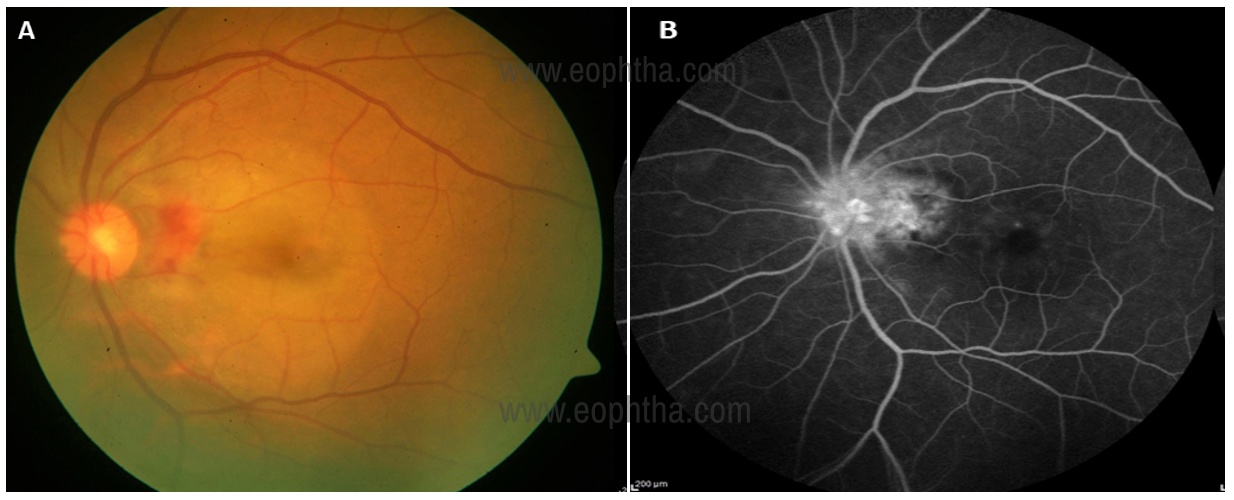
membrane (CNVM) adjacent to the disc with subretinal fluid at
the macula, B: fundus fluorescein angiogram showing hot disc
with dye leakage from the CNVM
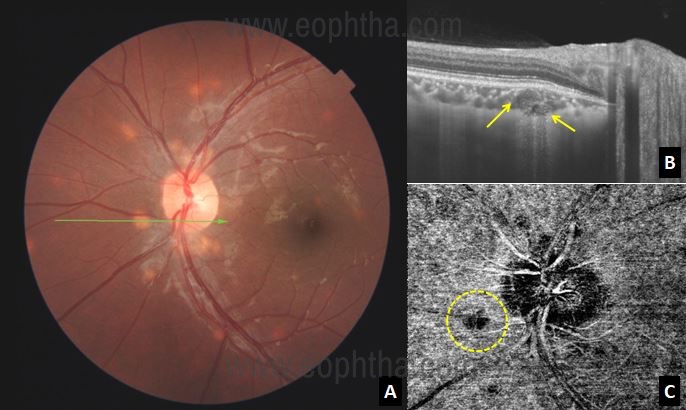
showing multiple choroidal granuloma B: Optical
coherence tomography over the lesion shows homogenous
lesion at the level of choroid with increased transmission suggestive
of choroidal granuloma
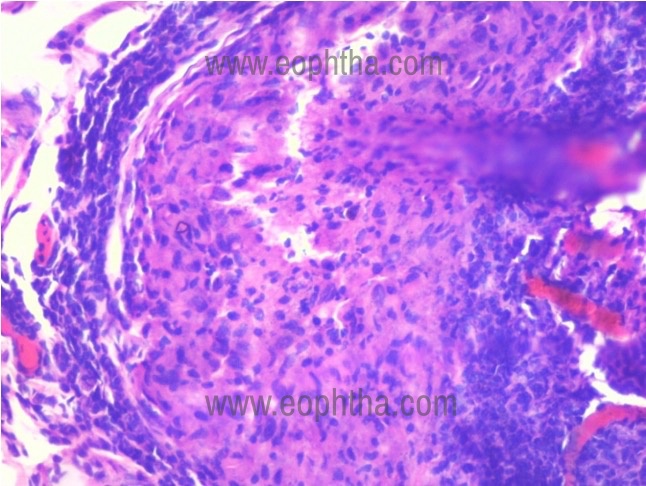
inflammation comprising of lymphocytes and macrophages
|
Investigations |
|
|
A |
Laboratory investigations |
|
Tuberculin Test |
|
|
Serum Angiotensin-Converting Enzyme and Lysozyme Level |
|
|
Abnormal Liver Enzyme Tests |
|
|
Lymphopenia |
|
|
Serum Interleukin 2 Receptor |
|
|
Krebs von den Lungen-6 |
|
|
Serum calcium level |
|
|
Micro Ribonucleic Acid |
|
|
B |
Radiological investigations |
|
Chest radiograph |
|
|
High-resolution computerized tomography scan |
|
|
Gallium Citrate Scanning |
|
|
18F-Fluorodesoxyglucose Positron Emission Tomography |
|
|
C |
Biopsy |
|
Transbronchial lymph node biopsy |
|
|
Conjunctival granuloma biopsy |
|
|
D |
Bronchoalveolar Lavage Fluid analysis |
|
E |
Ocular investigations |
|
Fluorescein angiography |
|
|
Indocyanine green angiography |
|
|
Optical Coherence Tomography |
|
|
Optical Coherence Tomography Angiography |
|
|
Aqueous CD4/CD8 Ratio |
|
|
Vitreous CD4+/CD8+ Ratio |
Differential Diagnosis of Ocular Sarcoidosis:
In addition to the clinical signs and laboratory tests indicative of sarcoidosis, exclusion of other entities that can be mistaken for sarcoidosis is equally important for the diagnosis of sarcoidosis. Granulomatous uveitis may be seen in tuberculosis, syphilis, Vogt-Koyanagi-Harada disease, toxoplasmosis, herpetic uveitis, and multiple sclerosis62. Chorioretinal granulomas may also be seen in tuberculosis, syphilis, Vogt-Koyanagi-Harada disease, birdshot retinochoroidopathy, and primary intraocular lymphoma.
Treatment:
There are no standardized therapies for ocular sarcoidosis-associated uveitis. The mainstay of treatment is corticosteroids and is given via topical, periocular, or systemic routes.
Topical Corticosteroids:
When the inflammation is confined to the anterior segment, topical steroids can be used.
