Know your cutter and machine.
Current generation vitrectomy cutters work by guillotine mechanism where vacuum holds the Vitreous fibers and presents it to guillotine blade that severs it in one swift motion without exerting significant on fibers. The cutting process needs a fine balance between the speed of cutting and the “pull” created by the vacuum. Higher cutting rates like 10000 cpm allow the surgeon to use a higher vacuum in the range of 600 mm Hg. The rate of vitreous removal also depends on duty cycle and bore size of lumen. Surgeons should test the efficiency of cutters either on cotton fibers or on the vitreous prolapsing out of sclerotomies. One has to learn to recognize when the cutter is pulling without cutting – the most dangerous situation. The surgeon should also familiarize himself with different functions of vitrectomy systems like air injection, Endodiathermy, venturi or peristaltic pumps, light source/s, Phaco Fragmatome, and Viscous fluid injection. It also helps to know about Cassettes and settings and the specifications for air compressor or the gas cylinder.
Little brush up with Surgical Anatomy
Pars plana
All beginners need to know that Ora serrata is 6 mmm from limbus nasally and 7 mm temporally. Conventionally Sclerotomies are to be created through Pars Plana at 3.0mm, 3.5mm, and 4.0mm from the limbus in aphakic, pseudophakic and phakic eyes respectively. (Figure 1) One has to be more cautious nasally where pars plana is narrower. Pediatric eyes have much narrower pars plana and that’s why sclerotomies are created even more anteriorly. The axial length of an emmetropic eye is 23 mm and the length of the globe from sclerotomy to the retina is lesser than that. Most vitrectomy instruments like cutter, endo-light pipes, and forceps are longer than that. This knowledge makes it easy for Surgeons to approach the posterior segment for different maneuvers but also cautions them to keep holding instruments in a stable position lest these instruments accidentally graze retina and inflict inadvertent injury.
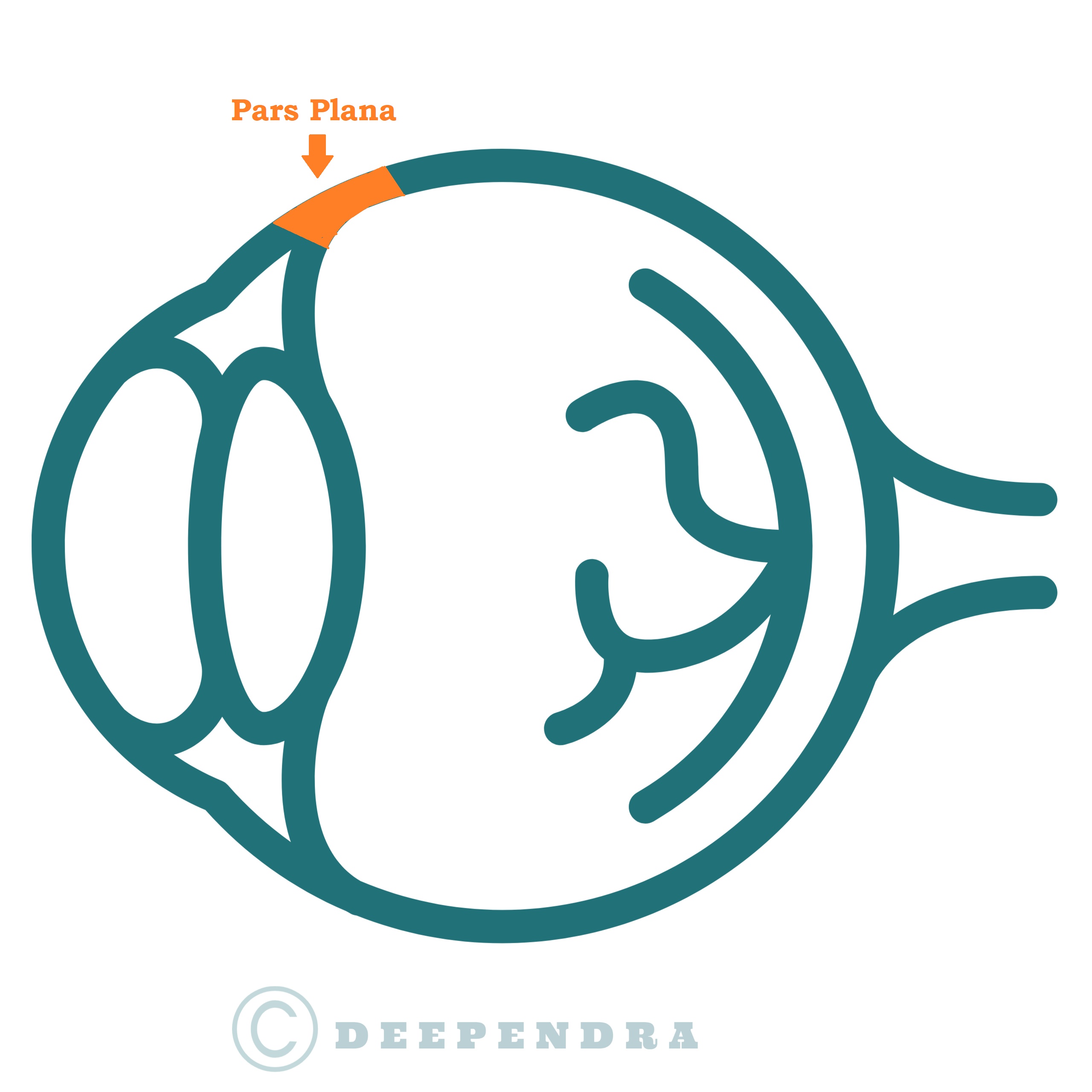
Schematic diagram of Human eyeball showing the location of pars plana. That’s the area through which we make our sclerotomies.
