Introduction
The diagnosis of primary open-angle glaucoma is based on the triad of increased intraocular pressure, visual field changes and optic nerve head changes. Apart from the glaucomatous changes in the optic nerve head, the other two factors in isolation lack diagnostic sensitivity. In fact, studies have shown that careful evaluation of the optic nerve head has high specificity and that an experienced observer may be better in distinguishing a normal from a glaucomatous disc as compared to any other technology, like the HRT or OCT.[1-3] IOP can be normal in 50% of patients of glaucoma.[4] Studies by Quigley et al have shown that up to 40% of the axons could be lost before a visual field defect develops on Goldmann perimetry and that 20% of axons are lost before a 5 db loss is detected on standard automated perimetry.[5]So to pick up early or pre-perimetric glaucoma, in the absence of imaging, a sound knowledge of assessment of optic nerve head changes in glaucoma is imperative.Therefore, this article is written with the aim of describing the morphological changes of optic nerve head in glaucoma, discuss the techniques of clinical evaluation and the differential diagnosis one should keep in mind.
Techniques of Optic Nerve Head Evaluation
1. Direct Ophthalmoscopy: This is the simplest and the commonest method of disc evaluation[6] although it lacks stereopsis and the subtle signs may not always be very well appreciated. When using a Welch-Allyn instrument, the size of the disc can be assessed using the Gross’s Technique[7] wherein a 5° light aperture on the direct ophthalmoscope produces a circular spot with a diameter of 1.5 mm and an area of 1.77 mm sq., which is just about smaller than an average-sized disc.
2. Indirect Ophthalmoscopy: This gives a three-dimensional visualization but due to poor magnification often renders it ineffective in detailed visualization of the disc especially blood vessels, disc hemorrhages, and neuroretinal rim[8]. However, it has an advantage in eyes with hazy media.
3. Slit-lamp Biomicroscopy: It is the gold standard technique since it provides both good magnification and stereopsis. It can be done in the following ways:
Contact method using Goldmann three-mirror lens or a Zeiss four mirror lens (central portion).Non-contact method using a Hruby lens, +78D, +60D, or a +90D lens.While using a non-contact biomicroscopy method, the angle between the illumination and the magnification system should be in 0o (retro) or no more than 10o to ensure stereopsis. The image obtained from this a real, inverted, and laterally reversed one. To determine the size of the disc, reduce the height of the slit beam to coincide with the disk margins, and then read the measurements from the graticule on the slit lamp (shown below). This measurement would need to be corrected as the size of the image would depend on the magnification properties of the eye and the instruments [9,10]. The correction factor, when using different convex lenses (+60D, +78D or +90D), which needs to be multiplied with the reading on the slit lamp graticule to get the final diameter of the disc (in mm) is shown below (EGS guidelines).
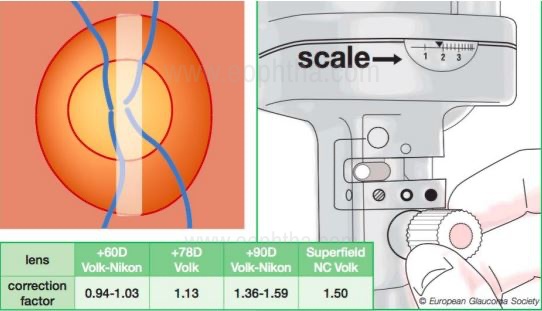
The 5 R’s of Optic Nerve and RNFL evaluation
Murray Fingeret in an excellent review article [11] described 5 major rules to diagnose the optic nerve head and retinal nerve fiber layer changes in glaucoma. He called it the 5R’s. It is unlikely that one can miss a diagnosis of glaucoma if a systematic approach as this is adopted to analyze the optic disc.
The 5 R’s are as follows:
- Observe the Scleral Ring to identify the limits of the optic disc and its size
- Identify the size of the Rim
- Examine the RNFL (Retinal Nerve Fiber Layer)
- Examine the Region of the Peri-papillary Atrophy
- Look for Retinal and Optic Disc Haemorrhages
Now, let's look at these 5 pointers one by one in greater detail.
