Introduction
Acute anterior uveitis (AAU) results from a breakdown of the blood-aqueous barrier and acute inflammation of the iris and ciliary body. It is the most common form of uveitis encountered the world over. Keratic precipitates (KPs) are characteristic of AAU and represent inflammatory cells that deposit on the corneal endothelium due to the upregulation of adhesion molecules in response to local chemoattractant cytokines.
The association between HLA (human leukocyte antigen)-B27, a human major histocompatibility complex (MHC), and its gamut of inflammatory diseases and HLA-B27 associated AAU was first described in 1973 and to date, it remains one of the strongest HLA-disease associations known.
Epidemiology
Though most epidemiological studies are based in tertiary referral centers and have a referral bias towards posterior and intermediate uveitis, anterior uveitis is the most common form of uveitis encountered in most parts of the world.
HLA-B27 AAU comprises cases of acute anterior segment inflammation occurring in association with HLA-B27 antigen with or without the presence of systemic disease. The prevalence of HLA-B27 is 8-10% in the general western population and about 50% in patients with AAU, with a range of 19-88% across various racial groups. According to a population-based study from the Netherlands, the lifetime cumulative incidence of AAU is 0.2% in the general population and 1% in the HLA-B27-positive population. HLA-B27 AAU is the most common identifiable cause of anterior uveitis. 