Vitreous body
Vitreous microfibers are composed of hyaluronic acid-collagen (Type-2 predominantly) complex that aggregates collagen fibrils into bundles that run in anterior-posterior orientation from pre-macular vitreous cortex to vitreous base. Vitreous has stronger attachments to disc margins, retinal scars, edges of lattice degeneration, anterior edge of retinal breaks, and fibrovascular or neovascular fronds.
Choosing your initial cases
Because vitreous surgery is performed for a wide variety of retinal diseases and asks for so many additional maneuvers besides vitrectomy, it’s important for a beginner to choose their initial cases more cautiously. Pseudophakic Eyes with retained lens matter, vitreous hemorrhage from vascular occlusions are good examples. If one is in institutional setups with other senior retina surgeons available in the team, then silicon oil removal in an eye with a low probability of retinal re-detachment is also a good case to start with. After a few cases, one can proceed to more complex situations like retinal detachment, macular surgeries, and then diabetic retinopathy.
How to start? – Getting ready, Anesthesia, and making sclerotomies
The surgeon should ensure that the following things are in working condition; Cutting and Vacuum, Airflow (test it in a bowl of water), Endodiathermy, Viscous fluid injection, Laser (test the endo-probe and firing). Most vitrectomies for adults are done under local peribulbar anesthesia. Because incisions for small gauge surgery are made transconjunctivally, one should avoid large sub-conjunctival blebs. These blebs can pose problems with conjunctival displacement and also create a crater on the cornea where fluid can collect and hamper visualization. If one gets blebs, it's better to drain it before making sclerotomies.
Sclerotomies
Site
For standard 3 port vitrectomy, we create 2 sclerotomies on the temporal side and one on nasal. These sclerotomies are created just above and below the horizontal meridian (3 O’ clock and 9 O’clock). (Figure-2 & 3). Sclerotomies at this position give maximum maneuverability of instruments and also prevents infusion cannula from interfering with the rotation of the globe.
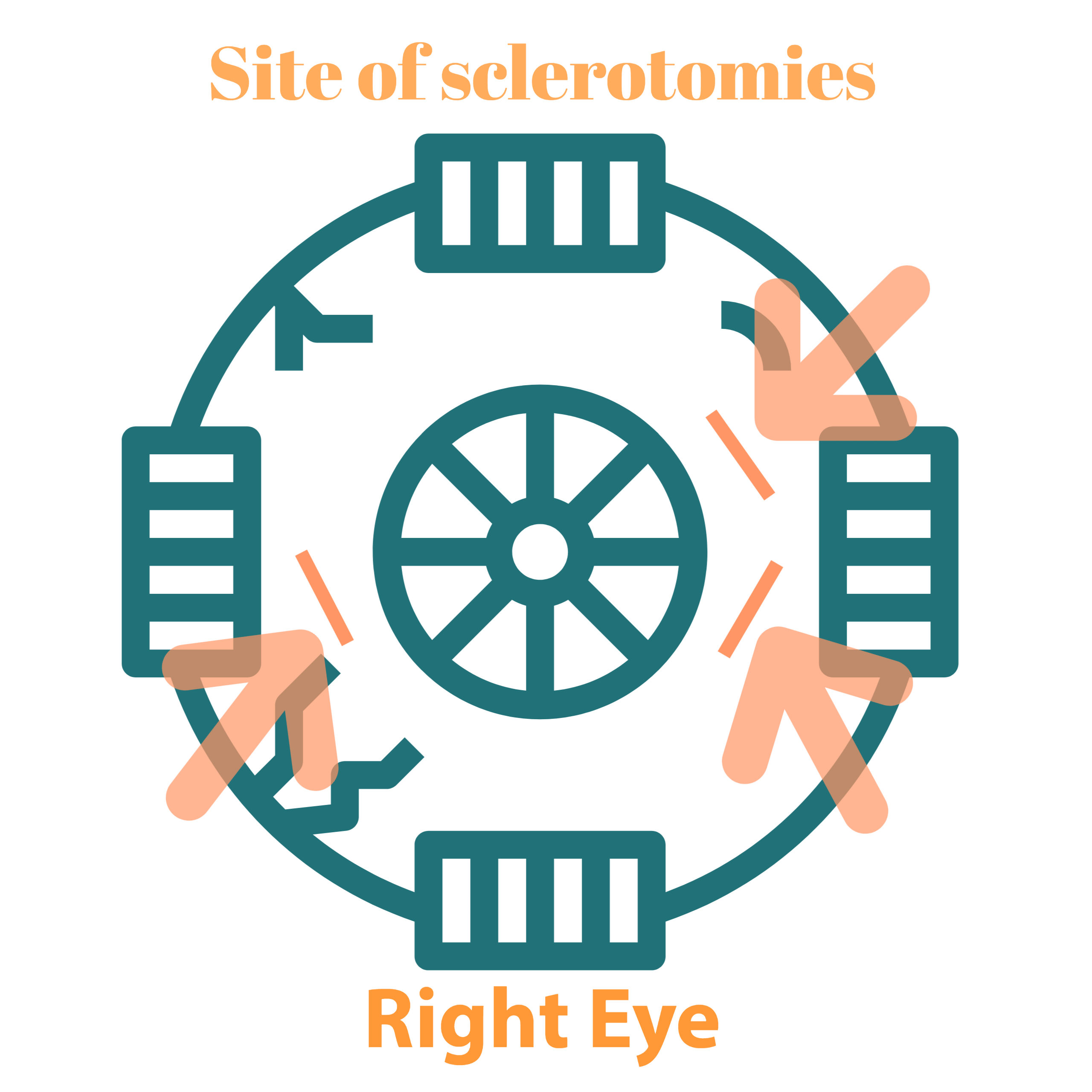
Diagram showing the sites where sclerotomies are conventionally placed.