1. Identifying the Scleral Ring and size of the Optic Disc
The optic disc margin is defined as the innermost border of reflective tissue that is internal to any pigmented tissue and within which only neural tissue is present [12,13]. Borders of the optic disk can be defined by looking at the thin white band encircling the disk. This white band is called the scleral ring. With the techniques described above, the size of the optic disc can be hence measured.
There is a large interindividual variability in the size of the so-called “normal” optic disc. Though the normal optic nerve head size is reported to be 1.5 mm in diameter it can vary from 0.96 mm to 2.91 mm. Even within the Indian population, the variability is clear from the different studies as enumerated below. The Caucasian population reports a range of 0.80 to 5.54 mm [14]
|
Study |
Mean Optic Disc Area |
|
Central India Eye and Medical Study (CIEMS) |
2.25 ± 0.51 mm2 |
|
Vellore Eye Study(VES) |
2.58 mm2. |
|
Andra Pradesh Eye Disease Study |
3.37 mm2. |
|
Chennai Glaucoma Study |
2.82 ± 0.52 mm2 |
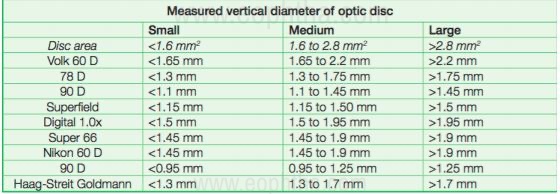
B. Medium-sized disc (1.5-2.5mm) with a medium cup;
C. Large disc (>2.5mm) with large cups
Clinically it is important to determine the size of the optic disc- whether its large, small or average. The size by itself doesn’t conclude anything, it only gives us a better relative assessment of the cup disc ratio (C:D ratio) and therefore of the neuroretinal rim(NRR). A small cup with a small disc may be abnormal whereas a large cup with a large disc may be normal. It is the relative C:D ratio and the contour of the NRR that is more important. Glaucoma tends to be over-diagnosed in large discs and often missed in small discs.
ONH diameter (mm) = (X/H) x D x C
(X = height of beam (mm); H = height setting on the beam height indicator (mm); D = diameter of the optic disc measured by the beam height indicator (mm); C = correction factor).
ONH area (mm2): r/4 X horizontal disc diameter X vertical disc diameter (r = the correction factor based on used lens diopter) [22,23]
NRR suggesting “pseudo-normal” glaucomatous mini cup;
Right: Large disc with the large cup but normal
NRR suggesting a “pseudo glaucomatous” cup in the large disc.
Optic Disc Shape
A normal optic disc is vertically oval The vertical axis is about 6% longer than the horizontal in Indian eyes[15] and 9% in a Caucasian population[14].
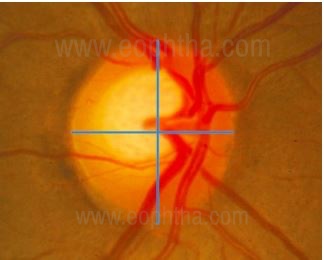
Evaluation of the Optic Nerve Head in Glaucoma.
J Curr Glaucoma Pract. 2013)
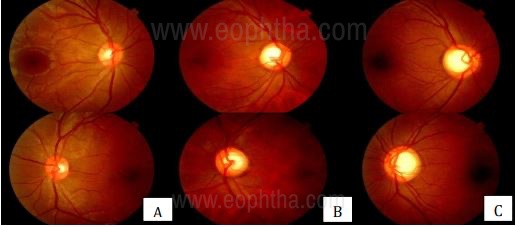
This ideal shape of the disc may not always be followed. Variations in disc shape can happen in myopes, astigmatism, amblyopia, congenital disc anomalies etc. In these cases, the assessment of glaucoma becomes tricky as the ISNT rule will not be followed and one may have to resort to investigations for diagnosis than relying on clinical assessment only. The following are a few examples.