18-32% of all anterior uveitis cases in Western countries and 6-13% of anterior uveitis cases in Asia can be attributed to HLA-B27 AAU. The relatively lower frequency of HLA-B27 AAU in Asian countries is partly due to the lower frequency of HLA-B27 and also a comparatively lower strength of association between AAU and HLA-B27 in the population. HLA-B27 AAU affects males between 1.5 to 2.5 times more commonly than females and the age of onset is between the second and fourth decades of life.
HLA-B27 subtypes and disease association
HLA-B27 is unique in that it is only one of the few HLA class I molecules to be associated with the disease. Most HLA-autoimmune disease associations are with HLA class II molecules. HLA-B27 is a serological specificity that comprises 28 different alleles which code for 24 different subtypes. HLA-B*2705 is the most prevalent subtype in the Western population and has a clear association with ankylosing spondylitis (AS) and related spondyloarthropathies (SpA), including AAU. HLA-B*2704 is the predominant subtype in the Asian population and has a weaker association with AAU, which may contribute to the lower frequency of HLA-B27 AAU seen in these populations.
HLA-B27 AAU Clinical features
The typical presentation of HLA-B27 positive AAU is as follows:
- Acute onset
- Unilateral, alternating, nongranulomatous anterior uveitis
- Significant cellular and protein extravasation in the anterior chamber chamber
- Fibrin and hypopyon in the anterior chamber
- Multiple recurrences
- Significant association with other HLA-B27 diseases.
There is a high tendency for recurrences, with a mean number of 0.6-3.3 attacks per patient per year of follow-up. The mean duration of each episode is 4-6 weeks. There may be a decrease in the frequency of acute attacks with increasing duration of disease.
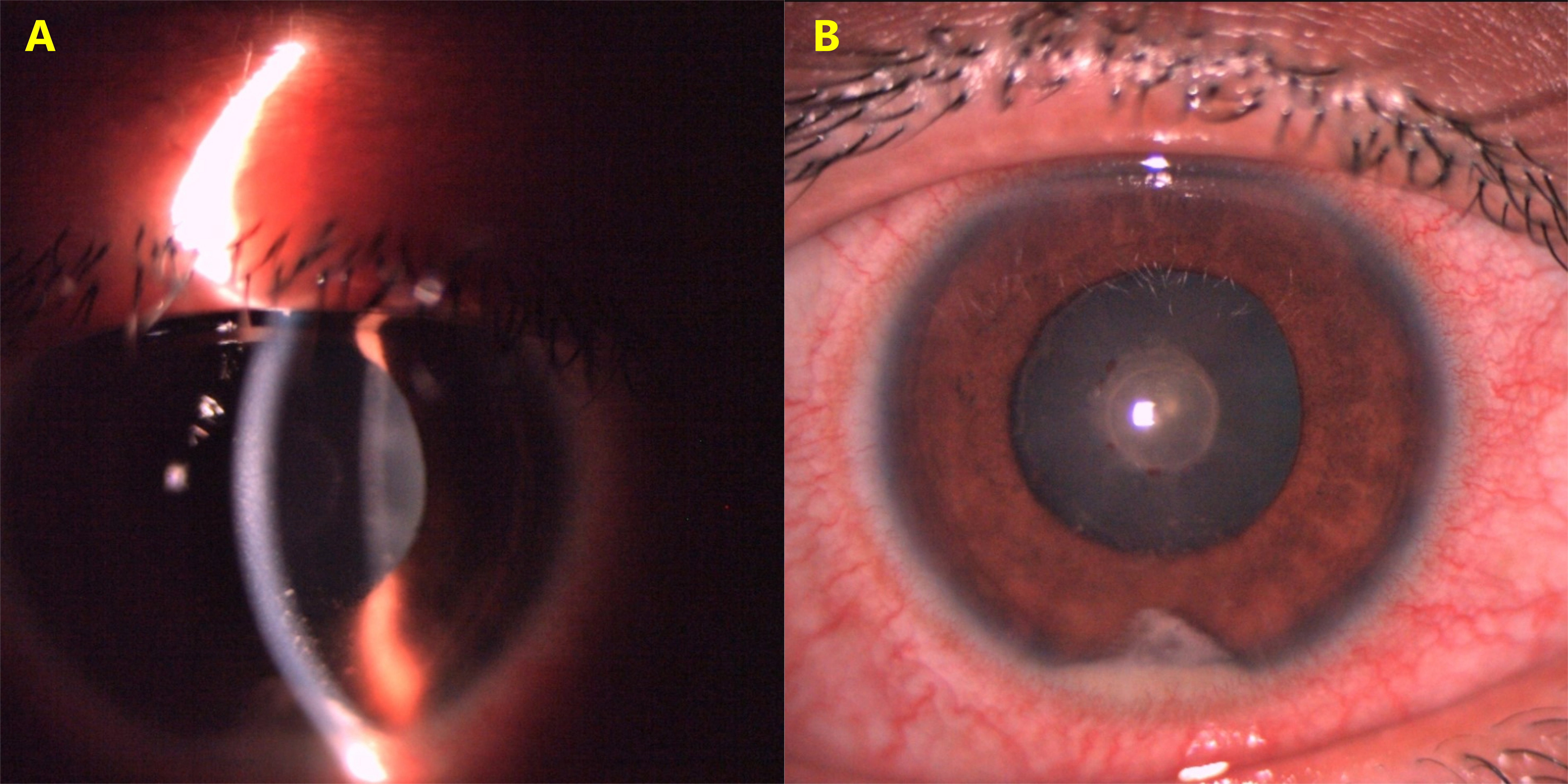
Figure 1: HLA-B27 acute anterior uveitis showing fresh nongranulomatous keratic precipitates (A). Fibrin with hypopyon in the anterior chamber (B).
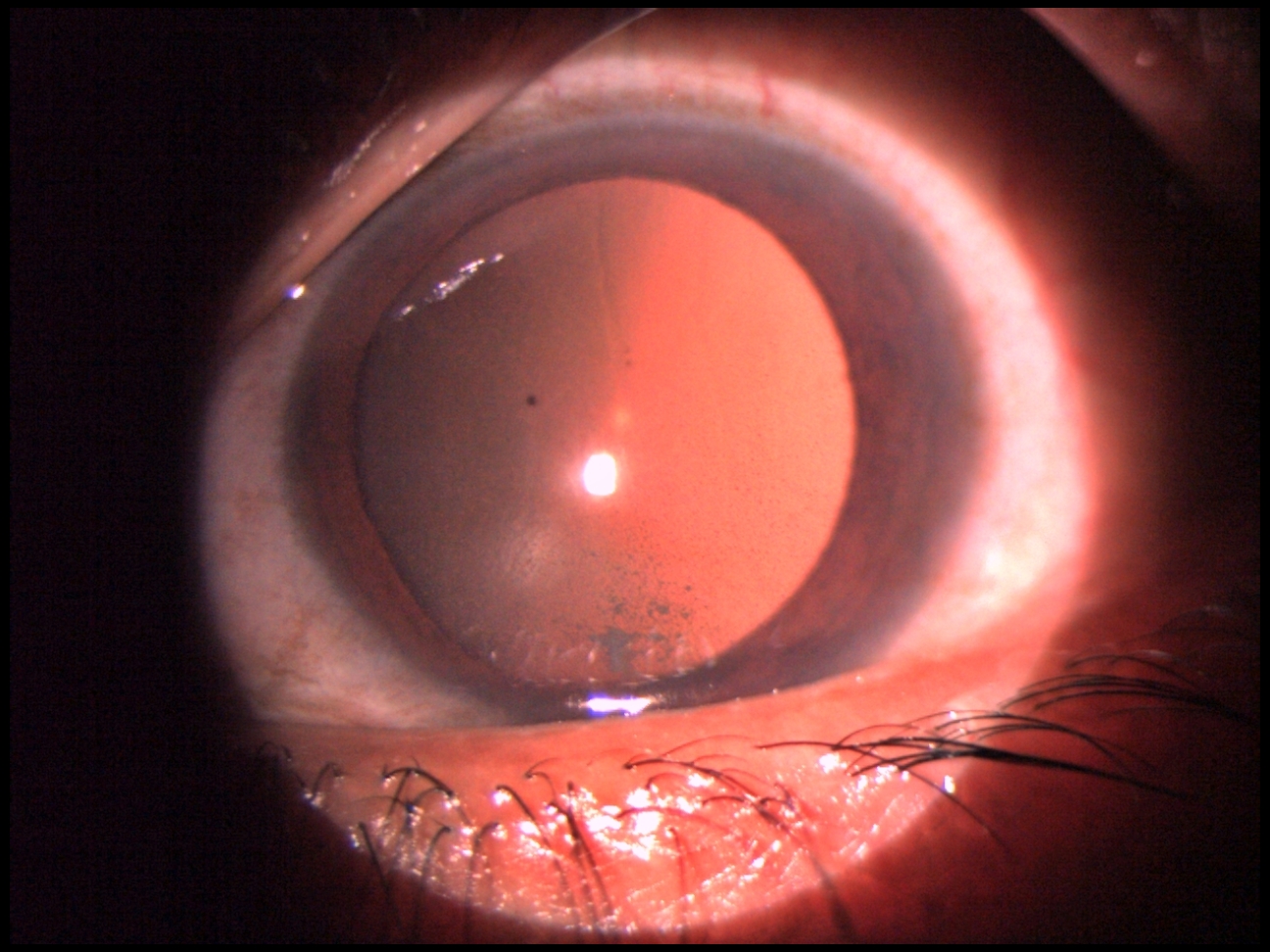
Figure 2: HLA-B27 acute anterior uveitis showing nongranulomatous fine keratic precipitates seen in retroillumination
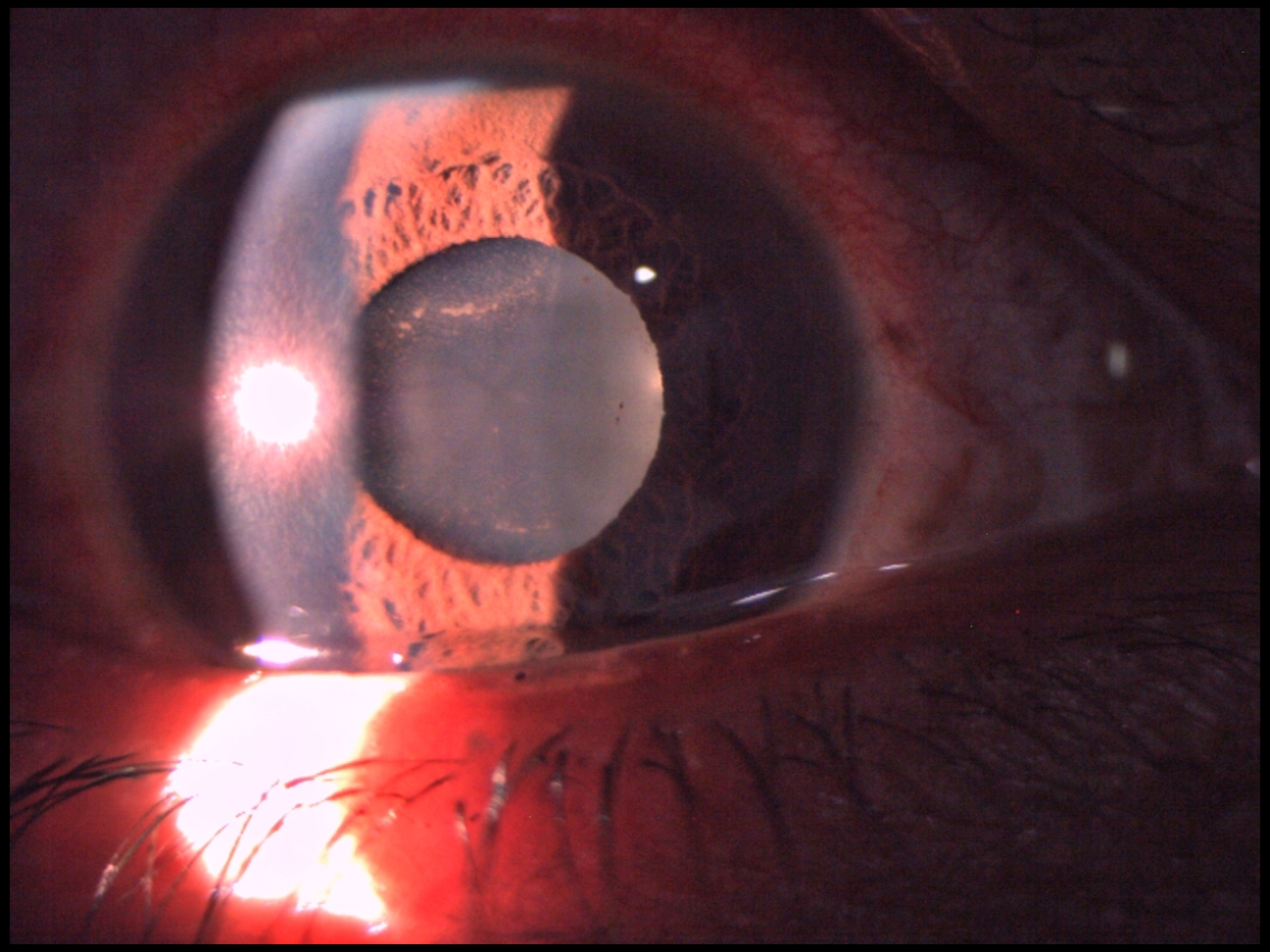
Figure 3: HLA-B27 acute anterior uveitis showing pigment deposits on the anterior lens capsule following released posterior synechiae.
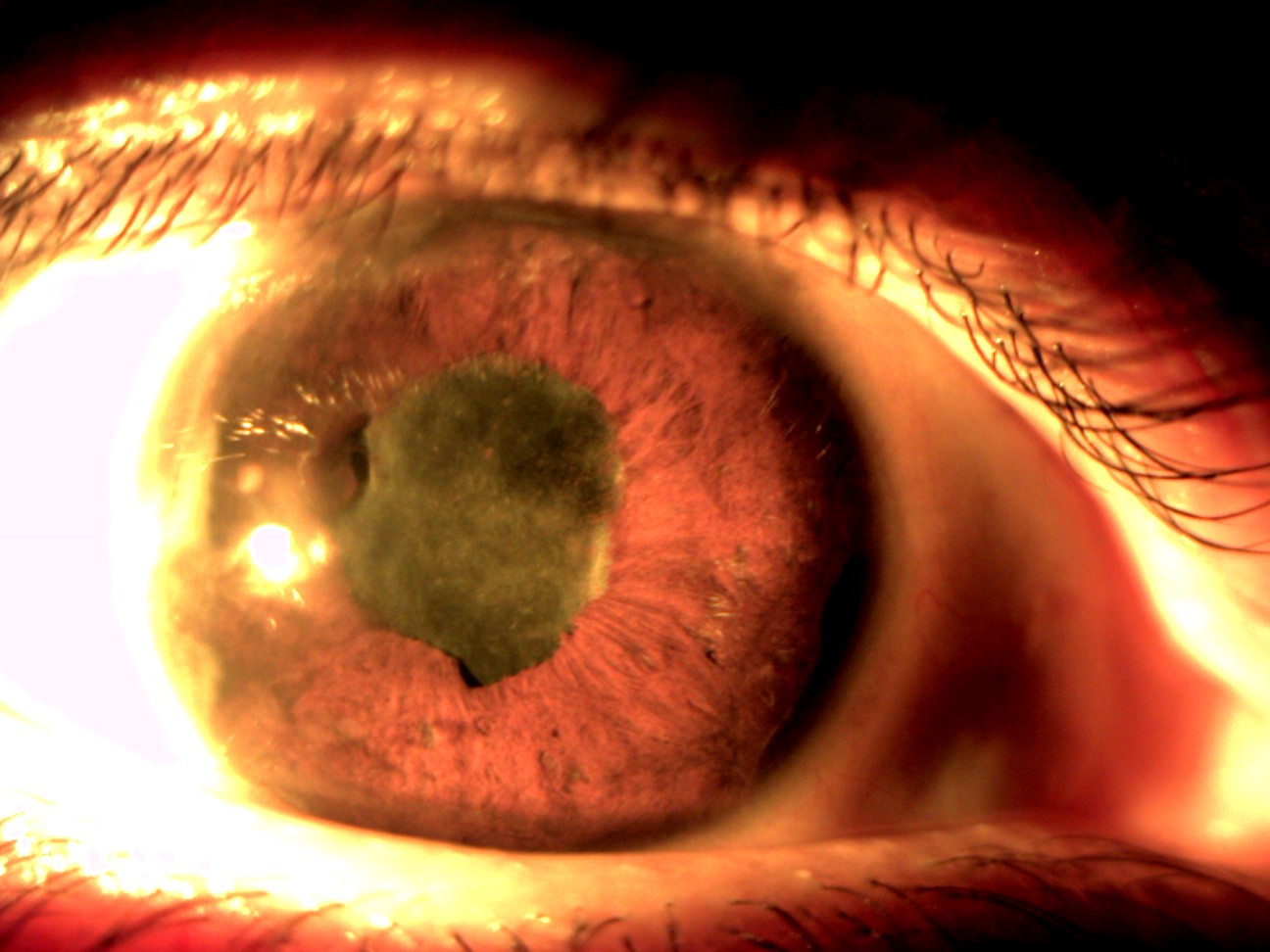
Figure 4: Chronic anterior uveitis with posterior and peripheral anterior synechiae in a case of HLA-B27 positive ankylosing spondylitis.