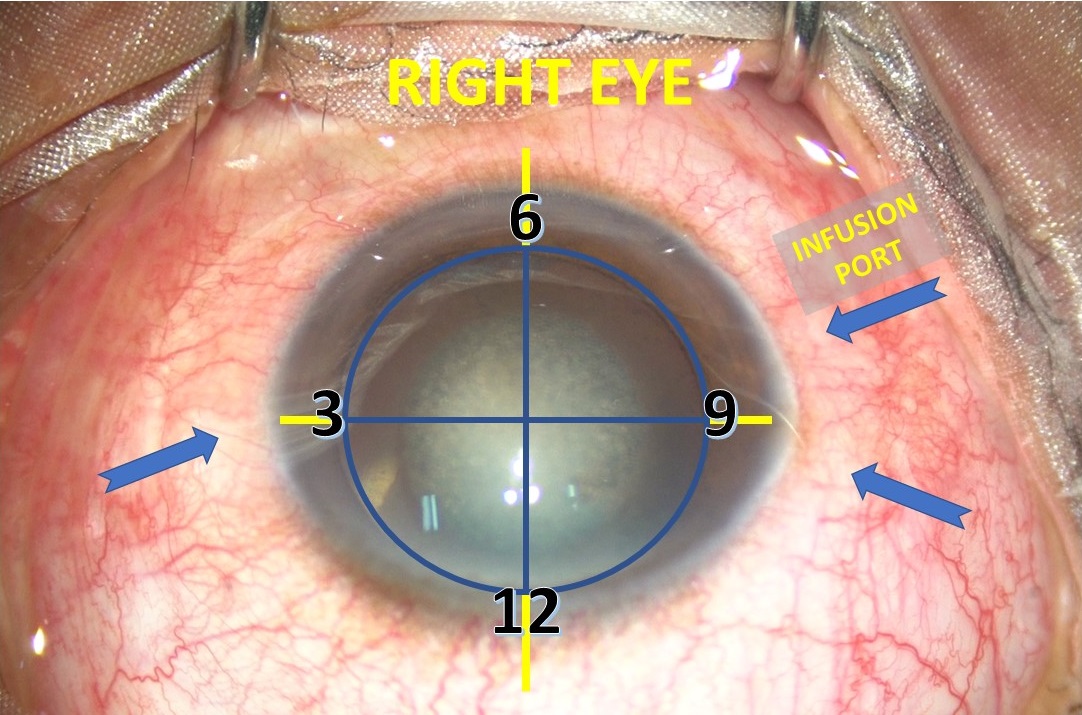
Intraoperative photograph of OD showing the potential sites for 3 sclerotomies
Sequence
The sclerotomy for infusion port (inferior-temporal) is created first unless there is choroidal detachment or a leaking incision from phacoemulsification.
Technique
The preferred technique for sclerotomy by 25 or 27 G trocars is the 3-step approach.
- Measure your site (3-3.5mm from limbus) and hold conjunctiva with toothed forceps and displace it towards limbus.
- Keep your trocar parallel to the limbus and at a very small angle to the globe (30 to 40°).
- Make an angled entry through 80% depth of sclera.
- After you have crossed 80% of scleral depth, change the angle of the trocar and make it perpendicular to the globe (90°) and enter the vitreous cavity. The direction of the trocar should be towards the optic disc. Note, it’s easier to hit crystalline lens than to hit retina during this process, so keep the direction of your trocar away from the lens as much as possible.
- Now, hold the cannula with forceps and withdraw the trocar slowly. If you find some resistance, then you can do little twisting (rotatory) movements of the trocar while pulling.
- After removing trocar, the infusion cannula with fluid turned on is fixed at first sclerotomy.
- In cases with thick choroid or choroidal detachment, the light pipe can be introduced into infusion port first to confirm that cannula is in the vitreous cavity and not in subretinal position.
- Rest 2 sclerotomies are created in a similar manner.