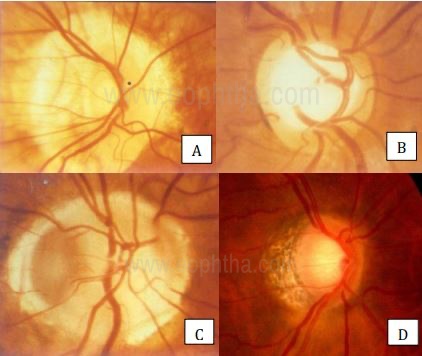
A. Myopic disc;
B. Dysplastic disc;
C. Congenital disc anomaly;
D. Tilted myopic disc.
Pic Courtesy: Dr. Ravi Thomas
Cup Disc Ratio
Cup Disc ratio is the ratio of the vertical cup diameter upon vertical disc diameter. Its relevance comes into play only when the size of the disc is defined along with. C:D ratio in itself doesn’t mean very much. A large cup may be normal in a large disc. A small cup may be abnormal in a small disc.
To define the margins of the cup there are two indicators. Firstly, on stereoscopic examination, there is a sharp drop off when the NRR ends and the cup margin starts. One has to understand that the cup is nothing but an empty space devoid of axons and reflecting its color due to the underlying lamina cribrosa. Secondly, focus on the course of the circumlunar blood vessel. It kinks at the cup-rim junction and then traverses on the NRR tissue before exiting the disc. The horizontal cup diameter is slightly larger than the vertical cup diameter.
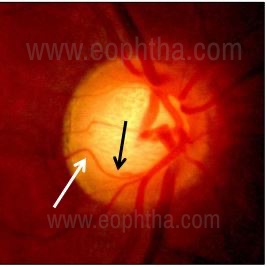
of the circumlinearblood vessels
delineating the cup rim junction.
The direction or point of deviation of small blood vessels on the surface of the ONH is used to determine the size of the optic cup (contour method) but not the area of pallor in the center of the optic disc (color contrast method) [25]
Once the cup margin is delineated, the vertical diameter can be measured in the same manner as the disc diameter with the help of the slit lamp graticule. The normal cup disc ratio varies with the size of the disc. Though the normal optic nerve head size is reported to be 1.5 mm in diameter it can vary from 0.96 mm to 2.91 mm.As a result, the physiological cup can be as small as 0.1:1 or as large as 0.8:1.
The C:D ratio should be an indirect indicator of the area and focal contour of the NRR, which is what will define the amount of glaucomatous damage. The European Glaucoma Society guideline also says that the use of CDR to classify patients is not recommended and the attention should be focused on the disc rim.
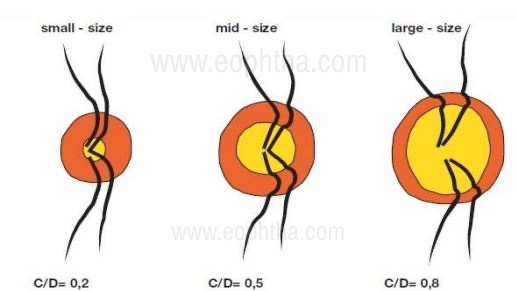
and the same number of retinal nerve fibers: small size disc (disc area less than
2 mm2 and C/D=0.3), mid-size disc (disc area between 2 and 3 mm2, C/D=0.5)
and largedisc (disc area greater than 3 mm2 and C/D=0.8).
Courtesy:European Glaucoma Society
2. Identifying the Neuroretinal Rim
Identifying the neuroretinal rim is the most important parameter in the assessment of glaucomatous damage. It is the pink or orange area between the disc margin and the cup margin all around. It is made up of 1-1.2 million retinal ganglion cell axons leaving the optic nerve head. In a normal disc, the inferior (I) NRR is thickest, followed by superior (S), nasal (N), and then temporal (T) being the thinnest. This is referred to as the ISNT rule, a violation of which greatly increases the suspicion of glaucomatous damage.
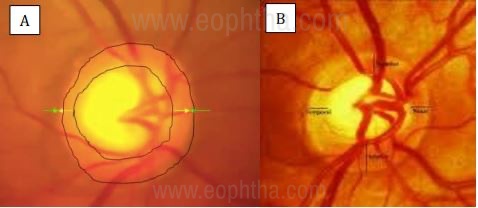
A- Area within the two circles is
the neuroretinal rim in a healthy disc ;
B- ISNT rule in a normal ONH.
