Modern imaging modalities in the field of the retina have expanded our understanding of common pathologies including diabetic retinopathy (DR), age-related macular degeneration (AMD), central serous chorioretinopathy (CSCR), and many rare retinal diseases such as dystrophies, which were poorly understood before.
This chapter describes newer emerging techniques in retinal imaging which enables advanced screening, monitoring and provide objective measures for treatment monitoring.
MULTICOLOUR IMAGING
Compared to standard fundus cameras, multi-colour images produced by the scanning laser ophthalmoscope(SLO) have a higher resolution due to reduced light scatter and allow image acquisition at different planes and higher contrast through a non mydriatic pupil using different wavelengths. SPECTRALIS Optical Coherence Tomography SDOCT(SPECTRALIS SD-OCT, Heidelberg Engineering,Heidelberg, Germany) uses the confocal scanning laser ophthalmoscope(cSLO) to capture three simultaneous reflectance images using three monochromaticlaser sources and these different wavelengths of light which penetrate the retinal surfaces at different depths to demonstrate details at the various layers of the retina.1
(a)Blue reflectance (BR; 488 nm)– This enables better detail of the inner retina and the vitreoretinal interface such as epiretinalmembranes, retinal nerve fiber layer thinning and macular pigment changes.
(b) Green reflectance (GR; 515 nm) – This enables details of deeper layers such as retinal blood vessels and intra retinal lipid exudation.
(c) Infrared reflectance (IR; 820 nm)– This helps to visualize structures at the level of the outer retina and choroid includes drusen,retinal pigmentary epithelium alterations.
Multicolor imaging application is found to be beneficial in following conditions.
A.Reticular pseudodrusen–Reticular drusen is associated with all stages of AMD that may represent a high-risk for progression, can be imaged more distinctly.2-4It appear as a central hyper reflective lesion with a hypo reflectiveborder.
B.Geographic atrophy (GA) -The boundaries of GA are more clearly demarcated on multicolor imaging.
C.Polypoial choroidal vasculopathy (PCV) –PCV lesions appears as an orange-red nodule in colour fundus photo. In contrast to multicolor imaging wherethe lesion appears darker red with a greenish tinge..This is likely due to the location of lesion deep under the retinal pigment epithelium, in contrast to more superficial subretinal bleeding. Hence, it enables us to distinguish lesion at different layers of the retina, although this finding will need to be verified in larger studies.
D.Diabetic retinopathy(DR) - Standard field stereoscopic colour fundus photos proposed by the Early Treatment of Diabetic Retinopathy Study (ETDRS) group is cumbersome for widespread screening for DR. Multicolor imaging in combination with spectral-domain optical coherence tomography (SD-OCT) has the potential to improve detection rates of diabetic eye screening programs with more ease. It also helps in easy identification of residual fibrotic tractional epiretinal membrane, and new vessels at the disc and diabetic macular edema (DME) in the absence of hard exudates.5, 6
E. Retinal Vascular Occlusions–Multicolor imaging helps in accurate documentation of disease progression and the response to treatment.
F. Central serous choreoretinopathy- The boundaries of the subretinal fluid pocket and the multifocal areas of neurosensory detachments in CSCR are seen more distinctly on multicolor imaging.(figure1) It detects subtle changes in the retinal structure which may not be clinically apparent after the resolution of the subretinal fluid from CSCR.
G. Inherited Retinal Dystrophies: Dystrophies such as Stargardt’s disease where demarcation of the central area of atrophy is more distinctly seen and correlates better with fundus autofluorescence. Pseudoxanthoma elasticum complicated by a choroidal neovascularization where in the early signs such as haemorrhages can be detected much easily on multicolor imaging.
H. Choroidal tumors- Pigmented lesions such as choroidal nevus,choroidal melanomas show a varying degree of colour with better delineation of the depth of the lesion, the degree of pigmentation and surrounding changes such as blood, fluid or atrophy.
Limitations of multicolor imaging
Limitations include a longer period of fixation which is required to take the three separate images. Artefacts including ‘ghost’ maculopathy7with no actual pathology are not uncommon. Conventional color fundus photos can be sometimes more comparable to clinical examination, in features such as collateral and sclerosed vessels in vascular occlusion.
