Introduction
Peripheral retinal degenerations (PRD) are considered a risk factor for rhegmatogenous retinal detachment (RRD) [1, 2]. The annual incidence of RRD varies between 6.3 and 18.2 cases per 100,000.
RRD develops when liquefied vitreous humor accelerated by rotary eye movements passes through retinal tears or retinal holes into subretinal space and detaches neurosensory retina from the underlying pigment epithelium. PRDs when missed during retinal examination can lead to vision threatening complications.3
Classification [3]

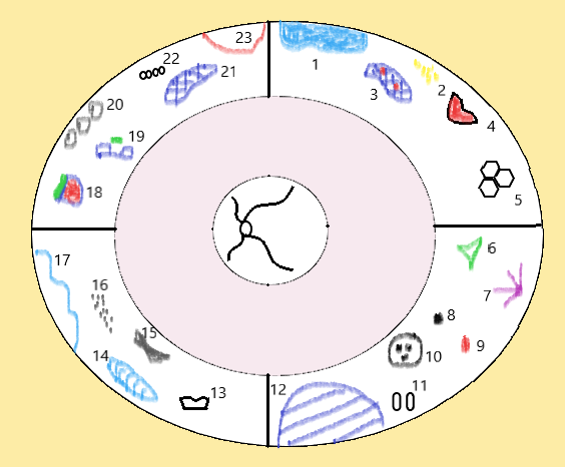
- Retinal detachment
- Retinal drusen
- Lattice with holes
- Full-thickness tear
- Honeycomb degeneration
- Preretinal fibrosis
- Retinal tuft
- Unifocal hypertrophy of the retinal pigment epithelium
- Hemorrhage
- Grouped congenital hypertrophy of the retinal pigment epithelium (“bear tracks”)
- Pearl degeneration
- Retinoschisis
- Atrophic retinal hole
- Snail-track degeneration
- Dark-without-pressure
- Snowflake degeneration
- White-without-pressure
- Flap tear
- Operculated retinal tear
- Paving-stone degeneration
- Lattice degeneration
- Peripheral cystoid degeneration
- Retinal dialysis
Developmental
Zonular traction tuft
A zonular traction tuft is a thickened zonule that is displaced posteriorly and attached to the anterior retina. This results in an abnormal tuft of tissue drawn from the retinal surface toward the ciliary body. With continual traction from the zonules on the tuft, thickening and degeneration occurs, and results in tears at the base of these lesions. 4
These lesions are present at birth and occurred with equal frequency in all decades beyond the first. In more than 2/3rd cases, it is single and present nasally.
Trophic complications included holes in the base of tufts (partial in 10%; full thickness in 4%). Tractional complications included rupture of tuft (5%) and full-thickness tears. These tufts do not require prophylactic treatment but require careful monitoring.
INTRARETINAL DEGENERATIONS
1. Senile retinoschisis
Senile retinoschisis is defined as the abnormal splitting of the neurosensory retina, at the outer plexiform layer. There is no genetic transmission and no gender predilection. Progression of peripheral cystoid degeneration results in retinoschisis when the glial supporting elements stretch, muller cells break and shallow cavities develop which split the retina. It is usually asymptomatic and non-progressive. Quite often noted in hyperopic eyes (70 %), It is usually bilateral, asymptomatic, and found in the inferotemporal, or less commonly in the superotemporal quadrants.5It is clinically seen as a transparent, round dome, smooth, immobile elevation in the inferotemporal fundus.
2. White-without-pressure
White with or without pressure is not degeneration, but rather a finding on fundus examination seen between equator and ora serrata. White with pressure (WWP) is seen in about 15 % of moderate to high myopes. Prevalence of WWOP increases with axial length and high degrees of myopia.