(Pic Courtesy: Dr. Ravi Thomas)
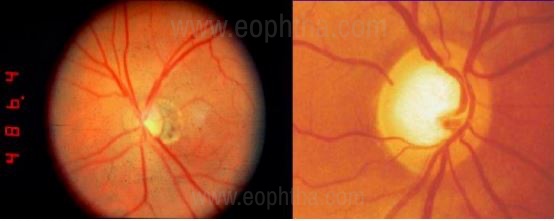
When evaluating NRR in glaucoma, the contour of the NRR is more important than the color. Figure 5 shows NRR pallor out of proportion to the cupping, suggesting non-glaucomatous neurological damage supported by the visual field findings.
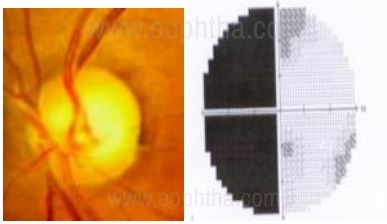
neurological field defect.
(Pic Courtesy: Dr. Ravi Thomas)
In Indian eyes, the mean neuroretinal rim area is also not interindividual constant. See the table below:
|
Study |
Mean Neuroretinal Rim Area |
|
Vellore Eye Study(VES) |
1.6 ± 0.37 mm2, |
|
Andra Pradesh Eye Disease Study (APEDS) |
2.8 ± 0.53 mm2 |
|
Chennai Glaucoma Study |
2.29 ± 0.39 mm2 |
Clinically, loss of NRR from its inner edge is a cardinal feature of glaucomatous disc damage. It can be a focal loss or a generalized one. The focal loss is more common inferotemporally and superotemporally so these areas should be carefully scrutinized for picking up early disease. A localized defect of NRR is called a NOTCH. A complete loss of NRR tissue in an area is defined as EXCAVATION. NRR notching usually does not occur in the temporal or nasal region. Temporal NRR notching without other glaucomatous optic disc changes is most likely associated with an optic disc insertion anomaly.
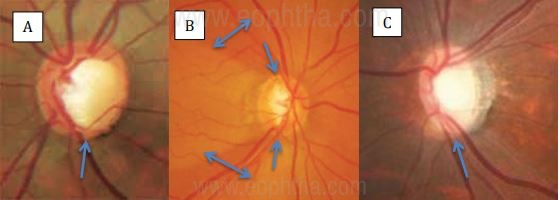
(Black Greg, Monitor or Medicate, Review of Optometry,2011) ;
B- Bipolar notch- superior and inferior with corresponding RNFL defects
(Gandhi M, Dubey S. Evaluation of the Optic Nerve Head in Glaucoma.
J Curr Glaucoma Pract. 2013);
C- Inferior Excavation (Pic Courtesy: Dr. Ravi Thomas)

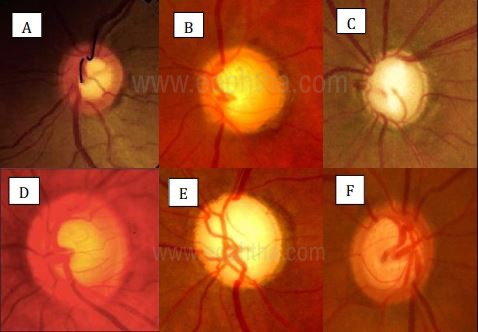
can be ancillary signs of glaucoma
: A- Bayonetting;
B- Inferior Saucerization;
C Nasalization Of Vessels;
D- Concentric Cupping;
E- Overpass Cupping;
F- Baring of Circumlinear Blood Vessels.
Disc Asymmetry
Disc asymmetry between the two eyes can be asymmetry in
- Size of the cup
- Width of the NRR
- Vessel course
- Peripapillary atrophy
Asymmetry of C:D ratio of 0.2 or less is present in 96% patients but the asymmetry of >0.2 with symmetrical discs and no anisometropia greatly increases the suspicion of glaucoma.