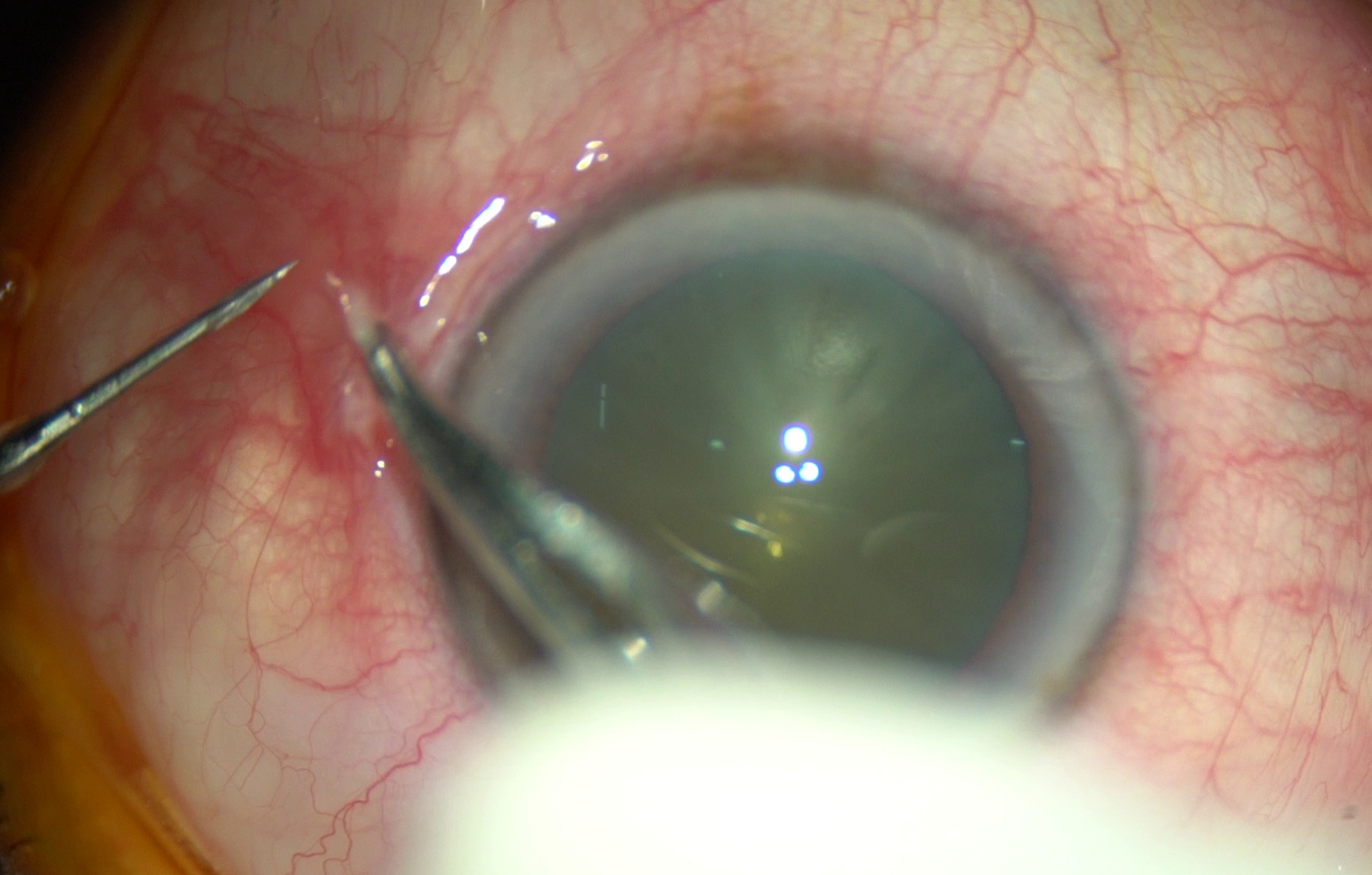
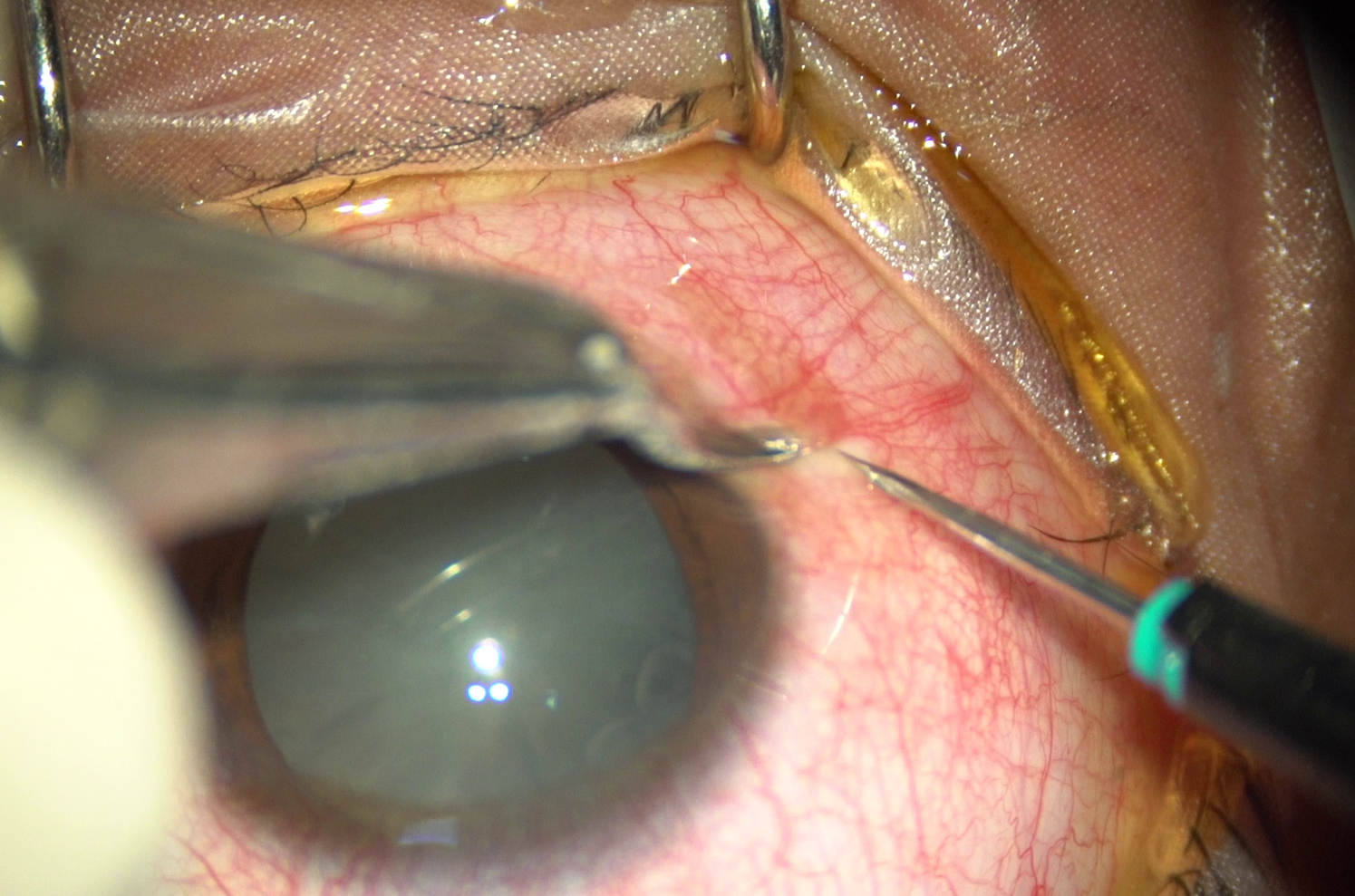
Intraoperative photograph of OD showing conjunctival displacement towards the limbus

Schematic Diagram showing how we direct trocar towards optic disc during making sclerotomy. This helps us avoid lens injury.