Local Corticosteroids:
Periocular injections or intravitreal injections of steroids may be useful in unilateral disease in the absence of a systemic disease. Intravitreal steroids or a sustained steroid drug delivery device like dexamethasone implants (Ozurdex, FDA approval—2010), is very useful in treating cystoid macular edema63.A fluocinolone acetonide implant called Retisert (Bausch & Lomb) was approved by the FDA in 2005, for the treatment of chronic non-infectious uveitis affecting the posterior segment. However, it is not used nowadays, as it is associated with serious adverse events like glaucoma (glaucoma with 34% of filtering surgery at 4 years) and cataract(s)64.
Systemic Steroids:
Systemic corticosteroids should be used in cases with optic neuropathy, macular edema with visual acuity <20/200, or occlusive retinal vasculitis with retinal ischemia. In addition, systemic therapy is used in patients who are resistant, and/or intolerant to local treatment, and/or have active systemic disease requiring treatment. In the acute phase, pulse intravenous corticosteroids are recommended in case of severe disease. Mean initial dose of prednisone/prednisolone is 0.5-1.0 mg/kg/day, to a maximum dose of 80 mg/day. Oral prednisolone has to be slowly tapered over a period of 3-6 months.
Immunosuppressive Agents:
For corticosteroid resistant or steroid intolerant cases, steroid sparing immunosuppressive agents like methotrexate, mycophenolate mofetil, azathioprine, and cyclosporine are effective65,66 Study by Baughman et al.65 reported that both methotrexate and azathioprine were efficient in the treatment of ocular sarcoidosis, but discontinuation rate was higher with azathioprine (methotrexate- 3.8% and azathioprine – 19.1% ).
Anti-TNF-α Treatment
Anti TNF-α![]() agents have been used for the treatment of refractory cases of ocular sarcoidosis.
agents have been used for the treatment of refractory cases of ocular sarcoidosis.
Adalimumab (ADA) has been approved by the FDA for the treatment of non-infectious anterior uveitis in patients with inadequate response to corticosteroids, corticosteroid dependence, or contraindication for use of corticosteroids based on VISUAL 1 and 2 trials67,68.