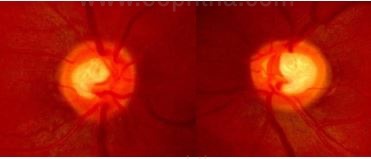
Asymmetry of CD ratio between the two eyes making
it a glaucoma suspect
(Rebolleda, G et al. Optic disc cupping after optic neuritis
evaluated with optic coherence tomography. Eye 2008)
3. Examine The Retinal Nerve Fiber Layer (RNFL)
RNFL defects are best visualized through a dilated pupil, at the posterior pole in the peripapillary region where they reach a critical thickness especially at the vertical poles of the optic nerve head in an arcuate fashion. RNFL defects are better appreciated using green light or red-free light on a slit lamp with a non-contact convex lens.
Normal RNFL is seen as bright white striations over the superior and inferior arcuate regions because it is the thickest in this area and reflects maximum light compared to the relatively darker background.
Patterns of RNFL defects:
Slit defect: These are very narrow defects emanating from the superotemporal or inferotemporal poles of the optic disc. Appears darker than the background retina on red-free light. Quigley et al [16] suggested that it could be the most sensitive parameter in glaucoma detection since it can preceed disc and visual field changes. Since it is very rare in normal individuals, the presence of slit RNFL defects greatly increases the suspicion of glaucoma.
Wedge defects: These defects flange out of the inferior and superior poles of the optic disc and fan out following the contour of the NFL layer in the arcuate regions. It represents the loss of many axons of ganglion cells of the corresponding area on the ONH and are very often associated with equivalent notching of the neural rim tissue on the disc and associated visual field defects
Diffuse defects: This is the commonest type of RNFL loss seen in glaucoma but clinically difficult to pick up due to loss of contrast with the background retina owing to broad generalized thinning of NFL layer over the arcuate regions.
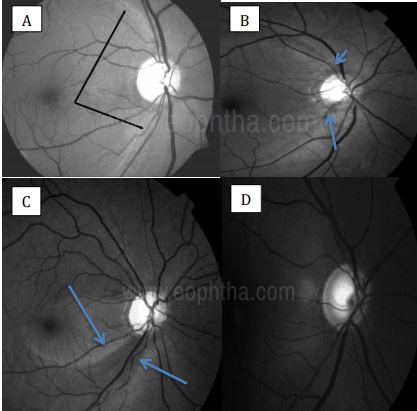
A. Normal RNFL as bright striations over the arcuate region;
B. Slit RNFL defects;
C.Wedge-shaped defect fanning out from the inferotemporal pole of the optic disc;
D. Diffuse RNFL defects.
Examination of the RNFL can detect early glaucomatous changes even before perimetry and is of importance in the detection of the pseudonormal glaucomatous mini cup in minidiscs[17] and classify an eye with pseudoglaucomatous normal large cup in a large disc. Differentiation of glaucomatous from nonglaucomatous also can be enhanced by the examination of the RNFL.
4. Examining the Region Of Peripapillary Atrophy (PPA)
ALPHA ZONE[Figure 14A, B] This is the outer zone of PPA which represents irregular hyper and hypopigmentation of the retinal pigment epithelium. It can be seen in normal eyes and is not specific to glaucoma. PPA is most commonly located in the temporal aspect of the disc.
BETA ZONE[Figure 14A, C] The inner beta zone is closer to the optic disc and is more remarkable due to the underlying visible sclera and large choroidal vessels. This is found more often in eyes with primary open-angle glaucoma and normal-tension glaucoma than in normal eyes. The location and extent of beta zone correlates with other disc changes and visual field changes. Its importance is especially apparent in small discs where a small cup may not unmask glaucomatous damage as well as an existence of the beta zone can. Hence, the presence of a beta zone should raise the suspicion of glaucoma but it should be in conjunction with other tests when making clinical decisions on diagnosis and management of glaucoma[3].