White without pressure indicates an area of the peripheral retina that has a white sheen with a sharply demarcated border which is seen even without indentation.(figure 1) White without pressure (WWOP) is an advanced form of white with pressure and maybe quite extensive (360°). There is obscuration of underlying choroidal markings. They contain islands of normal tissue wherein, making the distinction from retinal holes is difficult. However, on indentation, retinal hole diameter increases and pouts, making it easier to differentiate from the normal retina. The temporal half of the retina is involved more often than the nasal retina.6

Figure 1
The pathogenesis is still unknown- It has been hypothesized that it is a manifestation of peripheral vitreous traction or an abnormal reflex from a structurally normal VR interface. Spectral-domain optical coherence tomography (SD-OCT) has demonstrated hyper-reflectivity of the photoreceptor zone in eyes with WWOP.7White with pressure and white without pressure have increased incidence of vitreoretinal traction but not of retinal detachment.8 Giant Retinal Tears (GRT) are seen commonly in eyes with WWOP.9 If WWOP is found in the fellow eye of a patient with spontaneous GRT, a prophylactic barrage laser should be performed.
3. Dark-without-pressure
Dark without pressure are asymptomatic patches darker than surrounding retina. They are transient and change size and shape, can even completely disappear in few weeks. There are no functional defects and hence they do not need treatment. It is hypothesized that they represent altered reflex at the RPE and outer retinal level, because the RPE photopigment has a different spectral range compared to the rest of the retina. These patches are seen well on red free photography, explained by the altered photopigment that absorbs shorter wavelength light better than longer wavelength.10
4. Peripheral cystoid degeneration
Peripheral microcystoid degeneration is the most common form of peripheral retinal degeneration and is seen in almost all patients above 20 years of age. Cystoid degeneration begins at the ora serrata, particularly at the base of dentate processes, at the level of the retinal nerve fiber layer, and extends posteriorly and circumferentially to form a band that encircles the eye. It presents as tiny vesicles with indistinct boundaries on a greyish white background making the retina appear white, thickened, and less transparent. Scleral depression makes the area appear whiter. The cystoid spaces themselves appear reddish because of the increased visibility of the choroidal reflex.11
On histopathology, cystoid cavities are seen to be separated by pillars composed of compressed Muller cells and photoreceptor axons that coalesce to form interlacing tunnels.12
CLINICAL SIGNIFICANCE
- Retinal Holes- Develop due to rupture of the inner wall of cystoid cavity.
- Retinoschisis

5. Snowflake degeneration
Snowflake vitreoretinal degeneration appears as tiny yellow-white crystalline, frost like spots in the far periphery. Retinal transparency is lost. It extends from the ora to the areas anterior to the equator with irregular borders. Sometimes the snowflakes can be distributed radially along retinal vessels in a sectoral fashion or in a circumferential fashion along with the ora. Snowflake degeneration is also seen more commonly in the supero temporal quadrant.13 It is usually bilateral.
SVD is a part of a spectrum of Vitreous fibrillary disorders that exhibits predominantly autosomal dominant (AD) inheritance. A small percentage of sporadic cases are also seen. A phenotypic overlap of SVD with Stickler syndrome type I and type II and Wagener’s syndrome has been researched and they have found to have over lapping features but all three are established distinct entities.14The spectrum of SVD involves corneal gutta, early onset cataract, fibrillar anomaly of the vitreous, lattice degeneration, radial paravascular retinal degeneration, pigment clumping along the snow flake or just posterior to it, sheathing of vessels upto posterior pole, ONH dismorphism and retinal detachment.15 Small holes can be seen within the snowflake degeneration. Neovascular retinal tufts adjacent to areas of snowflake degeneration have been seen and these patients need photocoagulation to the neovascular tufts along with laser to holes if any. SVD starts as vitreous degeneration and then affects the inner retina. 16, 17
6. Pearl degeneration
Pearl degeneration (ora pearls, pearls of the ora serrata) is a rare congenital pathology of the peripheral retina that may look like white beads resembling retinal drusen. Ora pearls appear as milk-white dense glistening spheroids located at the ora serrata or pars plana.18 there is usually a single isolated pearl, or less frequently – multiple pearls that are bilateral and symmetrical. Pearl degenerations are benign and do not require prophylactic laser treatment.19