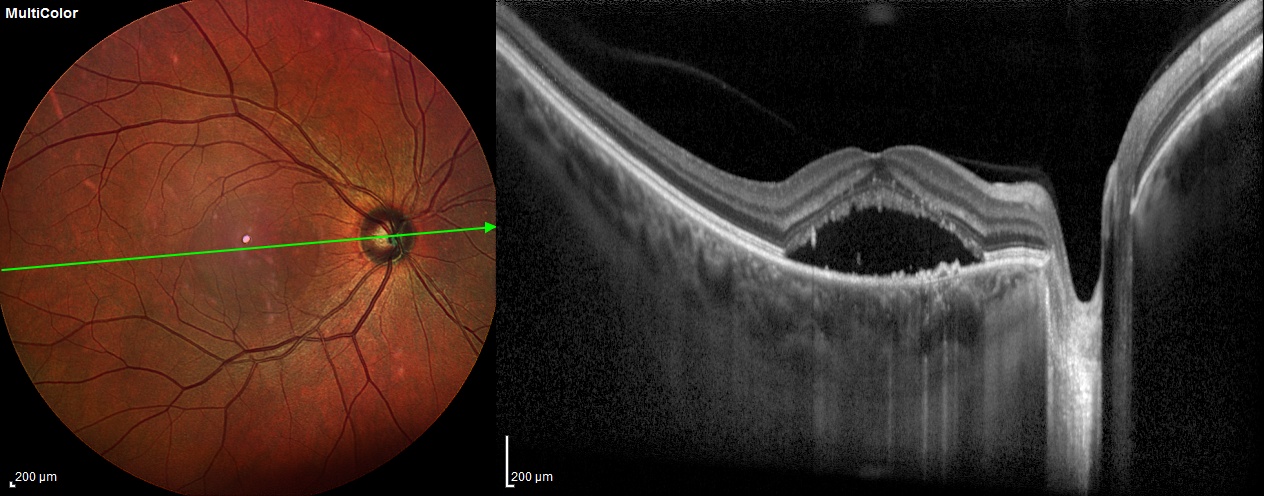
Figure 1. Multicolor imaging of an eye with central serous chorioretinopathy showing well-defined subretinal fluid pocket (left panel) corresponding to the neurosensory detachment on swept-source optical coherence tomography scan (right panel)
ULTRAWIDEFIELD (UWF)IMAGING
UWF imaging (Optos camera (Optos 200Tx, Dunfermline, UK)is based on SLO with two wavelengths of laser light[green (532 nm) and red (633 nm)] to recreate a composite pseudo-colour image of the posterior segment along with the use of a large ellipsoid mirror, through which a 200-degree field of view can be obtained in an undilated pupil. With UWF imaging, approximately 80 % of the total retinal surface area can be visualized. It can also capture wide-field fundus autofluorescence, and fluorescein/indocyanine green angiography.8
Diabetic Retinopathy –UWF angiography provides a more comprehensive assessment of the extent of a retinal disease process, and may lead to the detection of abnormalities that may alter treatment plans and also give new insights into the pathogenesis of DME.UWF imaging helps in detecting subtle neovascularization (NV) in the periphery which could not be seen using conventional 7 field angiography. Further studies have demonstrated the association between peripheral retinal nonperfusion and the occurrence of NV and DME.9, 10Identification of specific areas of retinal nonperfusion with UWFA allow targeted retinal photocoagulation rather than pan photocoagulation in the treatment of NV.11This approach can minimize laser-induced side effects such as field loss and macular edema.12A recent study evaluated the efficacy of UWFA in the detection of diabetic pathology which demonstrated retinal nonperfusion and NV in 10% of eyes that would have been missed by standard FA.13
Uveitis: UWF technology allowed clear identification of peripheral retinal lesions and greatly enhanced objective documentation of disease activity and progression. It ishelpful in monitoring areas of old or new retinal inflammatory activity in patients with non-infectious vasculitis, which may lead to earlier treatment and better visual outcomes.14
Retinal vascular occlusive disease-UWFA (ultra-wide angle fluorescein angiography) helps in measuring the “Ischemic index” in retinovascular occlusions which describe a ratio of the nonperfused retina over the whole retinal area measured manually from UWFA images.15
Retinopathy of prematurity(ROP) -UWFA images showed clear views of the different stages of ROP features at the posterior pole and peripheral retina and helps in identification of skip areas missed by initial laser treatment in the peripheral retina and managed accordingly.16 It also guides in monitoring the regression of ROP following laser and intravitreal bevacizumab treatment.