Other Biologics
Reports of treatment with rituximab in aggressive cases of sarcoid have been reported in literature69.Silpa-Archa, et al. reported that one case out of 17 cases studied had multidrug-resistant ocular sarcoidosis improved with tocilizumab therapy with no major side effects70. Janus kinase inhibitors like tofacitinib or ruxolitinib have been used in patients with systemic sarcoidosis without ocular involvement and has been found to be effective71-73.The treatment algorithm has been outlined in Figure 10 74.
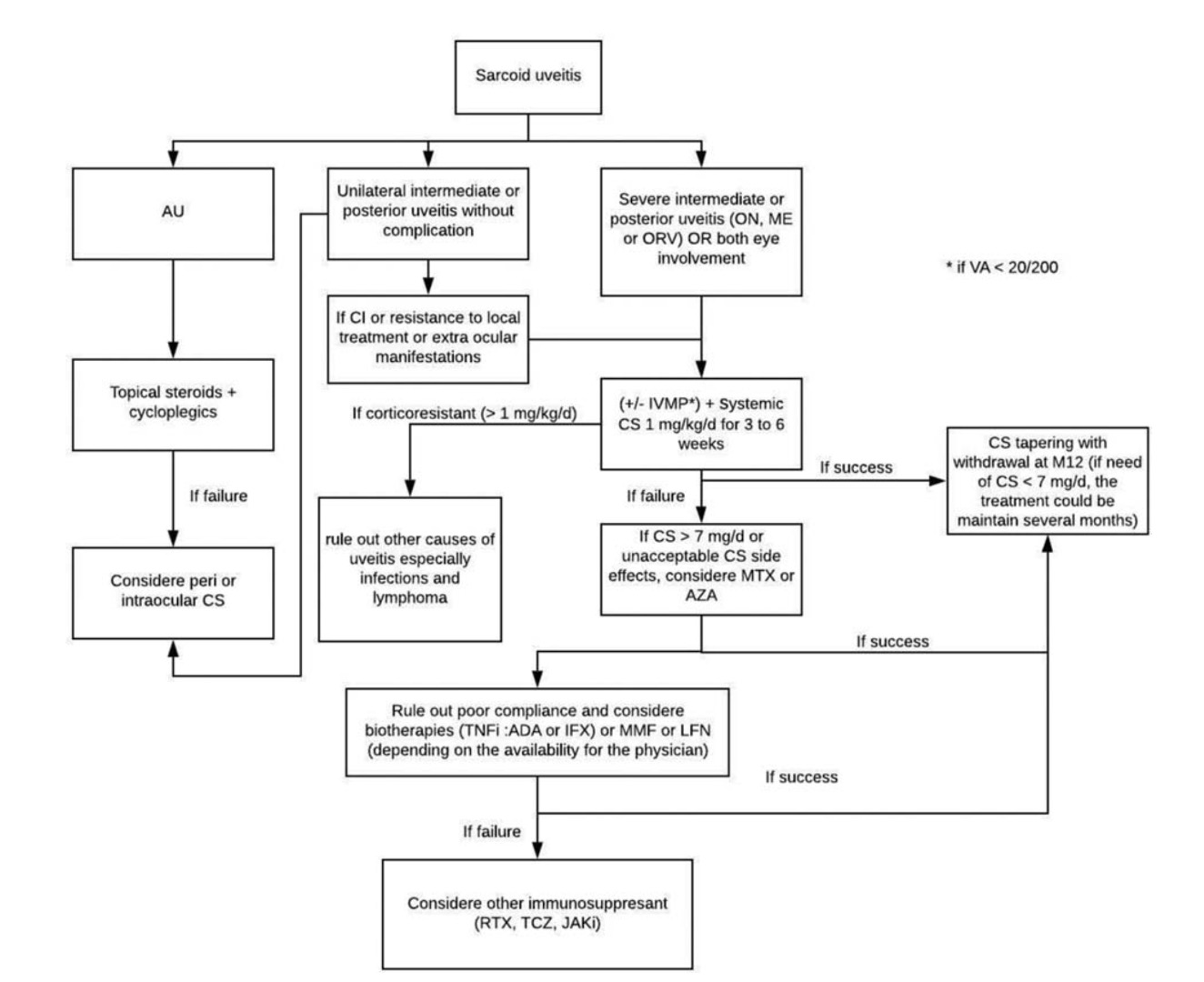
Figure 10: Algorithm for the treatment of patients with ocular sarcoidosis. The algorithm has been adopted from Seve P, et al.74 ADA: adalimumab; AU: anterior uveitis; AZA: azathioprine; CI: contraindication; CS: corticosteroids; IFX: infliximab; IVMP: intravenous methylprednisolone pulse; JAKi: Janus kinase inhibitor; LFN: leflunomide; ME: macular edema; MMF: mycophenolate mofetil; MTX: methotrexate; ON optic neuritis; ORV: occlusive retinal vasculitis; RTX: rituximab; TCZ: tocilizumab; TNFi: tumor necrosis factor inhibitor; VA: visual acuity
Treatment of Complications
Neovascularization may regress with systemic anti-inflammatory treatment, anti-vascular endothelial growth factors, and in some cases may require laser treatment. Associated complications like cataract and glaucoma may need to be treated appropriately to prevent visual morbidity.