Vitreoretinal degenerations
1. Snail-track degeneration
Snail track degeneration is a glistening white frost like area, elongated oval or spindle shaped and has multiple white dots within it. They are seen in the temporal quadrant and the majority are seen at least 2DD anterior to the equator. It is seen predominantly in myopes and is bilateral.20, 21
This lesion resembles lattice degeneration and can sometimes be noted as an early lattice. But maybe simultaneously present in the same eye also. Both may have a single or multiple hole within it. Lattice appears as isolated areas of retinal thinning circumferentially oriented in the retinal periphery with networks of white lines passing across it, suggestive of sclerosed vessels. Snail track lesions occurred as elongated patches of retinal thinning with irregular borders and no sclerosed vessels. Round holes, lamellar holes, and progression to lattice degeneration with vitreoretinal adhesion have been noted in the snail track degeneration.22
Management Snail tracks with holes need barrage laser22, 23Lesions without holes can be observed
2. Lattice degeneration
Lattice degeneration is not an uncommon peripheral vitreoretinal disorder present in 6-10% of the population. Incidence is higher in moderate myopes (4D-6D). Only 0.5-1% of eyes with lattice degeneration will develop a retinal detachment, on the other hand, approximately 20-30% of patients with rhegmatogenous retinal detachments have lattice degeneration.24
Clinical features-Lattices are circumferentially oriented linear or ovoid areas of retinal thinning sometimes crossed by whitish lines that represent hyalinized retinal vessels. There can also be superficial whitish-yellow flecks or patches of varying degrees of pigmentation. Within the lattice, there may be round or linear holes and small atrophic round holes.

Figure 2
Pathogenesis- The three main factors in pathogenesis are - Localized thinning of the inner retinal layersVitreous liquefaction overlying thinned retina with vitreous condensation with exaggerated vitreoretinal attachments at the posterior margins and lateral horns of the lesion.Vitreoretinal traction at the margins of the break where the vitreoretinal adhesion is strong. This intermittent traction aggravated by eye movements results in development of tears resulting in retinal detachment, when left untreated.
Indications for treatment of lattice
1. Lattice with tractional holes or breaks within it
2. Symptomatic lattice- Flashes and floaters due to vitreoretinal traction at the edge of the lattice
3. RD in the other eye
4. Patients undergoing refractive procedures
5. Lattice with subclinical RD (Surrounding SRF less than 1DD size) even if pigmented and appears old.
6. One-eyed patient
7. Family history of RD
8. Atypical lattice degeneration- Radial or paravascular Lattice (Seen in Hereditary vitreoretinal degenerations like Stickler syndrome). Usually within equator.
Indications to observe
- Asymptomatic lattice
- Lattice with no breaks/ holes
- Pigmented lattice with no breaks/holes
- Lattice with atrophic holes
These patients can be explained the symptoms of vitreoretinal traction (flashes, floaters) and can be regularly followed up at 6 monthly intervals
Investigations
Thorough Indirect ophthalmoscopy examination with scleral indentation with documentation- Mentioning specifics about the location of the lattice with the location of holes or breaks within the lattice (usually on the edges of the lattice). The location should be specified with respect to a vessel/ pigment/ vortex vein/ any anatomical landmark.
Treatment - Barrage laser (Figure 3)
Mechanism of action of Barrage Laser- It generates a stiff chorioretinal adhesion surrounding the lattice. This counters vitreoretinal traction and prevents the fluid vitreous from entering the subretinal space.

Figure 3
Three rows of confluent grade 2-3 burns covering all the margins of the break should be applied. Special attention should be paid to the anterior margin near the ora. If inadequately visualized, scleral indentation during laser will be useful. If the break is remarkably close to ora, the posterior edge is lasered and extended to the sides of the break connecting it to the ora. This forms U shaped rows of laser enclosing the break with the ora.
The most common cause of retinal detachment in an eye treated with barrage laser for lattice is, insufficient treatment of anterior edge where the vitreous adhesion to the base is strongest keeping the break open leading to RRD.
