Introduction
Fungal or mycotic keratitis is a leading cause of ocular morbidity, opacification and preventable blindness.1 The approximate annual incidence of fungal keratitis in India is said to be 11.3/10,000 population.2 Frequently it is caused by filamentous fungi (Aspergillus, Fusarium, Curvularia) in the tropical areas, while in temperate regions it is predominantly caused by Candida species (C. albicans, C. Parapsilosis). It is considered as one of the most difficult forms of microbial keratitis for the Ophthalmologist to diagnose and manage. Medical therapy alone is often inadequate and surgical therapy like therapeutic penetrating keratoplasty, conjunctival flap, lamellar keratoplasty or cryotherapy is required for control of infection.
Epidemiology and Demographics
Fungal keratitis incidence or prevalence is more in tropical regions of the world like India and Southern USA and is less commonly encountered in the temperate zones.3 In East India, fungal keratitis was found to be more prevalent than other microbial keratitis.4 Fungal species causing keratitis differ depending on the different geographical regions.3 In South India, studies have suggested that Fusarium spp. was commonly responsible for the fungal keratitis cases as also in tropical places of South Florida, Ghana, Paraguay, Nigeria, Tanzania, Hong Kong, and Singapore. Whereas in the temperate regions of subtropical countries of Nepal, North India, and Bangladesh Aspergillus was found to be predominant cause.5
In temperate regions Candida species are the predominant cause of fungal keratitis.5 Candida spp. was found to be prevalent in London and Melbourne with the incidence rate of about 60% and 32.7%, respectively. These variations signify that temperature and humidity plays a major role in determining the organism found in the environment.5
As with other microbial keratitis, fungal keratitis incidence is more common in male due to involvement in outdoor activities exposing them to corneal injury with external agents and this is more common in age group (21-50 years).5 Seasonal variation has been observed in the incidence of fungal keratitis. Incidence of fungal keratitis increases during the paddy harvesting and also during the time of the year when agricultural activities are increased (i.e. between June and September).5 However, in South-central India the incidence of fungal keratitis is more during the winter season (from October to January) and monsoon season (from June to September).5
Risk Factors
The ocular surface is normally protected from microbial invasion by its anatomic and physiological microenvironment. External trauma remains the most important risk factor for the development of fungal keratitis.
