The Implant
Durysta (ALLERGAN) is the first-ever intracameral sustained-release bimatoprost implant. The glaucoma treatment paradigm has shifted towards earlier intervention and targeting the actual pathology. The main problem one faces with glaucoma management is compliance with the medications. The intracameral bimatoprost addresses the problem satisfactorily.Durysta, a 10-microgram bimatoprost implant, becomes the first biodegradable, intracameral sustained release implant indicated to reduce Intraocular pressure (IOP). It is hard for patients to maintain quality of life while being on topical anti-glaucoma medications – The Intracameral Bimatoprost makes it possible.
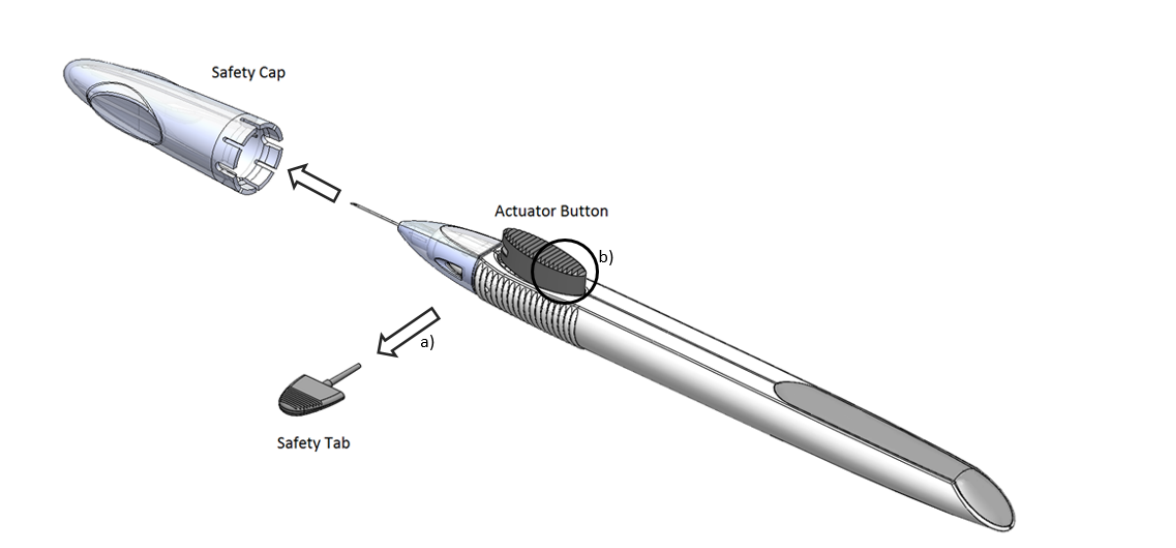
Administration of the Implant
Durysta is a rod-like implant preloaded within a sterile applicator with a 28-gauge needle tip. The needle is inserted under an aseptic condition in the anterior chamber and then with the press of a trigger button implant is released. After the implantation, the needle is removed and the patient is instructed to sit upright for at least an hour. The implant settles down to inferior anterior chamber angle.
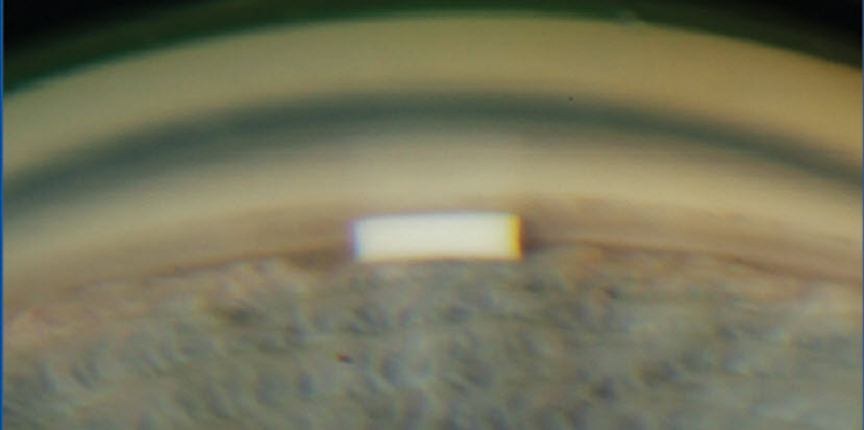
Mechanism of the implant?
DURYSTA acts in the same way as other PG Analogue molecule by increasing the uveoscleral outflow. They have an exciting prospect to remodel of the ciliary body and Trabecular meshwork by the 24-hour continuous release of bimatoprost molecules. The total dose of bimatoprost contained in the 10-microgram implant is similar to the amount in a single drop of the ophthalmic bimatoprost 0.03% solution. The maximum bimatoprost concentration observed in any patient was 0.00224ng/ml.

The evidence behind its safety & efficacy
To answer it we should know a little bit about two landmark trials that were conducted on this intracameral implant
ARTEMIS Study
The FDA approval of Durysta was based on two-phase 3 ARTEMIS study from the 20 months (including 8 months of extended follow-up) evaluating 1122 subjects on the efficacy and safety of DURYSTA with twice-daily timolol in patients with open-angle glaucoma (OAG) or Ocular Hypertension (OHT). DURYSTA reduced IOP by approximately 30% from baseline IOP over the 12-week primary efficacy period.
APOLLO Study
Phase I/II of Apollo trial was a 24-month prospective study on 75 patients with open-angle glaucoma, in whom one eye received topical bimatoprost once-daily dosage and fellow eye received bimatoprost sustained-release implant. Bimatoprost implant showed favorable efficacy and safety profile up to 24 months and was non-inferior to topical bimatoprost.
Topical bimatoprost has few well-known complications like eyelash growth, conjunctival hyperemia, cystoid macular edema, and peri-orbitopathy. Phase 1 and 2 of the ARTEMIS study does not show any sign of eyelash growth or peri-orbitopathy in the follow-up. The Bimatoprost sustained-release group did not report any case of endophthalmitis. The conjunctival hyperemia was more in sustained-release (DURYSTA) group for the initial 2 days but had fewer rates beyond 2 days compared to the topical bimatoprost group. Cystoid macular edema was noted in the sustained release bimatoprost implant group also seeking careful insertion of the implant in pseudophakic and aphakic patients. The endothelial density was slightly less at the end of 2 years than the topical group. The Bimatoprost SR group had a higher rate of non-specific adverse effects like eye pain, eye irritation, lacrimation, and conjunctival hemorrhage.
Duration of Action & Ideal Candidates
The implant is designed to last for 4-6 months but the effect lasts longer than expected. Phase 1 and 2 showed a drop in IOP of 40%, maintained for nearly a year. At this time FDA has only approved Bimatoprost SR (DURYSTA) for single-use per eye, although the phase 3 trial is collecting data for multiple administrations.
The FDA has currently approved for administration in open-angle glaucoma cases and ocular hypertensives. Careful administration is required in aphakic and pseudophakic patients. The effect seems to be optimal in patients with a history of good response to topical bimatoprost and no underlying corneal endothelial issues.
The sustained release bimatoprost is the newest member of glaucoma armamentarium and we hope that it will swing the future treatment paradigm.