How to perform a Core vitrectomy?
It’s always good to test your cutter on the prolapsed vitreous that pops out of sclerotomies. This also reduces the traction on the vitreous base when you shove the instrument inside the vitreous cavity. The light pipe and cutter are then introduced into the vitreous cavity. The direction of movement should always be towards the optic disc and slide them forward till you can see them in the mid vitreous cavity. Bring your viewing system into an active position and focus it on the cutter in the mid vitreous cavity. Start clearing the vitreous close to sclerotomy ports first. In cases where retinal is not visible, one can start nasally or in the areas where partial PVD can be seen. In cases with thick PVD with vitreous hemorrhage or exudates, it often helps to create an opening in posterior hyaloid in the nasal area and look for retinal vessels after aspirating subhyaloid blood or exudates. This opening is then enlarged with the cutter. To avoid lens injury in eyes with a clear lens, beginners need to be aware of the curvature of the posterior surface of the crystalline lens. To approach the retinal periphery, one should rotate the globe and avoid crossing the posterior pole of the lens and approach it from the sides of the lens. Changing your hands is also a good strategy to avoid lens injury. Thick blood adherent to anterior hyaloid can be carefully cleaned in the center leaving the peripheral rim undisturbed. This peripheral rim of blood-stained anterior hyaloid is harmless and gets absorbed spontaneously.
How much vitrectomy?
The extent of vitrectomy is determined by the indication of vitrectomy, retinal pathology, lens status, and goals of the surgery. Cases with retinal detachment need a near-complete vitrectomy with the clearing off as much vitreous as possible especially over retinal tears and all predisposing lesions like lattice degeneration. In a phakic eye with vitreous hemorrhage from RVO, one need not be aggressive to clear the vitreous base. Most cases need induction of posterior vitreous detachment but the same may not be done for eyes with PDR or endophthalmitis. The detailed discussion of other vitreoretinal maneuvers is beyond the scope of this chapter.