Risk factors which have been found to be associated with fungal keratitis include
- Corneal injury, especially with the vegetative matter, has been found to be the most important risk factor in developing world6,7
- Use of topical steroids drops
- Diabetes mellitus
- Co-existing other microbial keratitis (polymicrobial keratitis)
- Contact lens use and contact lens solution (in developed world7)
In 2005, an outbreak of Fusarium keratitis across several countries was traced to contact lens solution, which was later withdrawn from the market.3
Pathogenesis and Clinical Features
The fungal pathogens gains access to the host stroma through breached epithelium, where its hyphae germinates. This leads to activation of innate neutrophils, macrophages and dendritic cells in the corneal stroma which mediates host immune defense through release of various chemical factors.8
The Most patients report to the clinic with pain, redness, photophobia and white opacit/ lesion in the affected eye of acute or sub acute onset and are known to have indolent progression.
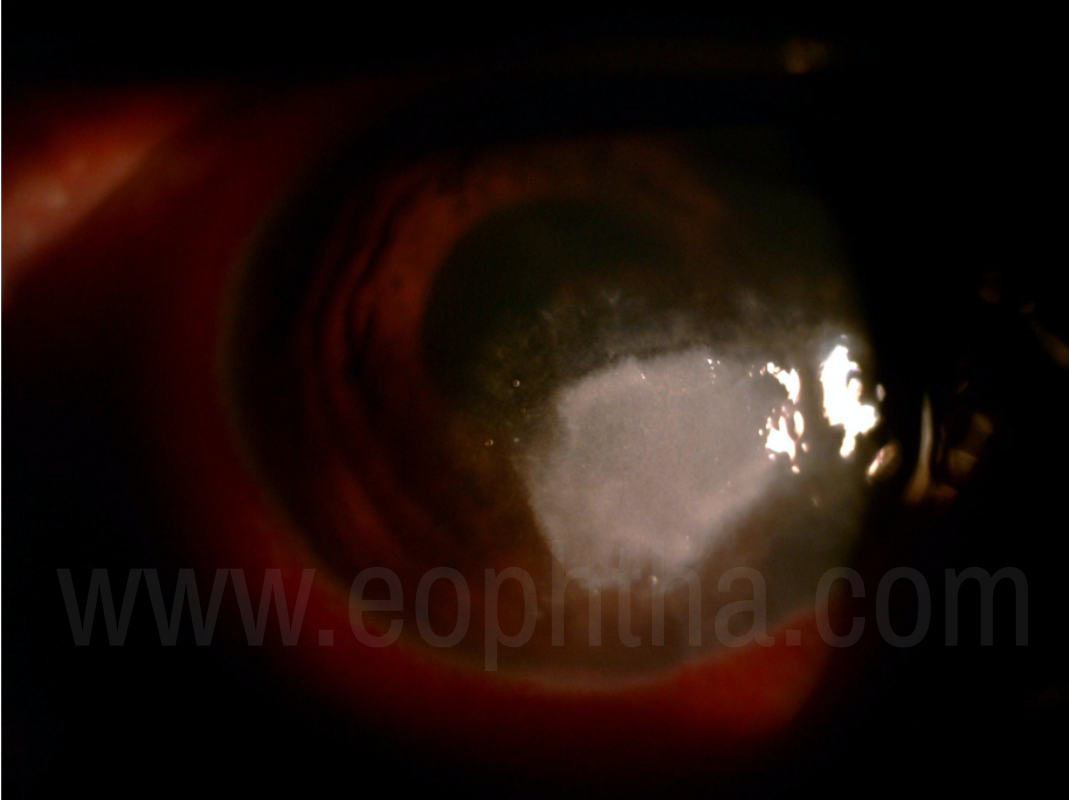
Figure 1: Dry raised Plaque in Fungal Corneal Ulcer
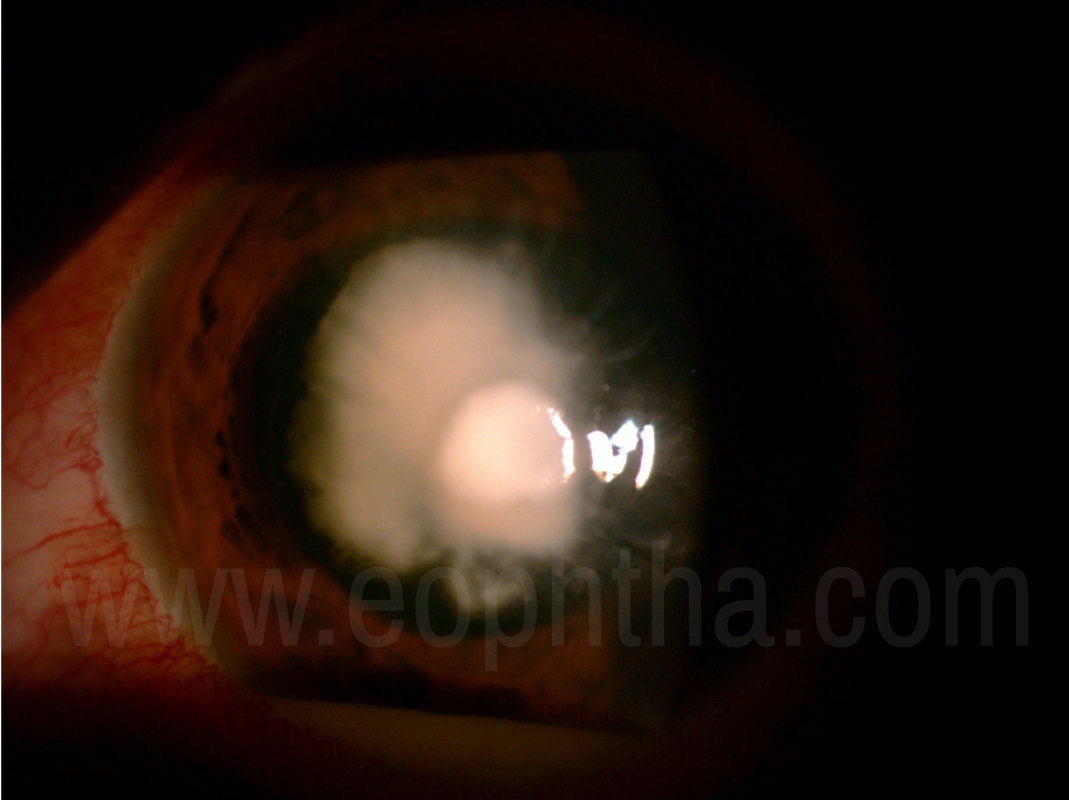
Figure 2: A fungal corneal ulcer withthick endothelial exudates
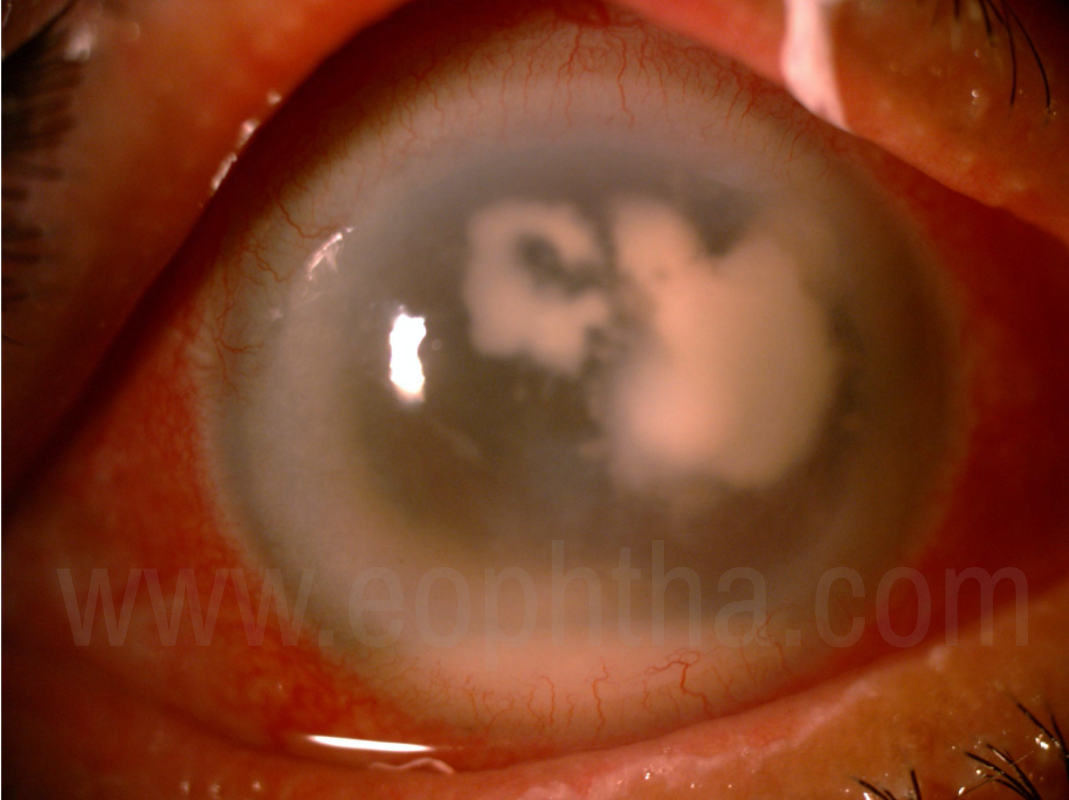
Figure 3: Fungal keratitis with satellite lesions
Signs of fungal keratitis in comparison to bacterial keratitis are generally minimal. Lid edema is minimal to absent in these cases. Clinical features in vast majority of fungal keratitis case can be obvious on the slit lamp examination unless the patient was initiated on several empirical non specific treatments.
Early cases of fungal keratitis can be missed for HSV viral dendrite keratitis because of the branching lesions. Understanding varied presentation, especially in developing world where it is more or if not equal in prevalence, is important in establishing microbiological evidence fungal keratitis with proof to continue appropriate medication. Various studies have shown that there are certain clinical features which are characteristics of fungal keratitis; dry gray to white infiltrate with feathery edges which have either a creamy base or raised exudate appearing as plaque with leathery texture infiltrate, satellite lesions, ring infiltrate, mild to moderate stromal edema and endothelial plaque. Other findings include descemets folds, posterior stromal abcess, yellowish to white hyopyon, immune ring and intact epithelium with deeper infiltrates.9–11 Since then, these features are commonly accepted as diagnostic of filamentous fungal keratitis.