3. Cystic Retinal tufts
Cystic tufts are congenital lesions where there are abnormal vitreoretinal adhesions causing small breaks or tears. They are well demarcated and may have pigmentation around it. They are characteristically seen in the equatorial and just anterior to equator regions.25 It can be present in any quadrant and is usually unilateral. Pathology lies in the outer retina and RPE which appear degenerative. Cystic retinal tufts may be avulsed by vitreous traction with or without posterior vitreous detachment and lead to horseshoe tears or irregular breaks leading to retinal detachment. Cystic retinal tufts are commonly seen in the general population and only a small percentage progress to retinal detachment. Hence prophylactic treatment is usually not needed.26
4. Peripheral retinal breaks
Definition- A retinal break is a full-thickness break in the neurosensory retina. It can be either a hole or a tear. The general prevalence of retinal breaks is up to 14% and is more in cases of moderate myopia due to thin retina. 27
Retinal Tears
Retinal tears are caused by degeneration of the inner retinal layers, overlying vitreous degeneration, and traction. These tears may be round, linear, or horseshoe-shaped, depending on the characteristics of the retina and vitreous traction that produced them (Figure 4). A flap of the sensory retina located above the break attached to the detached vitreous cortex is called an Operculated tear.
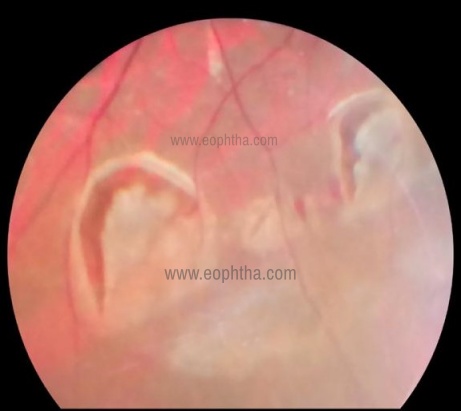
Figure 4
Retinal infections can also cause the formation of retinal breaks because of the destruction of the already friable inflamed retina.
Retinal Holes
A round break is called a hole. An atrophic retinal hole is a break that is not caused by traction. It is most likely produced by an atrophic process and is generally thought to be the result of underlying vascular disease insufficiency of the choroid and choriocapillaris. (Figure 5)

Figure 5
Symptoms
Photopsia or flashes from vitreous traction causing mechanical stimulation to the retina
Sudden onset of numerous tiny black floaters caused due to avulsion of small retinal vessels resulting in minor vitreous hemorrhage
The presence of a few floaters in the middle age group caused possibly by posterior vitreous detachment (PVD)should be explained to the patient
All the above symptoms should be thoroughly examined, if floaters are only due to PVD monthly review for 3-6 months until the patient’s symptoms resolve is necessary
Indirect Ophthalmoscopy with scleral indentation
Differentiation of a hole from a hemorrhage
A break tends to change color (from dark red to gray) during the depression because of compression of the underlying choroidal vasculature and the edges pout during indentation, whereas a hemorrhage does not change color or pout during scleral indentation
5. Microcystoid degeneration
Cystic degeneration in the periphery.
This is the most frequently seen degenerative lesion of the area and characterized by small bubbles or vacuoles in the peripheral retina near the ora. These occur in the outer plexiform and inner nuclear layers of the retina. Mostly symmetrical, they are more in the temporal retina than nasal, more superiorly than inferiorly The inner wall of a cyst may be absent giving the impression of that of a retinal hole. It thus does not predispose to retinal detachment.28
6. Pars plana cyst
Pars plana cysts are solitary or multiple clear blisters like cystoid spaces between the pigmented and nonpigmented epithelial layers of pars plana, seen extending between the ora and the ciliary process.(Figure 6) Their tricky location makes it difficult to examine and image.29 Most are acquired cysts secondary to ocular disease like chronic retinal detachments, chronic uveitis, post-traumatic uveitis, advanced glaucoma30 also in patients with multiple myeloma.31 They vary in size from 1 to 3 disc diameters and are usually located in the temporal side of the eye. Cysts may be clear or opaque with different contents. Pars plana cysts appear dome-shaped with the convexity anteriorly towards ciliary processes. Posteriorly, they narrow into neck like processes which are continuous with areas of cystoid degeneration in peripheral retina29 Pars plana cysts are present in 3% to 18% of all patients29 The cavities are filled with fluid that contains hyaluronic acid, got from the proliferation of multiple layers of inner non pigmented ciliary epithelium.32 Ciliary capillaries are shown to run from the ciliary stroma into the cystic cavity, these have gap junctions and hence leak fluorescein.32 The cystic cavity may also contain fibrocytes or zonular fibril bundles. The cavity of the opaque cyst is filled with amorphous globules. In patients with multiple myeloma the cysts are filled with paraaminosalicylic acid (PAS)-positive protein, which is the same myeloma protein (IgG) as is found in the patient's serum.31 Pars plana cysts are thought to be associated with peripheral cystoid degeneration, which causes the formation of holes leading to retinal detachment.