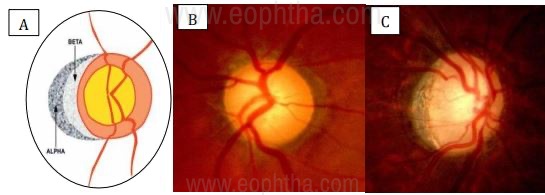
B- Alpha zone PPA;
C- Beta zone PPA
(Pic Courtesy: Dr. Ravi Thomas)
5. Retinal and Optic Disc Hemorrhages
The 5th or the final ‘R’ is to look for retinal and disc hemorrhages, also called Splinter, Flame-shaped or Drance hemorrhages. It may rarely be found in <0.2% normal. Prevalence in glaucomatous eyes ranges from 1.9 to 16.9% [18]. It is more common in patients with normal-tension glaucoma (5.9%) than primary open-angle glaucoma(4.3%). It is commonly located in the inferotemporal quadrant although can present anywhere around the disc. It can sometimes be the first sign of glaucomatous damage preceding RNFL defects, notching, or visual field defects. It can also result from large intraocular pressure fluctuations. It is a transient sign lasting anywhere from 2- 35weeks (average 10weeks), may disappear, and reappear again. The reappearance of drance hemorrhages either in the same or new location is indicative of progression.
Disc hemorrhages can be easily missed on clinical examination. They are better appreciated in red-free light and should be actively sought out for and the absence of it should be documented. One must note that the presence of drance hemorrhages in not only specific to glaucoma and can be seen in various other conditions like hypertensive or diabetic retinopathy, papillitis, venous occlusions, ischemic optic neuropathy etc.
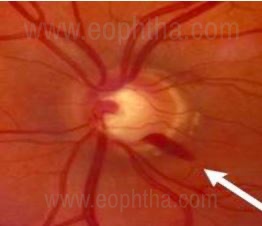
(Pic Courtesy: Dr Ravi Thomas)
Differential Diagnosis
It is of utmost importance to keep the differential diagnosis in mind in order to not miss a threatening neurological disc and to avoid overdiagnosis of glaucoma. Differentiating a glaucomatous from a non-glaucomatous disc can be quite often challenging as both may have cupped discs, along with the deceased diameter of retinal arterioles, focal arteriolar narrowing and reduced visibility of the RNFL[3,19]
The following are the warning signs that should alert the clinician to a different disease entity, other than glaucoma:
- Loss of visual acuity - If the visual acuity loss cannot be explained on the basis of other ocular conditions, suspicion of neurological conditions should arise.
- Rapid progression of field loss and visual acuity - This indicates one may be dealing with something else than glaucoma.
- Cup to field mismatch
- Hemianopic visual field loss - Indicates a neurological disease.
- Pallor more than cupping (band atrophy)
- Retinochoroidal venous collateral ("Optociliary shunt vessel") - may represent other pathologies like meningioma or old ischemic event.
Greenfield criteria consist of age less than 50 years, visual acuity less than 20/40, optic nerve pallor in excess of cupping, and vertically aligned VF defects.
- It can be used for the suspicious non-glaucomatous cupping in patients with normal pressure.
- It is recommended that patients who have one or more of the Greenfield criteria need to have neuroimaging performed to identify the causes of non-glaucomatous cupping.
- However, the absence of these criteria does not rule out the presence of non-glaucomatous cupping.[20]
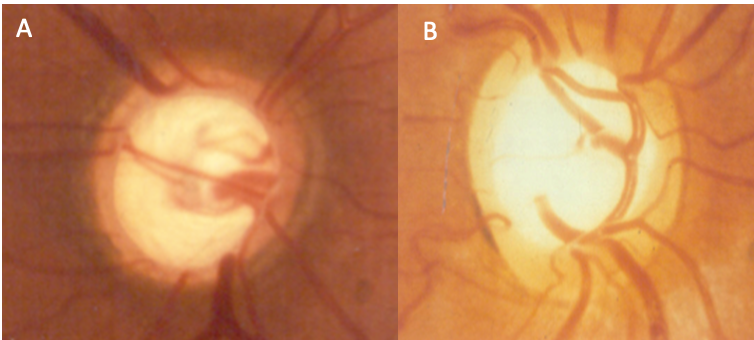
B- Dysplastic disc, note the anomalous vessels and funny shape of the disc.
(Pic Courtesy: Dr Ravi Thomas)
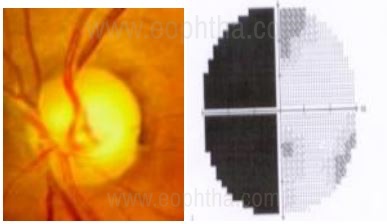
picture. The disc is cupped no doubt, but the pallor is
disproportionatly more than the cupping. The hemianopic fields
than respects the vertical meridian is proof of a neurological pathology.