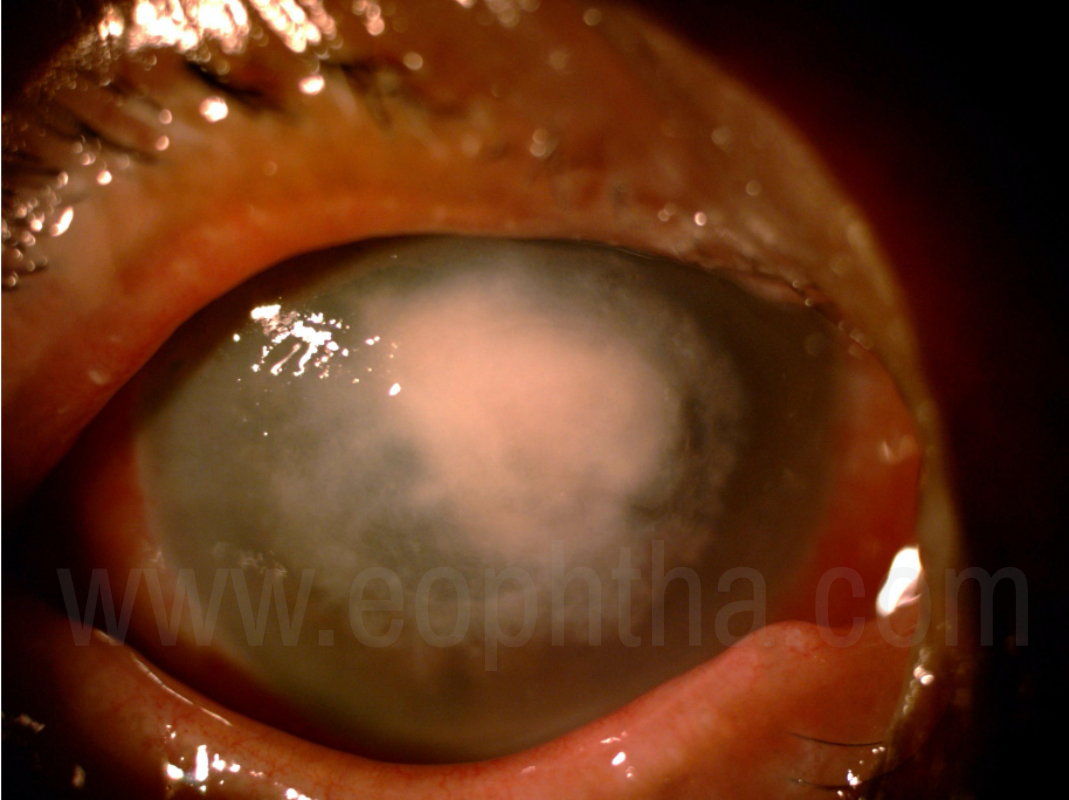
Figure 4:Severe fungal keratitis with hyphate edges
Dematiaceous fungi such as Curvularia and Bipolaris spp., apart from the above mentioned signs, shows pigmentation with raised lesions.12
Keratitis caused by yeast like organism such as Candida resembles non-specific keratitis and is usually seen in immunocompromised patients who are on long term topical steroids or systemically receiving immunological agents.
There are certain features that can be used to differentiate between the different fungal species like pigmentation in dematiaceous fungus, and endothelial plaque and ring infiltrates were found to be more common in Aspergillus species.12 However, there is considerable overlap in the clinical presentation among different etiological agents.
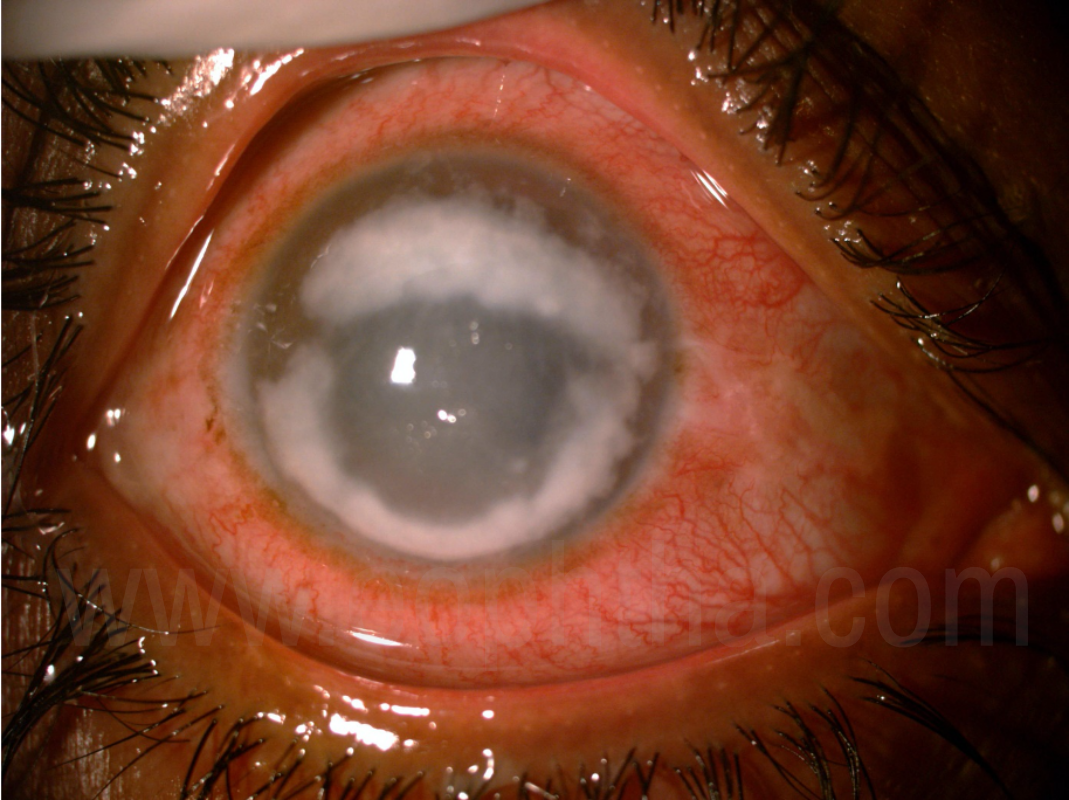
Figure 5:Ring Infiltrate
Comparison of bacterial and fungal keratitis clinical features have been done in many studies which showed following features, though can be present in both, are more common in fungal keratitis. These are longer history of symptoms, dry raised necrotic or fluffy surface, endothelial rings, satellite lesions and serrated margins.13 Also, apart from bacterial, acanthamoeba keratitis clinical picture can resemble that of a fungal keratitis. A study was done in South India, which showed that features of acanthamoeba were significantly different from that of fungal or bacterial keratitis and includes younger age, longer symptom duration, prior use of topical antibiotics, , presence of a ring infiltrate and disease confined to the epithelium.14 Though these features can point towards a certain etiology, but clinical judgement should be based on the whole clinical scenario including history, as the overlapping of clinical features is not uncommon. Hence, it has been suggested that, rather than giving importance to a particular clinical feature an Ophthalmologist should be looking for more than one clinical feature which can lead to increased certainty in diagnosing fungal keratitis.13
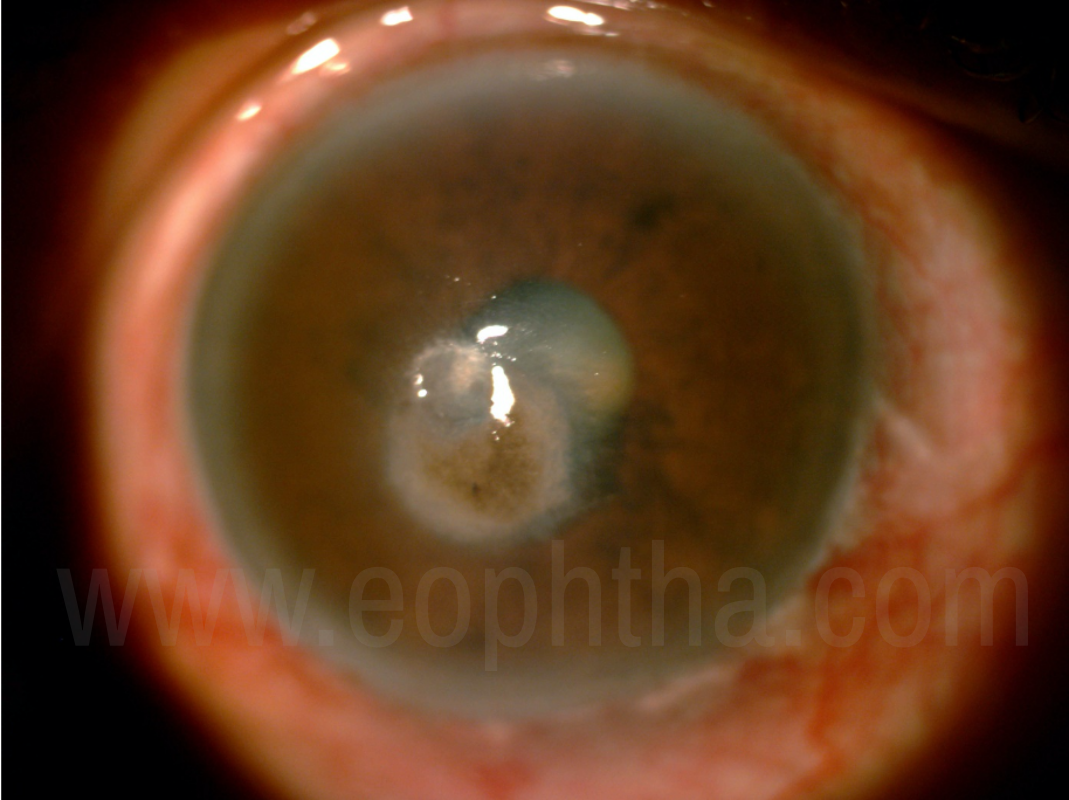
Figure 6:Dematacious Fungal keratitis
Though uncommon, the incidence of mixed microbial keratitis with bacterial and fungal etiology have been reported to be about 1.9% to 15.8%.15,16 The most common species isolated in the mixed microbial keratitis cases are Fusarium or Aspergillus among fungus and S. epidermidis among bacteria.15,16 The prognosis of mixed keratitis is usually poor and the most common risk factor was found to be ocular trauma. It usually doesn’t respond to either antibacterial or antifungal treatment alone and combination of both should be given. Keratitis cases not responding