Choroidal melanoma-UWF technology for more accurate evaluation of the criteria established by Shieldsincludingtumor thickness > 2 mm, presence of subretinal fluid, clinical symptoms, orange pigment overlying the surface of the tumor, and tumor margins touching or being located within 3 mm of the optic disc.17
Coats disease-UWF fundus photography and angiography helps in documentation and evaluation of finding including telangiectasia of retinal vessels with aneurysms, exudative retinal detachment and subretinal lipid deposits.(Figure 2 )
Further advancements in UWF technology includes portable fundus cameras, a handheld smartphone-based retinal camera capable of capturing high-quality wide-field fundus images which can directly transmit data from the phone via wireless telecommunication system for remote evaluation.18
Limitations of UWF technology-
- Artifacts due to eyelashes and eyelids which obscures superior and inferior field.
- Only two colors are used, red and green laser, therefore, the pseudo-color images are not similar to routine fundus images.
- Distortion or difference in measurements in central and periphery as 3D area portrayed in 2D.
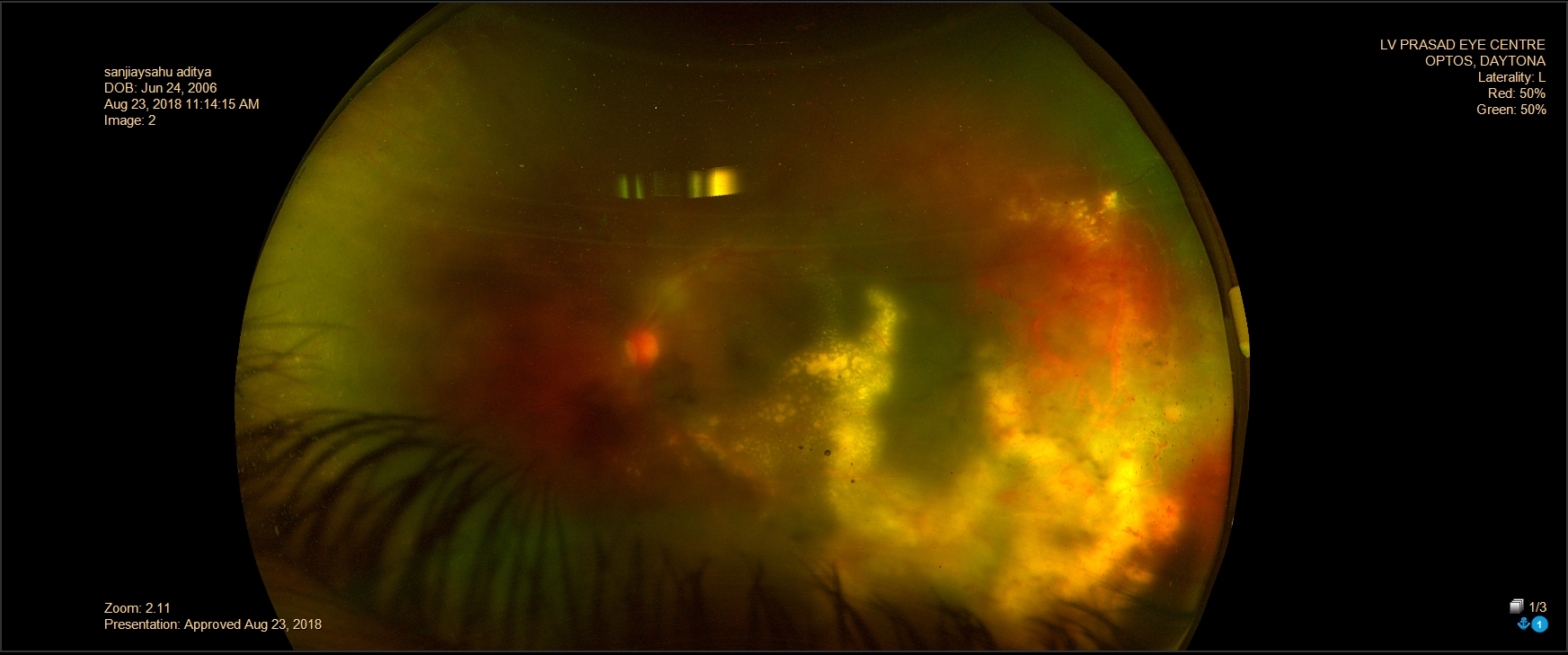
Figure 2- Ultrawide field color fundus photograph of an eye with Coat’s disease showing exudative retinal detachment with telangiectatic vessels temporally with massive exudation.