Glaucomatous Vs. Non-Glaucomatous Optic Nerve Head Changes
|
FEATURES |
Glaucomatous optic nerve changes |
Non-glaucomatous optic nerve changes |
|
Visual acuity |
Affected late |
Affected early |
|
Color vision |
Not affected |
Defective |
|
Rim changes |
Rim loss/thinning present |
Rim pallor present |
|
Cup to fields mismatch |
Absent |
Present |
|
Visual fields |
Defects respect the horizontal meridian |
Defects respect the vertical meridian |
|
RAPD |
Absent unless unilateral advanced glaucoma |
Present in unilateral disease |
|
Peripapillary changes |
Suggestive of glaucomatous damage |
Not suggestive |
Staging of Glaucoma on the Basis of ONH Damage
Categorizing patients on the basis of the severity of optic nerve head changes is essential in the management and prognosis of glaucoma.
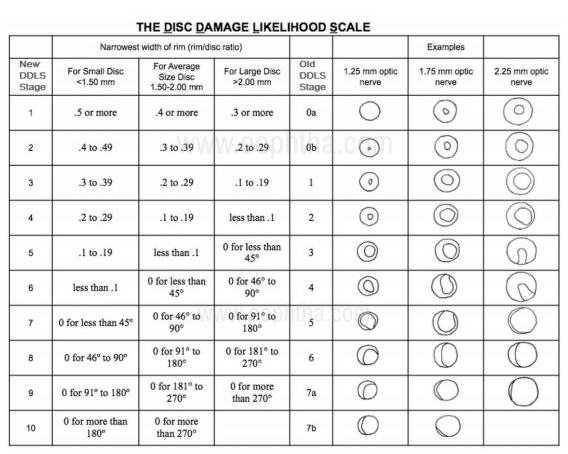
Many systems have existed namely the Armaly system, Read-Spaeth system, and Richardson system. The shortcomings of these systems lie in the fact that they take into consideration the cup/disc ratio which is not a valid indicator of disease. It assumes that the cupping starts centrally and progresses concentrically. Moreover, it does not take into account the size of the disc and by now we are well aware that the cup size varies with the disc size. The most widely accepted staging nowadays is the Disc Damage Likelihood Scale (DDLS) developed by Spaeth et al. [21]
DDLS allows quantification of the amount of damage that the optic nerve has sustained. Visual field loss will usually not occur before stage 5. And stages 6 to 10 strongly suggest aggressive treatment unless the glaucomatous progression has stabilized like in cases of steroid-induced glaucoma or traumatic glaucoma etc. The following table shows the DDLS nomogram taken from the original article.
Documentation of Optic Nerve Head Changes in Glaucoma
A systematic examination and documentation of findings will make an assessment of glaucomatous changes in the optic disc simple and enable picking up of subtle signs for early diagnosis.

Conclusion
A good stereoscopic evaluation of the optic nerve head with emphasis on changes of the neuroretinal rim and not estimation of the cup-disc ratio will aid in early diagnosis of glaucoma. The 5 R’s can be a useful checklist to incorporate into routine practice as it helps in sequentially looking for findings. Good documentation is key.