to initial medical therapy and behaving unusually should be repeatedly scarped for culture and mixed microbial etiology should be considered. Polymicrobial keraitits cases will usually present earlier than a typical fungal keratitis case and the size of infiltration and ulceration will be larger.16 Fernandes et al.16 have reported in their study that polymicrobial keratitis with infiltrate of >6mm in greatest dimension should undergo early penetrating keratoplasty in the wake of poor outcome and visual prognosis.
Diagnosis
The clinical diagnosis of fungal keratitis is not more than 70% by an experienced clinician.3 However, a well-experienced clinician can identify 38% of fungal keratitis cases based on the clinical photograph alone.17 Recently, newer imaging modality like in vivo confocal microscopy can act as an adjunct for the diagnosis of fungal keratitis and other microbial keratitis. Nevertheless, microscopic examination and culture of the samples remain the gold standard for the diagnosis of fungal keratitis.3 Newer molecular methods have increased the specificity and sensitivity of the diagnosis.
Conventional microbiological methods
The specimen for the diagnosis can include corneal scraping, corneal biopsy, or corneal button. The ideal corneal scarping can be obtained by Kimura platinum spatula or surgical blade no. 15 under topical anesthesia.3 Other instruments that can be used for the scraping are bent hypodermic needle, cataract knife, Beaver blade no. 64, calcium alginate swab, etc. The corneal scrapping specimen can be directly processed on the patient side or can be transported to the lab in a transport medium like Amies transport medium without charcoal.
The specimens need to be handled carefully to avoid contamination while transportation to the laboratory. Also, if the patient happens to be a contact lens wearer the contact lens and the solution should also be submitted as such to the laboratory. The corneal biopsy or corneal button should be cut into small pieces with a sterile blade and the pieces should be subjected to the microbiological diagnosis as for corneal scrapings, and also should be sent for histopathology to aid in diagnosis.
Multiple scraping should be collected from the base and edge of the ulcer to obtain an appropriate specimen. Conjunctival specimens can give false-positive results and hence should not be taken for microbiological diagnosis of microbial keratitis.18
Direct Microscopy
Various stains that can be used for diagnosis are Gram, Giemsa, potassium hydroxide(KOH), KOH + calcofluor white, Giemsa + calcofluor white, acridine orange, Periodic acid Schiff, lactophenol cotton blue and Gomori methenamine silver stains.