Conclusion:
Sarcoidosis has been increasingly diagnosed in recent years, due to increased awareness of the disease and better availability of diagnostic modalities. Ocular evaluations contribute in making a diagnosis of systemic sarcoid. Early diagnosis and appropriate, adequate treatment reduces the visual morbidity in ocular sarcoidosis.
References:
- Babu K, Kini R, Mehta R, Abraham MP, Subbakrishna DK, Murthy KR. Clinical profile of ocular sarcoidosis in a South Indian patient population. Ocul Immunol Inflamm. 2010;18(5):362.
- Jones N, Mochizuki M. Sarcoidosis: epidemiology and clinical features.Ocul Immunol Inflamm. 2010;18(2):72-79.
- Rothova A, Alberts C, Glasius E, Kijlstra A, Buitenhuis HJ, Breebaart AC. Risk factors for ocular sarcoidosis.Doc Ophthalmol. 1989;72(3-4):287-296.
- Hunter DG, Foster CS. Ocular manifestations of sarcoidosis. In: Albert DM, Jakobiec FA, eds. Principles and practice of ophthalmology. Philadelphia: WB Saunders, 1994:443-450.
- Rothova A. Ocular involvement in sarcoidosis.Br J Ophthalmol. 2000;84(1):110-116.
- Babu K. Sarcoidosis in tuberculosis-endemic regions: India.J Ophthalmic Inflamm Infect. 2013;3(1):53. Published 2013 Jun 27.
- Pathanapitoon K, Goossens JH, van Tilborg TC, Kunavisarut P, Choovuthayakorn J, Rothova A. Ocular sarcoidosis in Thailand.Eye (Lond). 2010;24(11):1669-1674.
- Chan AS, Sharma OP, Rao NA. Review for disease of the year: immunopathogenesis of ocular sarcoidosis.Ocul Immunol Inflamm. 2010;18(3):143-151.
- Kim HS, Choi D, Lim LL, Allada G, Smith J R, Austin C R et al. Association of interleukin 23 receptor gene with sarcoidosis.Dis Markers. 2011;31(1):17-24.
- Song Z, Marzilli L, Greenlee BM, et al. Mycobacterial catalase-peroxidase is a tissue antigen and target of the adaptive immune response in systemic sarcoidosis.J Exp Med. 2005;201(5):755-767.
- Gupta D et al. Immune responses to mycobacterial antigens in sarcoidosis: A systematic review. Indian J Chest Dis Allied Sci. 2011;53(1):41-49
- Oswald Richter KA et al. Dual analysis for mycobacteria and propionibacteria in sarcoidosis BAL. J Clin Immunol. 2012;32(5):1129-1140.
- Zimmermann A, Knecht H, Häsler R, Zissel G, Gaede K I, Hofmann S et al.AtopobiumandFusobacteriumas novel candidates for sarcoidosis-associated microbiota.Eur Respir J. 2017;50(6):1600746. Published 2017 Dec 14.
- Ramos-Casals M, Mana J, Nardi N, et al. Sarcoidosis in patients with chronic hepatitis C virus infection: analysis of 68 cases. Medicine (Baltimore). 2005;84(2):69-80.
- Moller DR, Chen ES. What causes sarcoidosis? CurrOpinPulm Med. 2002;8(5):429-434.
- Chopra A, Nautiyal A, Kalkanis A, Judson MA. Drug-Induced Sarcoidosis-Like Reactions.Chest. 2018;154(3):664-677.