Inducing Posterior Vitreous Detachment
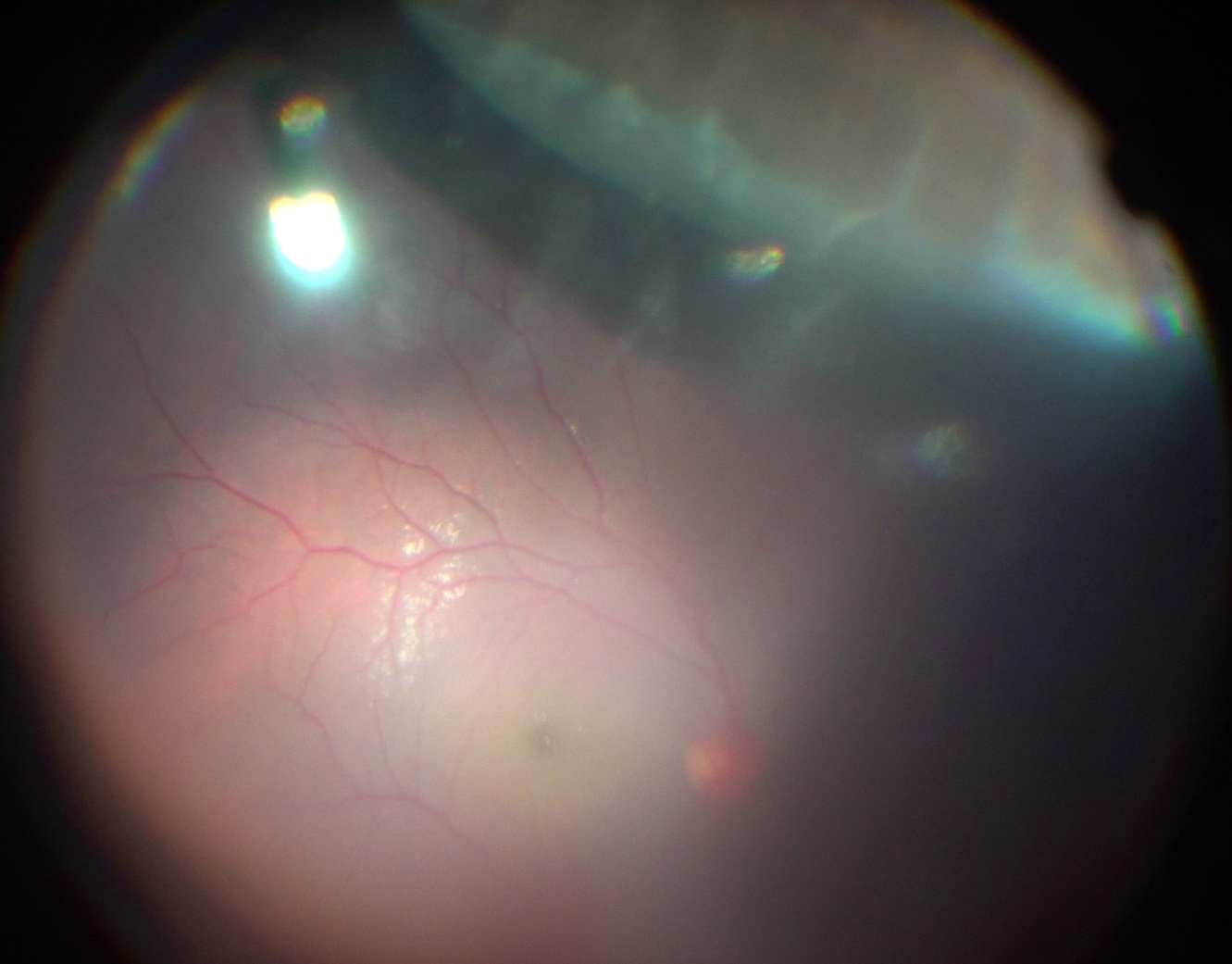
This is a critical step during vitrectomy. The technique described here is for all cases with the attached retina except those having endophthalmitis or active NV fronds. Although PVD induction can be done without it, it is facilitated by using triamcinolone acetate. Triamcinolone crystals enhance visualization of posterior hyaloid, especially over the posterior pole. Posterior hyaloid is firmly attached to disc margins and that is the preferred site to initiate PVD induction. PVD can be induced with either active or passive suction. For most eyes, it can easily and safely be done with 25 G cutter. A soft silicone tip attached to an active vacuum is a good alternative. The cutter is placed at a superior or inferior disc margin with “cutting switched off” and vacuum is raised in a linear fashion to engage the posterior hyaloid. Once a Weiss ring of posterior hyaloid is visible as being lifted off the disc, the vacuum is lowered and rest of the process can be completed with minimal or zero vacuum. It is advisable to extend posterior hyaloid separation in all quadrants to the point it keeps separating easily. Forceful extension of PVD beyond equator is not desirable especially in high myopes and diabetics where it can induce retinal breaks. For most eyes, it will automatically extend till ora serrata with passive lifting only. After PVD has been completed, the separated posterior hyaloid is removed with cutter on highest cut-rate and lower vacuum. Following this surgeon would go on to complete rest of the maneuvers depending upon retinal pathology to be treated.
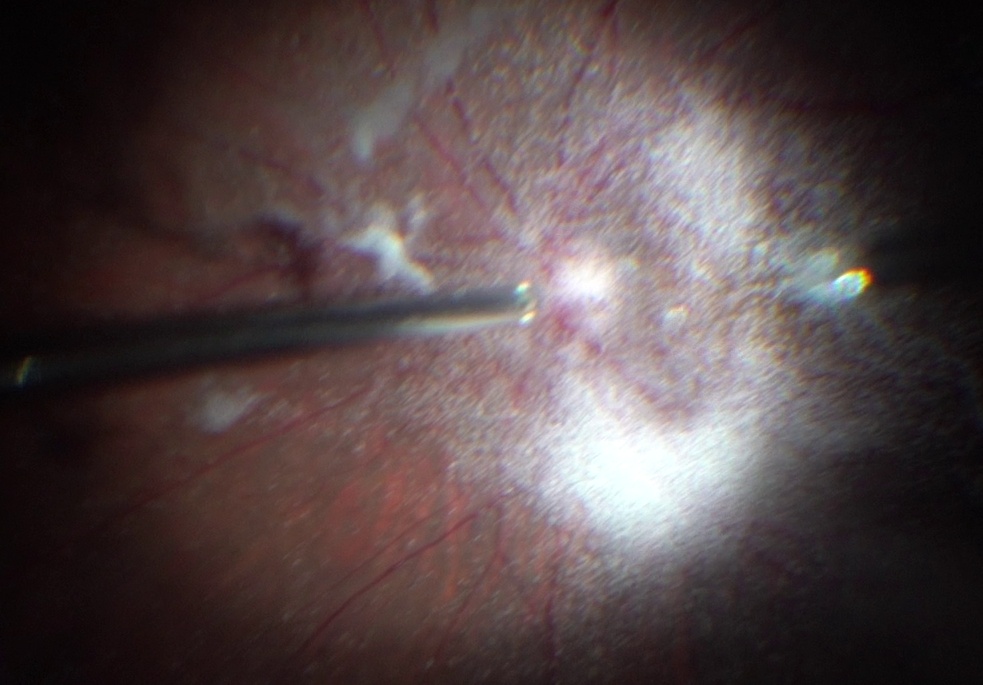
Intraoperative photograph showing nicely visualized posterior hyaloid by triamcinolone acetonide.
Intraoperative photograph showing posterior hyaloid being elevated by passive vacuum.