ADAPTIVE OPTICS
Adaptive optics (AO) is defined as a discipline to improve the performance of an optical system by reducing the effect of wavefront distortions19 using Hartman-Shack wavefront sensor19and the deformable mirror giving higher axial and transverse resolution with better contrast. AO provides high-resolution images of photoreceptors, retinal pigment epithelial mosaic, vascular flow in small capillaries.(Figure 3) It was tried in ophthalmology after its success in astronomy.
Upcoming AO fundus imaging methods include-
AO flood-illumination ophthalmoscopy (AOFIO)-which is able to observe structures like capillaries and the outer segment of cone.20
AO-OCT- which has high resolution both transversely and axially, enabling 3D cellular imaging which play an important role in fundus functional imaging in future.21
Limitation of AO-OCT including inability to detect fluorescent signals,the slow speed of 3D imaging, 22reduced imaging quality with eye movement which are overcome by AO-scanning laser ophthalmoscope which includes fluorescein angiographyAutofluroscence and ICG which has high recording speed of about 30 frames per second.23 Its application includes assessment of structural details which are difficult to be observed with conventional methods, such as the microscopic features of retinal micro aneurysms, different vessel parameters like arteriole thickness24, 25, velocity of blood cells in capillary26, parafoveal capillary density27 and area of foveal avascular zone.28, 29.

Figure 3-High resolution image of photoreceptors using adaptive optics
ADVANCES IN OCT IMAGING
1.Widefield OCT (WF-OCT)- (Optovue (Fremont, CA) Avanti RTVueXR) with 12 mm x 9 mm Bscans, along with both SD and SSOCT technologies helps in higher scanning speed of the SSOCT 1,060nm system, wider scans in the same acquisition time as of a standard scan on the SD system. These faster systems can collect between 684,000 and 1,368,700 A scans/second for outstanding volumetric acquisition speed. It offers 3µm digital resolution, providing detailed imaging of both the choroid and the retina with active eye tracking enhances image stability.30-34 It has been clinically utilized in following conditions. (figure 4)
A. Multifocal central serous retinopathy–WF-OCT provides improved visualization of multiple pigment epithelial detachments and numerous foci of subretinal fluid.
B. Peripapillary choroidal neovascular membranes-Utilizing WF-OCT to include the fovea and optic nerve simultaneously may allow for better monitoring of the proximity of subretinal fluid to the fovea, as well as treatment response.
C. Vitreoretinal relationship in pathologic conditions, such as vitreomacular traction, proliferative diabetic retinopathy, in which delineation with standard OCT scan lengths are difficult, WF-OCT can outline these relationships better, from both cross-sectional and three-dimensional perspectives.
D. In differentiation of retinal detachments and retinoschisis, targeted peripheral OCT allows clear delineation of the retinal layers and improved diagnosis of the splitting of retinal layers vs full thickness retinal elevation.
E. Characterization of choroidal lesions like choroidal nevus, malignancies could also improve using targeted peripheral OCT and WF-OCT.