Grams stain is commonly used in laboratories to stain bacterias, however, it can also be used to detect fungi. Badiee et al.18 have shown in their study that grams stain can detect 35-73% of the cases of fungal keratitis. The sensitivity of grams stain has been found to be ranging from 21% to 90% in detecting fungal species in a case of fungal keratitis.18,19
KOH smear is considered as a single most reliable screening tool for the diagnosis of fungal keratitis provided sufficient sample is taken and expertise in the detection of fungal filament is available.18 KOH smear is prepared by directly putting a drop of 10% KOH on the slide with the sample and observed under the microscope. KOH dissolves most of the mammalian cells and tissue allowing fungal elements to be seen, which dissolves very slowly.19The sensitivityof KOH ranges from 62% to 99% and specificity ranges from 73% to 100% in fungal keratitis cases.18–20
Giemsa stain stains the fungi blue and has been reported to have sensitivities of 40% to 85% and specificities of 70% to 96% in fungal keratitis cases.19
Calcofluor white (CFW) is a nonspecific fluorochrome and binds to the fungal cell wall which fluoresces in ultraviolet light. Calcofluor white has very high sensitivity, however, in a resource limited establishment 10% potassium hydroxide can be very helpful in diagnosing fungal keratitis.
Gomori methenamine silver stains the fungal cell wall silver and is often used to stain corneal biopsy specimens.
A combination of stains have been used in some studies to increase the diagnostic accuracy of direct smear. The addition of CFW to KOH or Giemsa have increased the sensitivity of diagnosis of fungal keratitis from 81% to 97% and 40% to 98%, respectively.21 The sensitivity and specificity of the direct microscopy is higher when the disease is in the advanced stage.22
|
Stain |
Appearance |
|
Fungal cell wall and septa appears as refractile structures |
|
Variable staining pattern; Yeasts stain violet in colour , and fungal filaments appear either as Gram negative (pink) or as faintly stained outline of fungus with unstained protoplasm. Helpful in identifying yeast and yeast like fugus |
|
Fungal filaments stain blue in color with purple blue nuclei |
|
Fungal filaments with septa appears as bright white structures against blue background under fluorescent microscope. Good visualization of morphology of fungus |
|
Valuable for demonstrating fungi in tissues. Fungal filaments are seen in black colour against light green background |
Culture
While the demonstration of fungal filaments in the microscopy is considered diagnostic of fungal keratitis, the culture of the organism is needed for the identification of particular etiological agents. The culture medium used for fungal identification is Sabouraud Dextrose agar (SDA) or Potato dextrose agar (which can show better sporulation);22 however, the fungus can also grow on blood and chocolate agar. Sabouraud Dextrose agar is incubated at 25-27oC. The typical incubation period, for the fungal organisms causing keratitis, to grow on SDA is usually 2 weeks.3 Hence, the media should be examined daily for growth and are incubated for about 2 weeks before discarding. Fungi causing ocular infections are usually saprophytic fungus and have been shown to grow earlier than SDA in blood agar and chocolate agar; however, for identification of species, the growth characteristics on SDA is essential. Whether the growth in the culture from the corneal specimen is significant or not is determined by one of the following criteria;3,13
- Confluent growth (more than 10 colonies) on the site of inoculation on solid culture media
- Growth on one solid media consistent with the microscopic demonstration of the organism in the clinical sample (appropriate staining and morphology)
- Growth of the same organism in more than one medium
- Growth of the organism from a repeated clinical sample
Although, being a standard procedure to diagnose fungal keratitis, the sensitivity of culture ranges from 57% to 68%.18,19Disadvantages of culture includes the length of time required for positive detection, a small amount of clinical material available from corneal scraping and false negatives as a result of prior treatment before scraping.19
|
Methods |
Comments |
|
Direct Microscopy |
Other less sensitive and specific are
|
|
Culture |
|
|
In vivo Confocal microscopy |
|
|
Molecular Diagnostic methods (Polymerase Chain Reaction) |
|
|
Histopathology |
|
Antifungal susceptibility testing
It can be done using the disc diffusion test. To determine the minimum inhibitory concentration (MIC) of antifungal drugs, broth or agar dilution methods can be used. Drugs that are commonly tested include 5FC (flucytosine), ketoconazole, miconazole, fluconazole, itraconazole, and Amphotericin B. It has been shown that the MIC of azoles and caspofungin is lowest against Aspergillus species, while voriconazole, amphotericin B and posaconazole have the lowest MIC against Fusarium species.3 However, the data of invitro susceptibility testing is not enough to incorporate or highlight their role in routine practice.3
In vivo confocal microscopy
It is a noninvasive tool to image the anterior segment of the eye. The cornea can be examined at multiple levels at variable magnifications. The sensitivity and specificity of in vivo confocal microscopy for the diagnosis of fungal keratitis in relation to the microbiological diagnosis, taking into consideration the inter and intra-observer variation, was found to be 88.3% and 91.1%, respectively.3,23 However, it has been advised that this modality cannot be used as a standalone for the diagnosis of fungal keratitis.3
On confocal microscopy with Heidelberg Retina Tomograph II-Rostock Cornea Module (HRTII-RCM), Fusarium hyphae appear as a high contrast line 200-300 µm in length and 3-5 µm in width, double-walled with septations and branches at 90° angles.23,24 Aspergillus hyphae appear as high-contrast lines 200–300 µm in length and 3–5 µm in width, double-walled with septations and branches at 45° angles.23,24 Candida pseudofilaments appears high-contrast elongated particles measuring 10–40 µm in length and 5–10 µm in width.24 The fungal elements are of uniform width with irregular branching pattern and are not seen in isolation. There are round to oval bright structures surrounding fungal elements, representing inflammatory cell infiltration. Heidelberg Retina Tomograph II-Rostock Cornea Module is better than the first generation confocal microscopes as it provides information of the epithelial tissues and peripheral structures of the cornea, has higher resolution and higher magnification (800x).24 These features increase the sensitivity of the imaging modality and can aid in the early diagnosis of fungal keratitis, thus providing a base to start early empirical therapy which may lead to a better prognosis.
There are case reports which suggests that confocal microscopy can be used to differentiate between ongoing infection and toxic effects of topical medications.25 This can help to modify the treatment according to the situation.