Periphery inspection and indentation
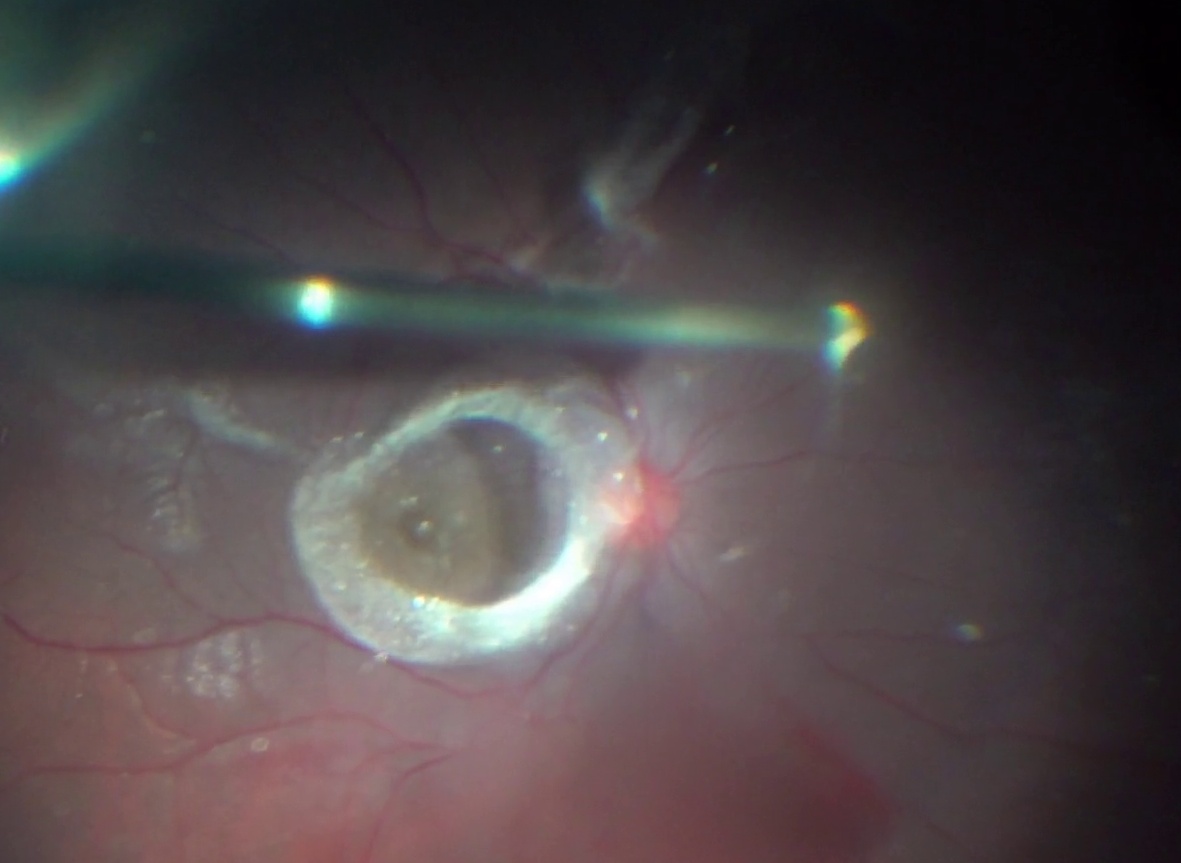
All eyes that have undergone vitrectomy need a careful examination of retinal periphery to detect any retinal breaks or dialysis. This can be easily done utilizing Wide angle viewing and peripheral indentation. Surgeon or assistant can indent with a cotton bud or any blunt instrument like a muscle hook. Muscle hook offers advantage of a long handle allowing easy maneuverability and larger area on contact with sclera, which in turn makes this inspection process very fast. Indentation can also be facilitated by reducing intraocular pressure to 25 mm Hg. If any retinal break is discovered during indentation, same can easily be lasered over the indent created by muscle hook or cotton bud.
Intraoperative photograph showing intraoperative scleral indentation to inspect retinal periphery for any retinal breaks
Fluid-Air Exchange and Choice of Tamponade
Choice of tamponade will depend on retinal pathology and other non-ocular factors like status of fellow eye, air travel. Most Surgeons would do a fluid-air exchange (FAE) for all eyes even if no tamponade is being planned to prevent postoperative hypotony. Eyes undergoing a combined surgery with scleral fixated IOLs, can be closed without FAE if integrity of sclerotomy wound is good or one decides to place a suture to avoid postop hypotony. The eyes undergoing silicon oil removal also need to undergo FAE to let all residual silicon droplets float on the top of fluid, where they can easily be aspirated by active vacuum and thus ensuring complete silicon oil removal. If Surgeon is planning to use Gas or Silicon oil Tamponade, same is infused after FAE.
Endolaser Photocoagulation
Although it is not needed for all vitrectomies, this is a very important step. Most Surgeons now use frequency-doubled YAG (Green) laser for vitrectomy but diode laser can also be used. Endolaser photocoagulation is utilized either to ablate ischemic areas or for retinopexy and occasionally for hemostasis. One can also use Endolaser to treat polyps or benign tumors or feeder vessels if these lesions are detected intraoperatively. The technique of Endolaser photocoagulation is similar to what we do for our outdoor patients with some key differences;
- Spot size cannot be changed much (some change is possible by altering the distance of the probe from the retina)
- The intensity of laser burns gets enhanced if it’s done under air or silicon oil or if it’s done over a buckle indent, so start with lower power and duration settings.
- Indentation can bring inaccessible areas into view and is a huge advantage. Similarly, curved probes can also help us reach the extreme periphery of the retina and also help us avoid inadvertent injury to the crystalline lens.
- Beginners also need to realize that failure to achieve a good burn can be due to multiple reasons like persistent subretinal fluid, gel vitreous in contact with the tip of the probe, a damaged or worn out probe and also if your laser beam is not perpendicular to the retinal surface.