Figure 6
Chorioretinal degenerations
1. Chorioretinal Atrophy and Paving stone degeneration
Chorioretinal atrophy (CRA) manifests as small depigmented, well-defined patches in the retina periphery showing the underlying prominent choroidal vasculature. Paving-stone degeneration (PSD) and chorioretinal atrophy is present in normal adults of the elderly age group and is a chance finding during fundus examination. These lesions are higher in myopic individuals. The lesions are more common in the inferior half of the fundus. It is usually bilateral.
Chorioretinal atrophy is seen anterior to the equator up to the ora serrata. Paving stone degeneration is also seen in the periphery but can be present up to the equator. SD in the macula is one form of geographic atrophy. PSD is also found anterior to peripheral intraocular tumors, such as choroidal malignant melanoma and nevi, and metastatic choroidal tumors.33 This is believed to be related to peripheral choroidal insufficiency induced by the space-occupying masses causing ischemia and overlying RPE and sensory retina.
2. Hypertrophy of the retinal pigment Epithelium
CHRPE is an asymptomatic congenital hamartoma. Prevalence is low about 1.25% in the general population.34 It can be classified into three types

Solitary CHRPE
Flat round hyperpigmented lesion smooth well-defined margins. Color may be from deep brown to black. (Figure 7) A marginal depigmented halo could surround the lesion and there could be punched out inner lacunae with hypopigmentation, suggestive of areas of normal retina. Seen commonly in the supertemporal or equatorial region. An overlying retina with vasculature is normal but sometimes vascular abnormalities like vessel obliteration, micro aneurysmal changes or neovascularization can also be seen. There is a slow increase in size in 45-80% lesions over 3-4 years have been documented34

Figure 7
Grouped CHRPE
When several lesions of varying size are arranged in a cluster resembling bear-tracks they are called grouped CHRPE. Each cluster may have 3-30 lesions which can be 100-300 microns in size.
Pathogenesis- They are thought to be a cluster of hyperpigmented atypical RPE cells that are derived from the edge of the optic cup and migrate along the embryologic lines akin to cutaneous lines of Blaschko35
Multiple CHRPE
Multiple CHRPE in FAP are smaller and present as small satellite lesions around larger lesions. More than 4 widely spaced lesions per eye or bilateral involvement are suggestive of FAP. (Figure 8) FAP is an autosomal dominant disease with numerous adenomatous polyps in the colon and rectum. This is a premalignant condition. Extra colonic manifestations of FAP is Turcot syndrome (With brain tumor lesions- Medulloblastoma, astrocytoma, ependymoma) and Gardener’s Syndrome (Part of FAP-Gastrointestinal and hamartomas of the skeleton and soft tissues. Tumors of the thyroid, adrenal liver, and bladder)

Figure 8
Multimodal Imaging
SD-OCT- General thinning of neural retina with photoreceptor loss with thickened irregular RPE. Areas of RPE hyperpigmentation caused shadowing of underlying choroidal structures. Areas of lacunae showed RPE loss with window defect and overexposure of underlying choroid.
Autofluorescence- Paradoxical hypoautofluorescence, since despite an RPE hyperplasia there is no lipofuschin produced by abnormally thickened RPE cells.