Fungal filaments in confocal microscopy should be differentiated from the other filamentous structures in the cornea by above-mentioned features and also characteristic features of the other filamentous structures. For example, Nocardia appears as a thin, beaded filamentous structure with right-angled branching.26 Corneal nerves are seen as bright, elongated, uniform (stromal nerves), or beaded (subbasal plexus) structures with acute-angled, regular branching pattern and 5 µm - 20 µm in thickness.26
To summarize, in vivo confocal microscopy can be useful in the following scenarios:23
- In routine keratitis cases for early identification of fungal etiology and early institution of specific therapy
- Deep infiltrates present in mid or posterior stroma not accessible to corneal scraping and require corneal biopsy
- When the patient is on antifungal medications which will render the anterior stroma sterile while the organism is still present in the posterior stroma
- Fungal keratitis after intracorneal implants like ring segments
- Fungal keratitis of the interface after LASIK surgery
- Fungal keratitis after radial or astigmatic keratotomy as the infiltrates are usually not anterior stromal
- Healing keratitis cases as the epithelium heals despite active underlying infiltrates,7 which can be identified by confocal scanning
Molecular methods
Though the molecular methods like polymerase chain reaction (PCR) are more specific and sensitive, they are to be used as an adjunct only, instead of replacing the microscopy and cultures for diagnosing fungal keratitis. PCR targeting different ribosomal DNA based primers of different regions 18S rDNA, 28s rDNA and internal transcribed spacer region (ITS) have been used to evaluate fungal keratitis cases. Out of the above three, ITS region is found to be significantly more sensitive than the other two for the diagnosis of fungal keratitis.3 Hence, ITS has become the ideal region to determine sequence-based identification of different species of fungi.3
PCR has the advantage that it is rapid, and needs only small amount of clinical sample but the test detects both viable and non-viable fungi. Microbiological culture diagnosis of fungus at genus and species level requires at least 3 and 7 days, respectively. However, PCR can give genus and species level identification within 24hours.27 The sensitivity, specificity, negative and positive predictive values for PCR in the study done by Badiee P et al.18 was found to be 75, 70, 50 and 87.5%, respectively.
As the fungal keratitis is caused by a larger number of saprophytic fungi, PCR is carried out in two ways either using universal fungal primers and DNA sequencing methods or using multicopy gene targets for the detection of fungal pathogens. However, due to limitation of false positive results, high cost, limited availability, this modality is currently used as a complementary tool for the diagnosis of fungal keratitis.18
Histopathology
Histopathological examination can also be used in diagnosis or as an add-on, especially post therapeutic penetrating keratoplasty. Various stains that can be used are Haematoxylin and Eosin, Gomori methanamine Silver stain(GMS) and Periodic acid Schiff(PAS). Fungal keratitis usually involves anterior two-third of the stroma first and later, as the disease progresses, posterior stroma, Descemets membrane and endothelium gets involved.28 Identification of fugus is easier with special stains like PAS and GMS; however, they may be not useful in advanced stages of the disease with severe necrosis and perforation.28 The fungal filaments measure upto 10µm in diameter with variable lengths and can have variable appearance.28
Findings observed on histopathology in fungal keratitis are fungal filaments, inflammatory cells, vascularisation and occasionally granulomatous inflammation. It is said that the distribution of fungal filaments is inversely correlated with the density of inflammatory cells.28 In case of dematiaceous fungus, a carpet like growth of fungal filaments on the surface can be seen.28
Treatment
Fungal keratitis is usually difficult to manage because of the delay in the identification by culture methods, limited commercial availability of antifungal agents, less ocular penetration of antifungals, the propensity of the fungus to invade intact Descemet membrane and differential susceptibility of the different causative organisms to the available antifungal agents. Resolution is usually slow and the duration of treatment can average for around 80 days.19
The prognosis and success of the treatment of Fungal keratitis depend upon the rapid identification of pathogens and the initiation of appropriate antifungal medication.
Response to treatment is assessed by improvement in symptoms, blunting of feathery edges, re-epithelialization, or reduction in the anterior chamber reaction.7 After about 1 week, if there is no improvement or worsening after initiation of medical therapy, the diagnosis should be reconsidered or changing to a different class of antifungal should be considered and/or antifungal susceptibility should be obtained from the microbiological laboratory.7
Medical therapy
Topical therapy
Topical therapy is the current standard for the treatment of fungal keratitis.
However, the two major concerns in the medical management of fungal keratitis is absence of data regarding drug susceptibility and poor topical penetration of the drugs.
Polyenes
Antifungals included in this group are Nystatin, Natamycin, and Amphotericin B. These binds to ergosterol present in the fungal cell membrane and lead to the formation of pores which disrupts the homeostasis of the fungus and leads to cell death. They have a concentration-dependent killing action. The ideal dosing schedule is not determined; however, it is used half-hourly for initial loading dose with a gradual reduction to six to eight times a day. They have poor penetration through the intact epithelium. Nystatin was the first antifungal discovered but has limited use now because of its toxicity and poor penetration.19
Natamycin was the only antifungal drug approved by Food and Drug administration in 1960s for the topical use in the cases of filamentous fungi, and remains the most popular treatment even now.19It has a slightly different mechanism of action, it acts by inhibition of amino acids and glucose transport across plasma membrane through ergosterol specific and reversible inhibition of membrane transport protein. Natamycin is the drug of choice for filamentous fungi, but the penetration of the drug is poor.20
It is available as a 5% suspension for topical use. Disadvantages includes poor penetration, hourly instillation and corneal deposits. Some clinicians have advised for periodic epithelial debridement for allowing better penetration of this drug.19 But, Prajna et al.29 have shown in their study that periodic epithelial debridement worsens the visual acuity outcome although it was not statistically significant. The adverse effect of topical natamycin are few and include deposition of suspended particles on the ulcer and ocular surface, mild irritation, redness, foreign body sensation, stinging, burning sensation, and tearing.