- Eser Öztürk H, Süllü Y. Sarcoid-like Granulomatous Intraocular Inflammation Caused by Vemurafenib Treatment for Metastatic Melanoma.Turk J Ophthalmol. 2020;50(1):50-52.
- Familial associations in sarcoidosis. A report to the Research Committee of the British Thoracic and Tuberculosis Association.Tubercle. 1973;54(2):87-98.
- Sverrild A, Backer V, Kyvik KO, Kaprio J, Milman N, Svendsen C B et al. Heredity in sarcoidosis: a registry-based twin study.Thorax. 2008;63(10):894-896.
- Rybicki BA, Kirkey KL, Major M, Maliarik MJ, Popovich J Jr, Chase GA et al. Familial risk ratio of sarcoidosis in African-American sibs and parents.Am J Epidemiol. 2001;153(2):188-193.
- Rybicki BA, Iannuzzi MC, Frederick MM, Thompson, BW, Rossman M D, Bresnitz E A et al. Familial aggregation of sarcoidosis. A case-control etiologic study of sarcoidosis (ACCESS). Am J Respir Crit Care Med. 2001;164(11):2085-2091.
- Rossman MD, Thompson B, Frederick M, Maliarik M, Iannuzzi MC, Rybicki BA et al. HLA-DRB1*1101: a significant risk factor for sarcoidosis in blacks and whites.Am J Hum Genet. 2003;73(4):720-735.
- Iannuzzi MC, Maliarik MJ, Poisson LM, Rybicki BA. Sarcoidosis susceptibility and resistance HLA-DQB1 alleles in African Americans.Am J Respir Crit Care Med. 2003;167(9):1225-1231.
- Adrianto I, Lin CP, Hale JJ, Levin AM, Datta I, Parker R et al. Genome-wide association study of African and European Americans implicates multiple shared and ethnic specific loci in sarcoidosis susceptibility.PLoS One. 2012;7(8):e43907.
- Zhu K, Yin X, Tang X, Zhang F, Yang S, Zhang X. Meta-analysis of NOD2/CARD15 polymorphisms with psoriasis and psoriatic arthritis.Rheumatol Int. 2012;32(7):1893-1900.
- Hofmann S, Franke A, Fischer A, Jacobs G, Nothnagel M, Gaede KI et al. Genome-wide association study identifies ANXA11 as a new susceptibility locus for sarcoidosis. Nat Genet. 2008;40(9):1103-1106.
- Hofmann S, Fischer A, Nothnagel M, Mrazek F, Pabst S, Grohe C et al. Genome-wide association analysis reveals 12q13.3-q14.1 as new risk locus for sarcoidosis.Eur Respir J. 2013;41(4):888-900.
- Fischer A, Schmid B, Ellinghaus D, Gieger C, Wichmann HE, Nebel A et al. A novel sarcoidosis risk locus for Europeans on chromosome 11q13.1.Am J Respir Crit Care Med. 2012;186(9):877-885.
- Fischer A, Ellinghaus D, Nutsua M, Hofmann S, Montgomery CG, Iannuzzi MC et al. Identification of Immune-Relevant Factors Conferring Sarcoidosis Genetic Risk.Am J Respir Crit Care Med. 2015;192(6):727-736.
- Pabst S, Baumgarten G, Stremmel A, Lennarz M, Knufermann P, Gillissen A et al. Toll-like receptor (TLR) 4 polymorphisms are associated with a chronic course of sarcoidosis.Clin Exp Immunol. 2006;143(3):420-426.
- Davoudi S, Chang VS, Navarro-Gomez D, Stanwyck L K, Sevgi D D, Papavasileiou E et al. Association of genetic variants inRAB23andANXA11with uveitis in sarcoidosis.Mol Vis. 2018;24:59-74. Published 2018 Jan 21.
- Umazume, A., Kezuka, T., Okunuki, Y., Ooshita, M., Usui, Y., Hirano, M., Yamashina, A, Goto, H., 2014. Prediction of severe cardiac involvement by fundus lesion in sarcoidosis. Jpn. J. Ophthalmol. 58, 81-85.
- Kojima K, Maruyama K, Inaba T, Nagata K, Yasuhara T, Yoneda K et al. The CD4/CD8 ratio in vitreous fluid is of high diagnostic value in sarcoidosis.Ophthalmology. 2012;119(11):2386-2392.