FFA/ICGA- Blocked fluorescence
Differential Diagnosis
- Choroidal nevus
- Melanocytoma
- Malignant Melanoma
- Melanocytoma
|
CHRPE |
Nevus |
Malignant Melanoma |
Melanocytoma |
|
Unilateral. Has a surrounding halo with lacunae within the lesion. Well defined. |
FLAT and round, have ill-defined borders. No halo |
Thickness >2mm, Orange pigment overlying |
Located near the ONH |
|
Jet black brown or darker colouration |
Slate grey colour |
May have a neurosensory detachment over it |
Uniform densely pigmented dark lesions |
|
Overlying Drusen |
B scan- Initial high spike with acoustic hollowness |
Grow more rapidly than typical nevi |
|
|
Scotoma in visual fields |
No scotoma |
3. Peripheral retinal drusen
Peripheral retinal drusen are extracellular deposits of proteins and fatty unstructured material between the basement membrane of RPE and inner collagenous layer of Bruch's. They form dome-shaped lesions. They are an age-related and bilateral phenomenon. 36
4. Myopic choroidal atrophy
Diffuse choroidal or chorioretinal atrophy is characterized by diffuse or circumscribed choroidal depigmentation with thinning of the overlying retina. Typically occurs on the posterior pole but can be located on the equator and beyond. Retinal holes developing in atrophic areas can lead to RD and such holes are difficult to visualize.37 (Figure 9)

Figure 9
5. Reticular degeneration
Reticular degeneration of the pigment epithelium is a distinct entity with many features that warrant consideration. The etiology and pathogenesis of RDPE are unknown. Fundamentally, RDPE increases with advancing age, is a bilateral zonal degeneration that affects the equatorial belt of the eye, is characterized by hypopigmentation with linear, geometric hyperpigmentation, and is associated with the presence of many small, subretinal drusen and hard drusen.38

Figure 10
These factors imply an aging-related degeneration that affects both eyes in a zone with similar vascular and metabolic characteristics. Relative loss of pigment, possibly with a thinning and spreading of the pigment epithelial cells, indicates a retrograde process in the pigment epithelial cells. Hyperpigmented lines in a geometric reticular pattern are produced by retained and often increased pigmentation, possibly with a thickening, compression, and reactive hyperplasia of the pigment epithelial cells at the borders of hypopigmented areas. Reticular degeneration of the pigment epithelium is a possible risk factor for age-related macular degeneration, particularly when RDPE is advanced in degree.
References
- Saksonova EO, Zakharova GI, Platova LA, et al. Prevention of retinal detachment in patients with peripheral vitreochorioretinal dystrophies. Oftalmol Zh. 1983;38(3):151–5. (In Russian)
- Conart JB, Baron D, Berrod JP. Degenerative lesions of the peripheral retina. J Fr Ophtalmol. 2014;37(1):73– 80. doi:10.1016/j.jfo.2013.09.001.
- Venera A. Shaimova; Peripheral Retinal Degenerations as a Risk Factor for Rhegmatogenous Retinal Detachment.
- Foos RY. Zonular Traction Tufts of the Peripheral Retina in Cadaver Eyes. Arch Ophthalmol. 1969;82(5):620–632. doi:10.1001/archopht.1969.00990020616009
- Kanski JJ, Bowling B. Clinical ophthalmology: a systematic approach. 7th ed. Edinburgh: Elsevier/Saunders; 2011.
- Shukla M, Anuja O P. White with pressure (WWP) and white without pressure (WWOP) lesions. Indian J Ophthalmol 1982;30:129-32
- Fawzi AA, Nielsen JS, Mateo-Montoya A et al. Multimodal imaging of white and dark without pressure fundus lesions.Retina 2014; 34:12:2376-2381
- Bedi N., Shah M., Association of peripheral retinal degeneration with retinal detachment in myopia. Int.J.Med.Sci. Educ 2019;6(4):92-95
- Taleb EA. Giant Retinal Tear Retinal Detachment Etiologies, Surgical Outcome and Incidence of Recurrent Retinal Detachment after Silicon Oil Removal. SM Opthalmol J. 2016; 2(1): 1008.
- Fawzi A A et al.Retina. 2014;34(12):2376-2387
- Yanoff M, Fine BS. Ocular Pathology: A Text and Atlas (2nd ed). Philadelphia: Harper & Row, 1982:504-505.