Amphotericin B is poorly soluble in water and is highly photosensitive; hence, it should be stored in dark bottles in refrigerator(2-8°C).30 It should only be given by topical route as systemic administration is associated with adverse effects. It is commonly used as a 0.15% solution prepared by reconstituting the parenteral formulation. It is the drug of choice for Candida keratitis.19 It can also be administered via intravenous route in a dose of 0.5-0.7mg/kg. It also has poor penetration through intact corneal epithelium. Other routes for administration include oral, subconjunctival, intracameral, intrastromal, intravitreal or intravenous. Subconjunctival administration in non-responsive cases is controversial as some reports have shown toxicity while others have shown favorable outcome without toxicity.19
It is available in deoxycholate form or lipophilic form. However, use of amphotericin B alone in the treatment of mycotic keratitis lacks the necessary studies to prove its effectiveness and hence is not advisable to use alone.30 But a recent randomized trial on collagen cross linking for moderate fungal keratitis also compared the effect of topical natamycin and amphotericin alone; and the authors have concluded that amphotericin alone can be used in the cases of mild to moderate filamentous fungal keratitis.31 Parenteral route can be used but is associated with renal toxicity. However, lipophilic form is less renal toxic. It is labelled as category B drug for pregnancy and is the safest antifungal drug for pregnancy.30
Azoles
It consists of two groups: Imidazoles and Triazoles. Azoles inhibit the synthesis of ergosterol through action on cytochrome P-450 dependent enzyme. Ergosterol is an essential component of fungal cell wall. They have concentration independent killing (time dependent). They are mainly fungistatic but can be fungicidal at high concentrations or growth phase of fungus.
Imidazoles include clotrimazole, econazole, miconazole, oxiconazole and ketoconazole; triazoles include fluconazole, itraconazole, voriconazole, posaconazole and ravuconazole. Triazoles have longer half-life and better penetration in intact corneal epithelium than imidazoles. Various azoles have different effectiveness towards fungus. Econazole has found to be as effective as natamycin for Fusarium spp.19
Voriconazole (VCZ), a derivative of fluconazole, is available in topical and oral formulations. It is approved by FDA for oral use in systemic fungal infections, 1% topical solution is off label use. It is known to have good systemic penetration into the eye.29 It is a broad spectrum antifungal efficacious against filamentous fungi and Candida, having efficacy similar to natamycin according to some studies.19 Two hourly dosing regimen does not achieve MIC levels to eradicate Fusarium spp.; however, it is sufficient to treat Candida and Aspergillus keratitis.30 Hence, hourly dosing is necessary in Fusarium keratitis cases. Topical formulation is known to cause contact dermatitis around the eye. Based on the currently available literature, VCZ is the alternative drug to use in recalcitrant cases of fungal keratitis, especially not responding to natamycin and can be used as an adjuvant also.
Posaconazole is relatively new and the only available in oral tablet form. Data for its efficacy in fungal keratitis is still limited. However, it can be used in cases of mycotic keratitis not responding to conventional antifungal therapy.30 It is teratogenic and has been labelled as a category C drug by FDA for use in pregnancy.30
It is imperative to choose appropriate antifungal medication for successful treatment of fungal keratitis. Various studies and randomized trials have been done to determine the efficacy of one antifungal over other. Voriconazole was being considered as a promising new first line antifungal medication instead of natamycin, given the theoretical advantage of having better penetration and less toxicity. Various trials have proven that there is no benefit of voriconazole over natamycin in terms of final visual acuity, scar size or adverse events like perforation.29 The Mycotic ulcer treatment trial (MUTT), have shown that natamycin is superior to voriconazole in terms of final visual acuity, and the rate of perforation and resorting to therapeutic keratoplasty, especially in case of Fusarium keratitis.32 Also, after 6 days of natamycin treatment the culture positivity rate is significantly reduced as compared to voriconazole among Fusarium keratitis. But the cases with Aspergillus keratitis in the study fared equal with both the antifungals. Thus, the author of the study have concluded that voriconazole should not be considered as a monotherapy in filamentous keratitis cases. Usual dosing schedule for topical antifungals is hourly for first week or till epithelial healing and then 6-8 times/day for 3 weeks or till the complete resolution.
Echinocandins
Echinocandins which include Anidulafungin, Micafungin and Caspofungin, inhibits 1,3-b glucan synthase which affects fungal cell wall. They have concentration dependent killing. They are active against Candida spp. and 0.5% Caspofungin topical solution is found to be as efficacious as 0.15% amphotericin B in some studies.19 In general, this group of drugs are effective against yeasts and Aspergillus spp and ineffective against Fusarium spp.
Caspofungin and Itraconazole are completely ineffective against Fusarium spp.20 However, Aspergillus was found to be significantly sensitive to itraconazole. In vitro studies have found that, except for natamycin and amphotericin B, the concentration of antifungals required to inhibit Fusarium spp. was higher than that for Aspergillus spp.20
Fluorinated pyrimidines (Fluocytosine) blocks fungal thymidine synthesis and have concentration dependent killing. Allyamine (Terbinafine) inhibits squalene epoxidase in the fungal membrane which leads to inhibition and or alteration of ergosterol biosynthesis. It has a concentration dependent killing and is effective against Aspergillus, Fusarium, Scedosporium and Candida.
It is popular practice to start single antifungal agent especially in filamentous keratitis cases as suggested by MUTT 1 trial.32 However, some studies advocates use of combination therapy in cases that are unresponsive to monotherapy.33 Natamycin and voriconazole is the most common combination used for filamentous keratitis, while amphotericin B and voriconazole was found to be more commonly used for yeasts.33
In vitro study done by Prajna L et al.34 have shown no synergistic action of amphotericin B and Natamycin combination in filamentous fungal keratitis. Though there are advantages of having a combination therapy, but amphotericin and natamycin being from the same group have almost similar mode of action and the author does not suggest the use of this combination.
Another interesting finding observed by Rees et al.35 in their in vitro study was that Ethylene diamine tetraacetic acid, timolol and 5-Fluorouracil have synergistic actions with natamycin and voriconazole especially over fusarium isolate. The mechanism of interaction is not clearly understood and the study is limited by its in vitro nature.
The outcome of fungal keratitis correlates with the in vitro susceptibility of the organism to the antifungal.36 Poor susceptibility to natamycin has been found to be associated with larger scar/ infiltrate size at 3 months and increased odds of perforation and /or therapeutic penetrating keratoplasty(TPK). As of now, Clinical and Laboratory Standards Institute (CLSI) has not proposed minimum inhibitory concentrations of topical antifungals for filamentous keratitis, instead values given by epidemiological cut off values (ECV) can be used as guide to the susceptibility of available antifungals for the treatment of fungal keratitis.36 However, this study was not able to identify any correlation between voriconazole susceptibility and outcome.