Associated Systemic Diseases
About 23-37% of AAU patients have associated seronegative SpA, while 49-84% of patients with HLA-B27 positive AAU have an associated systemic disease. Studies have shown that previously unknown systemic diseases have been diagnosed following ophthalmological examination after an episode of uveitis in more than half of patients with SpA and uveitis and up to 91% in undifferentiated SpA. AS is characterized by early morning low backache or stiffness, which is relieved with activity as the day progresses.
Ocular complications:
- Posterior synechiae, is by far the commonest with a frequency of 13%-91%.
- Cataract in 7%-28% cases.
- Ocular hypertension, secondary glaucoma and development of chronic anterior uveitis.
- Diffuse vitritis is the most common posterior segment complication.
- Cystoid macular oedema is the most frequent cause of visual impairment in uveitis and affects between 6%-13% of HLA-B27 positive cases of AAU.
Since HLA-B27 positive AAU tends to be recurrent, it is associated with a worse visual prognosis compared to HLA-B27 negative idiopathic AAU. About 10% of HLA-B27 AAU patients have been reported to suffer from legal blindness or severe visual impairment.
Pathology of AAU:
Endotoxin-induced uveitis (EIU): AAU can be induced in rats and mice by injection of endotoxin or liposaccharide (LPS) of Gram-negative bacteria at a site remote from the eye. EIU is a useful model supporting the etiological association between HLA-B27 AAU and Gram-negative bacterial triggers. Though EIU is predominantly anterior uveitis, posterior segment involvement has been noted.
Experimental Melanin-Protein-induced Uveitis (EMIU): Insoluble melanin-associated protein derived from bovine uveal tissue is used to induce experimental uveitis. EMIU is useful for histopathological studies as inflammation is limited to the anterior chamber of the eye.
Myelin Basic Protein (MBP)- induced anterior uveitis: A self-limited form of anterior uveitis develops in Lewis rats immunized with MBP. The inflammatory cellular infiltrates accumulate around iris nerve fibers suggesting autoreactivity to nerve antigens by MBP-specific T-cells.
Immunopathology:
Pro-inflammatory cytokines like TNF-⍺, IL-1 and IFN-𝛾 and immunoregulatory cytokines like IL-10 and TGF-𝛽 have been implicated in the pathogenesis of AAU. Patients with various forms of active uveitis have been shown to have higher aqueous and serum levels of TNF-⍺ and lower aqueous humor levels of the immunoregulatory cytokine IL-10.
Pathogenesis of HLA-B27 AAU:
HLA-B27-associated diseases are multifactorial occurring in genetically predisposed individuals, triggered by environmental factors.