- William L Jones. Peripheral ocular fundus (3rd edition). Butterworth, Heinmann, Elsevier. 1998:72-75
- William L Jones. Peripheral ocular fundus(3rd edition). Butterworth, Heinmann, Elsevier. 1998:102-105
- Lee MM, Ritter R, 3rd, Hirose T, Vu CD, Edwards AO. Snowflake vitreoretinal degeneration: follow-up of the original family. Ophthalmology.2003;110:2418–2426
- Xiaodong Jiao, Robert Ritter, J. Fielding Hejtmancik, Albert O. Edwards; Genetic Linkage of Snowflake Vitreoretinal Degeneration to Chromosome 2q36.Invest. Ophthalmol. Vis. Sci.2004;45(12):4498-4503
- Pollack A, Uchenik D, Chemke J, Oliver M. Prophylactic Laser Photocoagulation in Hereditary Snowflake Vitreoretinal Degeneration:A Family Report.Arch Ophthalmol.1983;101(10):1536–1539.
- Edwards, A. Clinical features of the congenital vitreoretinopathies.Eye22,1233–1242 (2008). https://doi.org/10.1038/eye.2008.38
- Byer NE. The peripheral retina in profile: a stereoscopic atlas. Torrance: Criterion Press; 1982.
- Goldbaum MH. Retinal examination and surgery. In: Peyman GA, Saunders DR, editors. Principles and practice of ophthalmology. Philadelphia: WB Saunders; 1980. p. 988.
- William L Jones. Peripheral ocular fundus (3rd edition). Butterworth, Heinmann, Elsevier. 1998:72-75
- Shukla M, Ahuja Or. A possible relationship between lattice and snail track degenerations of the retina. Am J OphthalmoI1981;92:482-485
- Hyams, S. W., Meir, E., Ivry, M., Krakowski, D., Barkai, S., Jedwab, E., and Neumann, E.: Chorioretinal lesions predisposing to retinal detach ment. Am. J. Ophthalmol. 78:429, 1974
- Morse, P. H.: Lattice degeneration of the retina and retinal detachment. Am. J. Ophthalmol. 78:930, 1974.
- The Retinal Atlas Edition 2-Peripheral Retinal Degenerations and Rhegmatogenous Retinal Detachment. Chapter 11. Page 971-973
- William L Jones. Peripheral ocular fundus (3rd edition). Butterworth, Heinmann, Elsevier. 1998:102-105
- Byer NE. Cystic Retinal Tufts and Their Relationship to Retinal Detachment.Arch Ophthalmol.1981;99(10):1788–1790. doi:10.1001/archopht.1981.03930020662007
- Straatsma BR, Foos RY, et al. Degenerative Diseases of the Peripheral Retina. In TO Duane, EA Jaeger (eds), Clinical Ophthalmology. Philadelphia: Harper & Row, 1976;1-30
- https://eyewiki.aao.org/Peripheral_Retinal_Degenerations
- Vinod Kumar, Pradeep Kumar, Shayeri Roy. Ophthalmic Surgery Lasers and Imaging. Retina49(10):e169-e170
- Okun E. “Gross and microscopic pathology in autopsy eyes. IV. Pars plana cysts. Am J Ophthalmol. 1961 Jun;51:1221-8.
- Sanders TE, Podos S. Pars plana cysts in multiple myeloma. Trans Am Acad Ophthalmol Otolaryngol. 1966 Nov-Dec;70(6):951-8. PMID: 5971478.
- Gärtner J. Fine structure of pars plana cysts. Am J Ophthalmol. 1972;73(6):971-984.
- Brown GC, Shields JA. Choroidal melanomas and paving stone degeneration. Ann OphthalmoI1983;15:705-708
- Stephen Ryan 6th Edition-Tumors of retina-Congenital hypertrophy of Retinal Pigment Epithelium- Page 2490-2494
- Meyer CH, Freyschmidt-Paul P, Happle R, Kroll P. Unilateral linear hyperpigmentation of the skin with ipsilateral sectorial hyperpigmentation of the retina. Am J Med Genet A. 2004 Apr 1;126A(1):89-92. doi: 10.1002/ajmg.a.20483. PMID: 15039978.
- Yannuzzi L. The retinal atlas. Amsterdam: Elsevier Science; 2010.
- Kanski JJ, Bowling B. Clinical ophthalmology: a systematic approach. 8th ed. Edinburgh: Elsevier/Saunders
- Kanski JJ, Bowling B. Clinical ophthalmology: a systematic approach. 8th ed. Edinburgh: Elsevier/Saunders