- Agostini C, Trentin L, Facco M, Sancetta R, Cerutti A, Tassinari C et al. Role of IL-15, IL-2, and their receptors in the development of T cell alveolitis in pulmonary sarcoidosis.J Immunol. 1996;157(2):910-918.
- Bronson LJ, Fisher YL. Sarcoidosis of the paranasal sinuses with orbital extension.Arch Ophthalmol. 1976;94(2):243-244.
- Umur KA, Tayfun B, Oguzhan O. Different ophthalmologic manifestations of sarcoidosis.Curr Opin Ophthalmol. 2012;23(6):477-484.
- Birnbaum AD, Oh FS, Chakrabarti A, Tessler HH, Goldstein DA. Clinical features and diagnostic evaluation of biopsy-proven ocular sarcoidosis.Arch Ophthalmol. 2011;129(4):409-413.
- Mavrikakis I, Rootman J. Diverse clinical presentations of orbital sarcoid. Am J Ophthalmol. 2007;144(5):769-775.
- Prabhakaran VC, Saeed P, Esmaeli B, et al. Orbital and adnexal sarcoidosis. Arch Ophthalmol. 2007;125(12):1657-1662.
- Demirci H, Christianson MD. Orbital and adnexal involvement in sarcoidosis: analysis of clinical features and systemic disease in 30 cases.Am J Ophthalmol. 2011;151(6):1074-1080.e1.
- Pasadhika S, Rosenbaum JT. Ocular sarcoidosis. Clin Chest Med. 2015;36(4):669-683.
- Babu K, Smitha KS, P Manjandavida F. Orbital Sarcoidosis in a High TB Endemic Country - A Case Series from South India [published online ahead of print, 2020 Feb 19].Ocul Immunol Inflamm. 2020;1-6.
- Biswas J, Krishnakumar S, Raghavendran R, Mahesh L. Lid swelling and diplopia as presenting features of orbital sarcoid.Indian J Ophthalmol. 2000;48(3):231-233.
- Dursun D, Akova YA, Bilezikci B. Scleritis associated with sarcoidosis. Ocul Immunol Inflamm. 2004;12(2):143-148.
- Heiligenhaus A, Michel D, Koch JM. Nodular scleritis in a patient with sarcoidosis. Br J Ophthalmol. 2003;87(4):507-508.
- Babu K, Kini R, Mehta R. Scleral nodule and bilateral disc edema as a presenting manifestation of systemic sarcoidosis. Ocul Immunol Inflamm. 2010;18(3):158–161.
- Dodds EM, Lowder CY, Barnhorst DA, Lavertu P, Caravella LP, White DE. Posterior scleritis with annular ciliochoroidal detachment. Am J Ophthalmol. 1995;120(5):677-679.
- Beardsley TL, Brown SV, Sydnor CF, Grimson BS, Klintworth GK. Eleven cases of sarcoidosis of the optic nerve. Am J Ophthalmol. 1984;97(01):62-77.
- Gudeman SK, Selhorst JB, Susac JO, Waybright EA. Sarcoid optic neuropathy. Neurology. 1982;32(06):597-603.
- Liu JP, Bender MB. Horner’s syndrome, bilateral sensory motor trigeminal and other cranial neuropathies in a patient with sarcoidosis. Neuroophthalmol. 1981; 2(2):117-122.
- Lewis GE. Sarcoidosis: Speculations as to the Stages of Infection.Br Med J. 1941;1(4188):552-554.
- Donin J. Sarcoidosis and the tonic pupil. In Smith JL, ed. Neuroophthalmology Symposium of the University of Miami and. The Bascom Palmer Eye Institute. Vol Hallandale, Florida, Huffman Publishing Co., 1970:81-87.
- Kupersmith MJ, Aleksic SN. Bilateral pupillary cholinergic supersensitivity in a case of sarcoidosis. Neuroophthalmology.1984;4(1):15-20.
- Kirkham TH, Kline LB. Monocular elevator paresis, Argyll Robertson pupils and sarcoidosis.Can J Ophthalmol. 1976;11(4):330-335.
- Poole CJ. Argyll Robertson pupils due to neurosarcoidosis: evidence for site of lesion.Br Med J (Clin Res Ed). 1984;289(6441):356.
- Verma A, Biswas J. Choroidal granuloma as an initial manifestation of systemic sarcoidosis. Int Ophthalmol. 2010; 30(5):603-6.260.