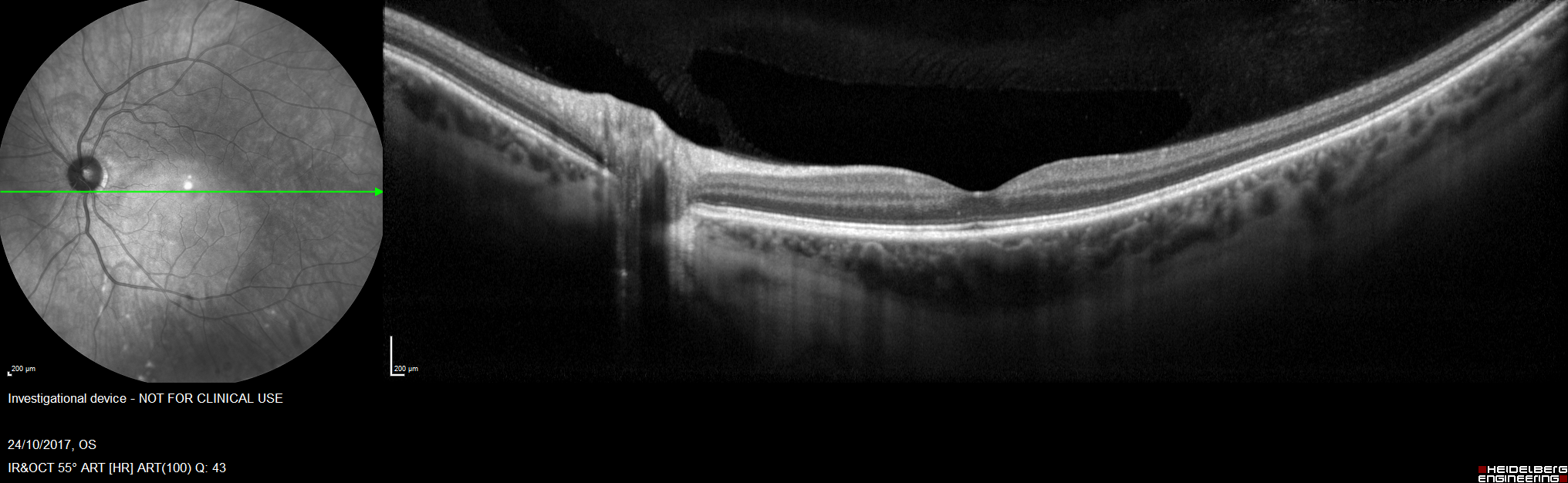
Figure 4- Wide Field Optical Coherence Tomography scan of a healthy eye
2.Hand-held OCT-Most of the OCT systems for ophthalmology is in a fix-mounted table-top form, which requires patients to sit in front of the machine and hold their eye open for several seconds to finish the imaging process. This is difficult for subjects with certain disabilities and children. To improve the portability and application of the OCT, different kinds of handheld OCT probes have been developed along with microelectromechanical (MEMS) scanning mirror, which makes the handheld probe more compact and lighter.30 Hand-held OCT has expanded our understanding in pediatric retina including the normal foveation process, premature retinal architecture and certain pediatric retinal diseases such as ROP. 30
3. Intraoperative OCT– Hand-held OCT was used for intraoperative OCT imaging. However, the primary drawback of the handheld OCT system was its restriction for use only during pauses in surgery. Maintaining the sterilization and increase in surgical time. These challenges are overcome by the advent of intraoperative swept-source microscope-integrated OCT (SS-MIOCT), which is capable of imaging and rendering up to 10 volumes per second and can perform volume rotation, allowing the surgeon to get a comprehensive 3-D view of the retina.34 This helps to guide intraoperative procedures in diabetic tractional retinal detachment, tractional schisis, detachment, dense membranes, and fluid at subretinal, intraretinal, preretinal levels and across multiple locations in the retina. (figure 5)
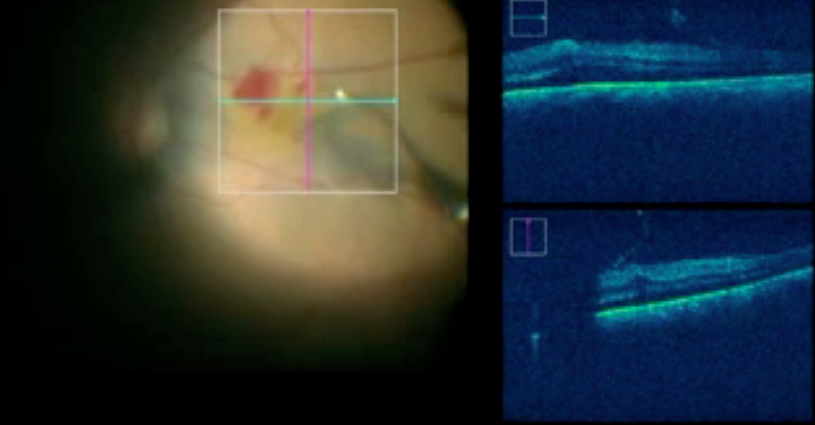
Figure 5.- Intraoperative view of swept-source microscope-integrated optical coherence tomography
E.OCT Angiography - It uses laser light reflectance of moving red blood cells to accurately depict vessels through a different segment in the retina, thus eliminating the need for intravascular dyes.It employs two methods for motion detection, amplitude decorrelation which detects differences in amplitude between two different