Hypotheses:
The following hypotheses have been proposed to explain the possible pathogenic link between HLA-B27 and its associated diseases:
Molecular mimicry: Molecular mimicry refers to the similarity between antigenic epitopes of microbial components and host (self) tissue. A humoral or cellular immune response initiated against a microbial agent may cross-react with a similar epitope present on host tissues, resulting in autoimmune disease. HLA-B27 abnormally shares short peptide sequences with proteins from Gram-negative enteric bacteria.
HLA-B27 as an antigen-presenting molecule: HLA-B27 may act as an antigen-presenting molecule which is the primary function of MHC class I molecules. This hypothesis proposes that autoimmune disease is induced by cytotoxic T-cell mediated anti-host immune reaction, via molecular mimicry to arthritogenic peptide (found only in joint tissues) and uveitogenic peptide (found only in the eye), which can specifically be bound and presented by all disease-associated HLA-B27 subtypes. Gram-negative bacterial proteins are an example of exogenous uveitogenic peptides.
Treatment:
Topical steroids and cycloplegics are the mainstay of treatment in the majority of patients of HLA-B27 AAU with good results. More severe presentations, like CME and sight-threatening uveitis, may require more aggressive treatments like periocular or intraocular steroid injections, systemic corticosteroids and/or systemic immunosuppressive agents. The Systemic Immunosuppressive Treatment for Eye diseases (SITE) cohort study showed that corticosteroid-sparing success (tapering to 10mg or less) was attained over 6 months in 47.2% with mycophenolate mofetil, 46.1% with methotrexate, 28.5% with cyclosporine and 16.6% with azathioprine.
The Multicenter Uveitis Steroid Treatment (MUST) trial and a Cochrane review have reported that combination systemic immunosuppressive treatment resulted in better visual outcomes, lower rates of development of cataract, glaucoma, retinal detachment and endophthalmitis compared to Retisert implants.
The disease-modifying antirheumatic drugs (DMARDs) with methotrexate and sulfasalazine have been shown to reduce relapse rates in HLA-B27 AAU. Methotrexate also reduces AAU-related macular oedema.
Potential new therapies:
Anti-tumour necrosis factor-⍺ Therapy
Infliximab is particularly useful in the management of treatment-resistant and sight-threatening uveitis. Total resolution of uveitis has been reported with infliximab as the sole anti-inflammatory agent.
HLA-27-peptide Oral Tolerance
Oral tolerance induces peripheral immune tolerance when a specific antigen is administered orally. Orally administered HLA-B27 derived peptide (B27PD) has been shown to markedly reduce intraocular inflammation, reduce systemic corticosteroids and maintain or improve visual acuity. Unlike systemic immunosuppressive therapies, oral tolerance does not induce non-specific inhibition of the immune system or side effects.
References:
- Chang, J. H., McCluskey, P. J., & Wakefield, D. (2005). Acute anterior uveitis and HLA-B27. Survey of ophthalmology, 50(4), 364-388.
- Wakefield, D., Chang, J. H., Amjadi, S., Maconochie, Z., El-Asrar, A. A., & McCluskey, P. (2011). What is new HLA-B27 acute anterior uveitis?. Ocular immunology and inflammation, 19(2), 139-144.
- Wakefield, D., Clarke, D., & McCluskey, P. (2021). Recent developments in HLA B27 anterior uveitis. Frontiers in Immunology, 3380.
- D’Ambrosio, E. M., La Cava, M., Tortorella, P., Gharbiya, M., Campanella, M., & Iannetti, L. (2017, November). Clinical features and complications of the HLA-B27-associated acute anterior uveitis: a metanalysis. In Seminars in ophthalmology (Vol. 32, No. 6, pp. 689-701). Taylor & Francis.
- Kempen, J. H., Daniel, E., Gangaputra, S., Dreger, K., Jabs, D. A., Kaçmaz, R. O., ... & Suhler, E. B. (2008). Methods for identifying long-term adverse effects of treatment in patients with eye diseases: the Systemic Immunosuppressive Therapy for Eye Diseases (SITE) Cohort Study. Ophthalmic epidemiology, 15(1), 47-55.
- Multicenter Uveitis Steroid Treatment Trial Research Group. (2010). The multicenter uveitis steroid treatment trial: rationale, design, and baseline characteristics. American journal of ophthalmology, 149(4), 550-561.
- Brady, C. J., Villanti, A. C., Law, H. A., Rahimy, E., Reddy, R., Sieving, P. C., ... & Tang, J. (2016). Corticosteroid implants for chronic non‐infectious uveitis. Cochrane Database of Systematic Reviews, (2).