References:
1. O’Connor DJ, Zeyen T, Caprioli J. Comparison of methods to detect glaucomatous optic nerve damage. Ophthalmology Oct 1993;100(10)1498-1503 2. Caprioli J. Clinical evaluation of optic nerve in glaucoma. Trans Am Ophthalmol Soc 1994;92:589-641 3. Jonas JB, Budde WM, Jonas SP. Ophthalmoscopic evaluation of the optic nerve head.. Survey of Ophthalmology 1999;43(4):293-320 4.Leibowitz HM, Kruger DE, Maunder LR, et al. The Framingham Eye Study monograph: An ophthalmological and epidemiological study of cataract, glaucoma, diabetic retinopathy, macular degeneration and visual acuity in general population. Surv Ophthalmol 24 (suppl):335-610, 1980. 5.Quigley HA, Dunkelbarger GR, Green WR. Retinal ganglion cell atrophy correlated with automated perimetry in human eyes with glaucoma. Am J Ophthalmol 107: 453-464, 1989. 6. Spaeth GL. Direct Ophthalmoscopy In: Varma R, Spaeth GL,(Eds): The optic nerve in glaucoma, Philadelphia 1993, JB Lippincort. 7.Gross PG, Drance SM .Comparison of a simple ophthalmoscopic and planimetric measurement of glaucomatous neuroretinal rim areas. J Glaucoma. 1995 Oct; 4(5):314-6. 8. Shields MB, Tiedman JS. Binocular Ophthalmoscopic techniques for evaluation of the optic nerve head. In: Varma R, Spaeth JL,(Eds): The optic nerve in glaucoma, Philadelphia 1993, JB Lippincort. 9. Ansari-Shahrezaei S, Maar N, Biowski R, Stur M. Biomicroscopic measurement of the optic disc with a high-power positive lens.Invest Ophthalmol Vis Sci. 2001 Jan; 42(1):153-7. 10.Garway-Heath DF, Rudnicka AR, Lowe T, Foster PJ, Fitzke FW, Hitchings RA.Measurement of optic disc size: equivalence of methods to correct for ocular magnification. Br J Ophthalmol. 1998 Jun; 82(6):643-9. 11.Fingeret M, Medeiros FA, Susanna R Jr, Weinreb RN. Five rules to evaluate the optic disc and retinal nerve fiber layer for glaucoma.Optometry. 2005;76(11):661‐668. doi:10.1016/j.optm.2005.08.029 12.Strouthidis NG, Yang H, Reynaud JF, et al., Comparison of clinical and spectral domain optical coherence tomography optic disc margin anatomy,Invest Ophthalmol Vis Sci, 2009;50(10):4709–18. 13. Strouthidis NG, Yang H, Downs JC, Burgoyne CF, Comparison of clinical and three-dimensional histomorphometric optic disc margin anatomy,Invest Ophthalmol Vis Sci, 2009;50(5):2165–74. 14. Jonas JB, Gusek GC, Naumann GOH. Optic disc, cup and neuroretinal rim size, configuration and correlations in normal eyes. Invest Ophthalmol Vis Sci 29:1151-1158, 1988. 15. Jonas JB, Thomas R, George R, Berenshtein E, Muliyil J.Optic disc morphology in south India: the Vellore Eye Study. Br J Ophthalmol. 2003 Feb; 87(2):189-96. 16. Quigley HA, Reacher M, Katz J, Strahlman E, Gilbert D, Scott R. Quantitative grading of nerve fiber layer photographs.Ophthalmology. 1993 Dec; 100(12):1800-7. 17. Jonas JB, Fernandez MC, Naumann GO. Glaucomatous optic nerve atrophy in small discs with low cup-to-disc ratios.Ophthalmology. 1990 Sep; 97(9):1211-5. 18.Healey PR, Mitchell P, Smith W, Wang JJ.Optic disc hemorrhages in a population with and without signs of glaucoma.Ophthalmology. 1998 Feb; 105(2):216-23. 19. Airakinsen PJ, Tuulonen A, Werner EB. Clinical evaluation of the optic disc and the retinal nerve fiber layer. In: Ritch R, Shield MB, Krupin T: The Glaucomas(Basic Sciences), Vol 1 2nd edition, St Louis, Missouri: Mosby Inc 1996;617-58 20.Greenfield DS, Siatkowski RM, Glaser JS, et al., The cupped disc. Who needs neuroimaging?,Ophthalmology, 1998;105:1866– 74. 21.Zangalli C, Gupta SR, Spaeth GL. The disc as the basis of treatment for glaucoma. Saudi journal of ophthalmology. 2011;25(4):381–7. pmid:23960952 22. Lim CS, O’Brien C, Bolton NM, A simple clinical method to measure the optic disk size in glaucoma,J Glaucoma, 1996;5:241–5. 23.Bhartiya S, Gadia R, Sethi HS, Panda A, Clinical evaluation of optic nerve head in glaucoma, J Curr Glaucoma Pract, 2010;4:115–32.