OCT B-scans and phase variance which is related to the emitted light wave properties, and the variation of phase when it intercepts moving objects.Using split spectrum amplitude decorrelation and volume averaging technique, improvement in visualization with reducing background noise from normal small eye movements can be achieved. These OCT-A algorithms produce an image (3mm2to 12mm2) that is segmented into four zones: the superficial retinal plexus, the deep retinal plexus, outer retina and the chorio capillaries.(figure 6)
Advances like SS-OCTA devices, has larger single acquisition scan patterns, such as 12 mm x 12 mm and 15 mm x 9 mm with the internal fixation point which can be circled centered at the fovea and in parafoveal regions.
Further techniques like variable interscan time analysis (VISTA), which has been applied to high-speed prototypes to compare not only flow or no flow but also relative flow speeds in the retinal vasculature, which helps in understanding the changes in flow speed in patients with diabetic retinopathy as well as in the vessels of choroidal neovascularization. In the future it may even allow for quantification of retinal and choroidal blood flow velocities.
Improvement in the segmentation schemes based on vascular plexus have also been described, which may provide a basis for standardization across devices and thus improve automated visualization of vascular pathology.
Problems of artefacts have been tackled with technologies like pixel and volume registration of OCTA which helps to view retinal vasculature in 3 dimensions. Semiautomatic algorithms are also being developed to improve segmentation of the retinal nerve fibre layer.35 .
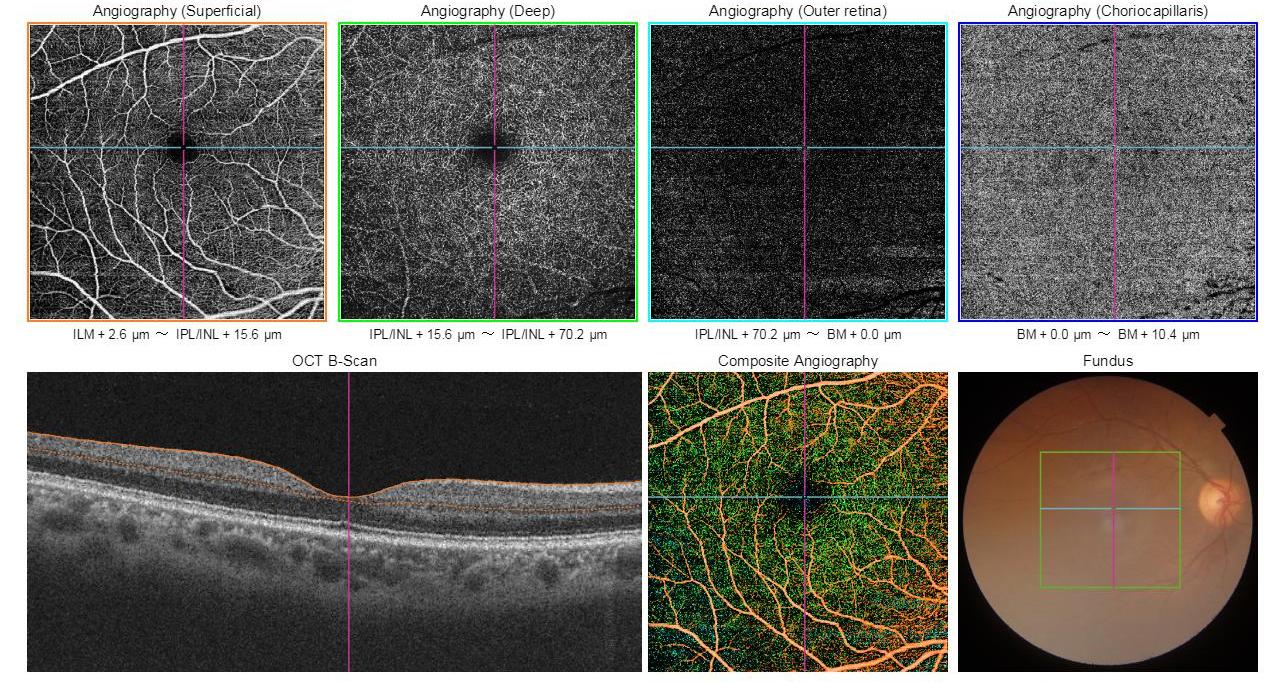
Figure 6- Optical coherence tomography angiography scan at four slabs: the superficial retinal plexus, the deep retinal plexus, outer retina, and the choriocapillaries
REFERENCES:
1.Tan AC, Fleckenstein M, Schmitz-Valckenberg S, et al. Clinical Application of Multicolor Imaging Technology. Ophthalmologica 2016;236:8-18.