Closing the sclerotomies
Closure of sclerotomy ports needs a cautious approach to prevent post-op hypotony and reduce the risk of infection. It’s always good to remove cannula over a solid instrument like a light pipe to avoid any vitreous “wick” getting trapped in the track. One can also tilt the endo light pipe while sliding cannula over it, the direction of tilt should be the same as was during creating sclerotomies. After cannula has been pulled out of the eye, the solid instrument is slowly withdrawn from the eye and the sclerotomy wound is pressed with a cotton bud. One needs to apply firm pressure on the anterior lip of wound for 1 minute at least and then the conjunctiva is retracted back over the wound. The same process is followed for the second port. For infusion port, the cannula and infusion tubing is pulled off in one go followed by firm pressure on sclerotomy wound. It’s also advisable to check IOP (palpation with finger) before removing the last (Infusion) port. Sclerotomies for eyes receiving silicon oil tamponade should be carefully inspected for any leaks. If there is persistent leakage that does not stop even after 3-4 minutes of firm pressure, then one should not hesitate to apply one suture in order to avoid subconjunctival silicon bubbles or silicon oil underfill postoperatively. Most surgeons prefer a single 8-0 Vicryl (Absorbable) suture passed transconjunctivally for suturing 25G ports.
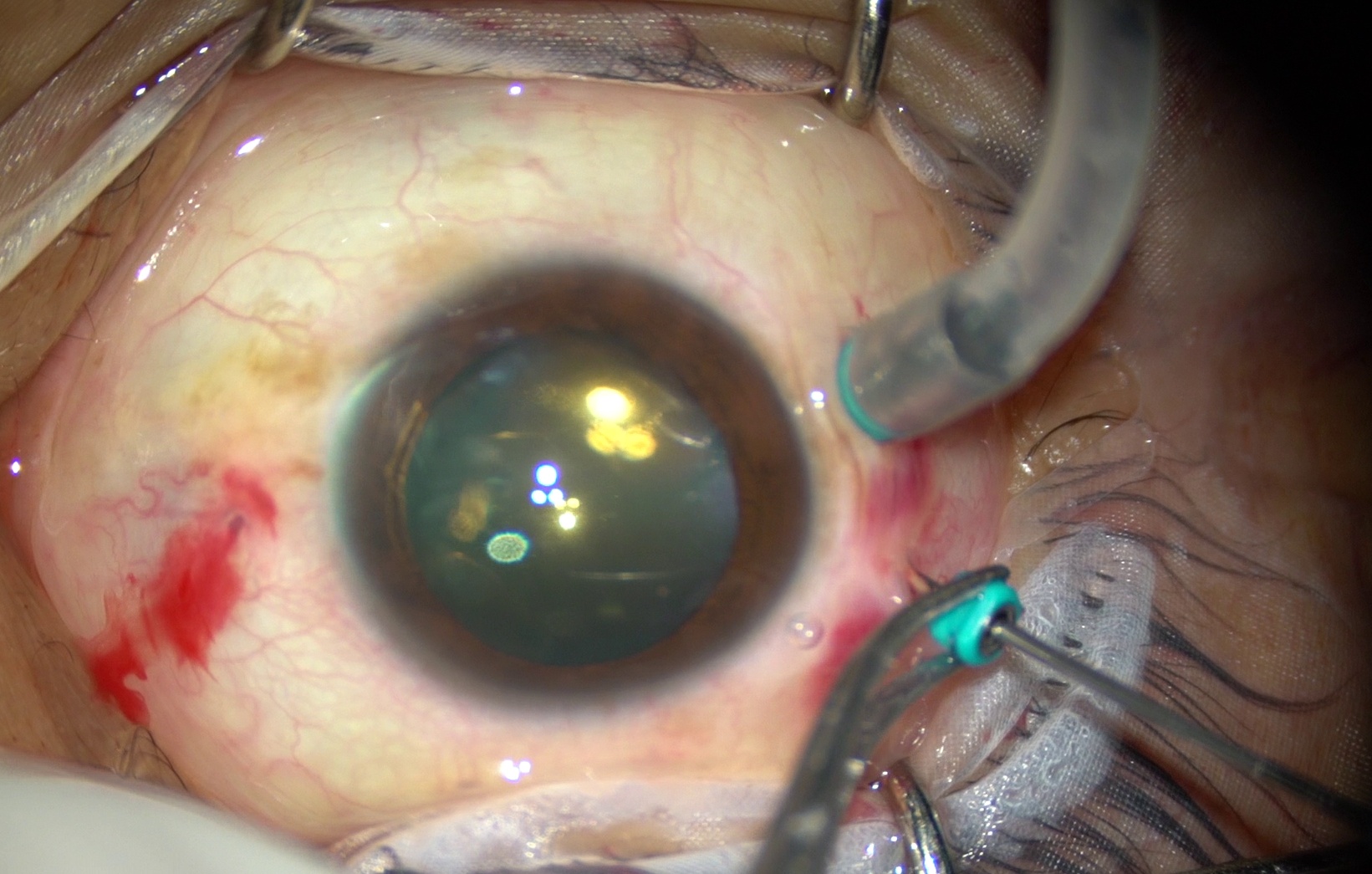
Intraoperative photograph showing how cannula is pulled over a light pipe to avoid vitreous wick.
Post-op care
Patients need to be instructed upon postoperative positioning, eye hygiene, schedules for eyedrops and other medicines. Most Surgeons would like to prescribe an antibiotic drop for 7 days, steroid drops in tapering doses for 4 weeks and a cycloplegic for 3-5 days. Patients with air or gas bubble in vitreous cavity should be warned against air travel.