- Herbort CP, Rao NA, Mochizuki M; members of Scientific Committee of First International Workshop on Ocular Sarcoidosis. International criteria for the diagnosis of ocular sarcoidosis: results of the first International Workshop On Ocular Sarcoidosis (IWOS).Ocul Immunol Inflamm. 2009;17(3):160-169.
- Takase H, Shimizu K, Yamada Y, Hanada A, Takahashi H, Mochizuki M. Validation of international criteria for the diagnosis of ocular sarcoidosis proposed by the first international workshop on ocular sarcoidosis.Jpn J Ophthalmol. 2010;54(6):529-536.
- Acharya NR, Browne EN, Rao N, Mochizuki M; International Ocular Sarcoidosis Working Group. Distinguishing Features of Ocular Sarcoidosis in an International Cohort of Uveitis Patients.Ophthalmology. 2018;125(1):119-126.
- Mochizuki, M., Smith, J. R., Takase, H., Kaburaki, T., Acharya, N. R., Rao, N. A., & International Workshop on Ocular Sarcoidosis Study Group (2019). Revised criteria of International Workshop on Ocular Sarcoidosis (IWOS) for the diagnosis of ocular sarcoidosis. The British journal of ophthalmology, 103(10), 1418-1422.
- Ohno S, Aoki K, Usui M, Uchio E, Rothova A. (1998) Posterior segment involvement in sarcoidosis. in Uveitis today . eds Ohno S, Aoki K, Usui M, Uchio E (Elsevier, Amsterdam), pp 207-210.
- Cowan CL. Differential diagnosis of ocular sarcoidosis. Ocul Immunol Inflamm.2010;18(6):442-451.
- Lowder C, Belfort R Jr, Lightman S, et al.; Ozurdex HURON Study Group. Dexamethasone intravitreal implant for noninfectious intermediate or posterior uveitis. Arch Ophthalmol 2011;129 (5):545–553
- Pearson PA, Comstock TL, Ip M, et al. Fluocinolone acetonide intravitreal implant for diabetic macular edema: a 3-year multi- center, randomized, controlled clinical trial. Ophthalmology. 2011;118(08):1580-1587.
- Baughman RP, Lower EE, Ingledue R, Kaufman AH. Management of ocular sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2012;29(1):26-33.
- Bhat P, Cervantes-Castañeda RA, Doctor PP, Anzaar F, Foster CS. Mycophenolate mofetil therapy for sarcoidosis-associated uveitis. Ocul Immunol Inflamm. 2009; 17(3):185-190.
- Jaffe GJ, Dick AD, Brézin AP, et al. Adalimumab in patients with active noninfectious uveitis. N Engl J Med. 2016;375(10):932-943.
- Nguyen QD, Merrill PT, Jaffe GJ, et al. Adalimumab for prevention of uveitic flare in patients with inactive non-infectious uveitis controlled by corticosteroids (VISUAL II): a multicentre, double- masked, randomised, placebo-controlled phase 3 trial. Lancet. 2016;388(10050):1183-1192.
- Lower EE, Baughman RP, Kaufman AH. Rituximab for refractory granulomatous eye disease. Clin Ophthalmol. 2012;6:1613-1618.
- Silpa-Archa S, Oray M, Preble JM, Foster CS. Outcome of tocilizumab treatment in refractory ocular inflammatory diseases. Acta Ophthalmol. 2016;94(06):e400–e406.
- Damsky W, Thakral D, Emeagwali N, Galan A, King B. Tofacitinib treatment and molecular analysis of cutaneous sarcoidosis. N Engl J Med. 2018;379(26):2540-2546.
- Rotenberg C, Besnard V, Brillet P-Y, Giraudier S, Nunes H, Valeyre D. Dramatic response of refractory sarcoidosis under ruxolitinib in a patient with associated JAK2-mutated polycythemia. Eur Respir J. 2018;52(06):1801482.
- Wei JJ, Kallenbach LR, Kreider M, Leung TH, Rosenbach M. Resolution of cutaneous sarcoidosis after Janus kinase inhibitor therapy for concomitant polycythemia vera. JAAD Case Rep. 2019;5(4):360-361.
- Sève P, Jamilloux Y, Tilikete C, Gerfaud-Valentin M, Kodjikian L, El Jammal T. Ocular Sarcoidosis.Semin Respir Crit Care Med. 2020;41(5):673-688.