2.Sarks J, Arnold J, Ho IV, et al. Evolution of reticular pseudodrusen. Br J Ophthalmol 2011;95:979-85.
3.Pumariega NM, Smith RT, Sohrab MA, et al. A prospective study of reticular macular disease. Ophthalmology 2011;118:1619-25.
4.Klein R, Meuer SM, Knudtson MD, et al. The epidemiology of retinal reticular drusen. Am J Ophthalmol 2008;145:317-326.
5.Prescott G, Sharp P, Goatman K, et al. Improving the cost-effectiveness of photographic screening for diabetic macular oedema: a prospective, multi-centre, UK study. Br J Ophthalmol 2014;98:1042-9.
6.Litvin TV, Ozawa GY, Bresnick GH, et al. Utility of hard exudates for the screening of macular edema. Optom Vis Sci 2014;91:370-5.
7.Pang CE, Freund KB. Ghost maculopathy: an artifact on near-infrared reflectance and multicolor imaging masquerading as chorioretinal pathology. Am J Ophthalmol 2014;158:171-178 e2.
8.Shoughy SS, Arevalo JF, Kozak I. Update on wide- and ultra-widefield retinal imaging. Indian J Ophthalmol 2015;63:575-81.
9.Patel RD, Messner LV, Teitelbaum B, et al. Characterization of ischemic index using ultra-widefield fluorescein angiography in patients with focal and diffuse recalcitrant diabetic macular edema. Am J Ophthalmol 2013;155:1038-1044 e2.
10.Wessel MM, Nair N, Aaker GD, et al. Peripheral retinal ischaemia, as evaluated by ultra-widefield fluorescein angiography, is associated with diabetic macular oedema. Br J Ophthalmol 2012;96:694-8.
11.Muqit MM, Marcellino GR, Henson DB, et al. Optos-guided pattern scan laser (Pascal)-targeted retinal photocoagulation in proliferative diabetic retinopathy. Acta Ophthalmol 2013;91:251-8.
12.Kaines A, Oliver S, Reddy S, et al. Ultrawide angle angiography for the detection and management of diabetic retinopathy. Int Ophthalmol Clin 2009;49:53-9.
13.Wessel MM, Aaker GD, Parlitsis G, et al. Ultra-wide-field angiography improves the detection and classification of diabetic retinopathy. Retina 2012;32:785-91.
14.Leder HA, Campbell JP, Sepah YJ, et al. Ultra-wide-field retinal imaging in the management of non-infectious retinal vasculitis. J Ophthalmic Inflamm Infect 2013;3:30.
15.Prasad PS, Oliver SC, Coffee RE, et al. Ultra wide-field angiographic characteristics of branch retinal and hemicentral retinal vein occlusion. Ophthalmology 2010;117:780-4.
16.Fung TH, Yusuf IH, Smith LM, et al. Outpatient Ultra wide-field intravenous fundus fluorescein angiography in infants using the Optos P200MA scanning laser ophthalmoscope. Br J Ophthalmol 2014;98:302-4.
17.Shields CL, Shields JA, Kiratli H, et al. Risk factors for growth and metastasis of small choroidal melanocytic lesions. Ophthalmology 1995;102:1351-61.
18.Maamari RN, Keenan JD, Fletcher DA, et al. A mobile phone-based retinal camera for portable wide field imaging. Br J Ophthalmol 2014;98:438-41.
19.Zhang B, Li N, Kang J, et al. Adaptive optics scanning laser ophthalmoscopy in fundus imaging, a review and update. Int J Ophthalmol 2017;10:1751-1758.
20.Ra E, Ito Y, Kawano K, et al. Regeneration of Photoreceptor Outer Segments After Scleral Buckling Surgery for Rhegmatogenous Retinal Detachment. Am J Ophthalmol 2017;177:17-26.
21.Jonnal RS, Kocaoglu OP, Zawadzki RJ, et al. A Review of Adaptive Optics Optical Coherence Tomography: Technical Advances, Scientific Applications, and the Future. Invest Ophthalmol Vis Sci 2016;57:OCT51-68.
22.Liba O, SoRelle ED, Sen D, et al. Contrast-enhanced optical coherence tomography with picomolar sensitivity for functional in vivo imaging. Sci Rep 2016;6:23337.
23.Lu J, Gu B, Wang X, et al. High-speed adaptive optics line scan confocal retinal imaging for human eye. PLoS One 2017;12:e0169358.
24.Hillard JG, Gast TJ, Chui TY, et al. Retinal Arterioles in Hypo-, Normo-, and Hypertensive Subjects Measured Using Adaptive Optics. Transl Vis Sci Technol 2016;5:16.
25.Arichika S, Uji A, Murakami T, et al. Correlation of retinal arterial wall thickness with atherosclerosis predictors in type 2 diabetes without clinical retinopathy. Br J Ophthalmol 2017;101:69-74.
26.Bedggood P, Metha A. Direct visualization and characterization of erythrocyte flow in human retinal capillaries. Biomed Opt Express 2012;3:3264-77.
27.Pinhas A, Razeen M, Dubow M, et al. Assessment of perfused foveal microvascular density and identification of nonperfused capillaries in healthy and vasculopathic eyes. Invest Ophthalmol Vis Sci 2014;55:8056-66.
28.Chui TY, Zhong Z, Song H, et al. Foveal avascular zone and its relationship to foveal pit shape. Optom Vis Sci 2012;89:602-10.
29.Dubis AM, Hansen BR, Cooper RF, et al. Relationship between the foveal avascular zone and foveal pit morphology. Invest Ophthalmol Vis Sci 2012;53:1628-36.
30.Campbell JP, Nudleman E, Yang J, et al. Handheld Optical Coherence Tomography Angiography and Ultra-Wide-Field Optical Coherence Tomography in Retinopathy of Prematurity. JAMA Ophthalmol 2017;135:977-981.
31. Justis p. ehlers et al.Widefield OCT: Current and Future ApplicationsWhen retinal pathology goes beyond the macula, imaging can pose a challenge.Retinal Physician 2014:4
32. Raiji V, Walsh A, Sadda S. Future directions in retinal optical coherence tomography. Retinal Physician. 2012:9(4):3337.
33.Povazay B, Hermann B, Hofer B, et al. Widefield optical coherence tomography of the choroid in vivo. Invest Ophthalmol Vis Sci. 2009?50:18561863.
34. Hesham Gabr et al.Intraoperative 4-D Microscope-Integrated OCTReal-time image acquisition can provide new types of information to the retina surgeon.Retina today. 2017:12.
35. Malvika arya et al.Upcoming Advances in Optical Coherence Tomography Angiography Improvements could make OCTA a mainstay for retinal imaging. Retinal Physician.2018:01.