Table 1. Antifungals topical concentrations, oral dosage and spectrum of activity
|
Topical concentrations |
Oral dosage |
Spectrum of activity |
|
|
Polyenes |
|||
|
Amphotericin B |
0.05-.30% |
Not available |
First line therapy for Candida species. Good to moderate activity against Aspergillus, Fusarium species |
|
Natamycin |
2.5-5% |
Not available |
Good activity against most Fusarium, Aspergillus, less effective against Candida species |
|
Azoles |
|||
|
Clotrimazole |
1% topical; 1% cream |
Good activity against Candida and some Aspergillus species |
|
|
Econazole |
0.02-2% |
50-100 mg/day |
Effective against Fusarium, Aspergillus, and Candida species |
|
Fluconazole |
0.5-1% |
100-400 mg/day |
Effective against yeast, minimal Activity against filamentous fungi |
|
Ketoconazole |
1-2% |
200-400 mg/day |
Effective against Candida, Aspergillus, limited effectiveness against Fusarium species. |
|
Itraconazole |
1% |
200-400 mg/day |
Aspergillus, Candida species, not effective against Fusarium |
|
Miconazole |
1% |
Effective against Paecilomyces, Scedosporium species |
|
|
Posaconazole |
200 mg x3/day |
Limited information, has been used as savage rescue for Fusarium, Scedosporium. Spectrum similar to voriconazole |
|
|
Voriconazole |
1-2% |
200-400 mg/day |
Candida species, Aspergillus species. Non Fusarium solani species, Scedosporiumn apiospermum |
|
Echinocandins |
|||
|
Capsofungin |
0.5% |
Yeasts, Aspergillus species; not effective against Fusarium species |
|
|
Micafungin |
0.1% |
||
|
Fluorinated pyrimidines |
|||
|
Flucytosine |
25-37.5 mg/ kg x4 |
Effective against yeasts, Some Aspergillus species and Cladosporium species |
|
|
Allyamine |
|||
|
Terbinafine |
0.25% |
250 mg/day |
Aspergillus, Fusarium, Scedosporium, and Candida. |
All the topical antifungals have been studied at some point for their usefulness in fungal keratitis cases, however, the data are limited to draw a conclusion regarding their efficacy. Most of the studies have focused on amphotericin B, natamycin and voriconazole and hence a reliable evidence based conclusion regarding their efficacy, safety and clinical indication is available.
Other agents
In developing world where antifungal agents availability is limited, general antiseptics like chlorhexidine gluconate, topical silver sulphadiazine have been used with good efficacy according to some reports.19
Systemic therapy
Topical therapy is the mainstay of fungal keratitis, however, due to poor penetration severe or recalcitrant cases may require combination of topical and systemic therapy. Limited data is available to determine the efficacy of addition of systemic antifungal medications (oral or intravenous) in the treatment of fungal keratitis cases. Very few randomised trials have been done to determine the efficacy of oral adjunct antifungals in the treatment of fungal keratitis and have not shown any benefit of systemic therapy.37,38 The rationale behind the use of oral drug is that the topical drugs having poor penetration can lead to intervals of subtherapeutic levels of drugs in between the doses and giving oral adjuvant can help maintain a constant therapeutic levels in the aqueous.30,38
Systemic therapy is used as an adjunct in recalcitrant cases, in patients with ulcers >5mm or involvement of >50% of stromal depth, scleral or limbal involvement, endophthalmitis, impending perforation, or perforated corneal ulcer; pediatric cases; and post keratoplasty cases.30,33,39 Commonly used oral antifungals include Ketoconazole (KCZ), Itraconazole, Fluconazole and Voriconazole (VCZ).30,39
Ketoconazole is inexpensive and have good tissue distribution. Peak concentration in serum of 2-3µg/ml is achieved after 2-3 hours of 200mg oral dose. Its highly protein bound and it should be taken with a meal.30 Hepatotoxicity is a major side effect of KCZ. Hence, liver function tests should be monitored every 3 weeks. It also causes gastrointestinal, cardiac and dermatological side effects. It has been labeled as category C drug for use in pregnancy by the FDA.
In a study it was found that oral KCZ has no added benefit in cases of fungal keratitis when given as an adjunct to topical therapy, probably due to serum concentration achieved being lower than MIC.37
Voriconazole oral bioavailability is 90% and plasma protein binding is moderate, about 58%.30 It is metabolized by the liver and hence liver enzymes should be monitored at the outset and during therapy.30 It should be taken either one hour before or two hours after the meal to improve absorption. VCZ achieves more than MIC levels in plasma, aqueous and vitreous after oral administration.30The adverse effects of VCZ include visual disturbances, color vision changes, and photophobia. It is a category D drug and is contraindicated in pregnancy as it is teratogenic and causes skeletal and visceral abnormalities.30
In a study done by Sharma et al.39, the authors have compared the outcomes of adjunct oral VCZ with oral KCZ in fungal keratitis cases. They have found that cases treated with adjunct VCZ have a better outcome in terms of visual acuity and scar size as compared to the ones treated with adjunct oral KCZ, due to better in vitro profile and higher ocular tissue/ tear drug concentration achieved with oral VCZ. Also, the systemic side effects with VCZ are less than KCZ. Both are given in a dose of 200mg twice a day. Hence, it is recommended that depending on the local microbiological prevalence profile and availability of the drug, oral VCZ can be considered as first-line of oral adjunct in severe fungal keratitis cases, especially if Aspergillus spp. is the causative agent.39
The MUTT II trial, which investigated the use of adjunct oral voriconazole for the treatment of severe filamentous fungal keratitis, have concluded that there is no benefit of adding oral voriconazole.38 However, in a subsequent subgroup analysis, they have found a favorable outcome (not statistically significant) in severe Fusarium keratitis cases in terms of decrease in the rate of perforation by adding adjuvant oral voriconazole. Whatsoever, the exclusion of cases with scleral or limbal involvement, bilateral ulcers, endophthalmitis, impending perforation or perforated corneal ulcer, recalcitrant mycotic keratitis; pediatric cases; and post keratoplasty cases in MUTT II study still put the question mark on the efficacy of systemic antifungals in these severe cases.
Fluconazole has been found to have high ocular tissue concentration due to less protein binding and low lipophilicity. In a study, it was found that fluconazole has higher ocular tissue concentration than KCZ and itraconazole in the normal and inflamed eyes.30 It has been labeled as category D drug for use in pregnancy by the FDA and hence should not be used.30
Other drug delivery methods (targeted drug delivery)
It is not possible to achieve the therapeutic drug concentrations of antifungals in the deep stromal level or aqueous due to poor penetration of the topical drugs. There are some methods that have been tried to deliver the drug at the site of interest to achieve therapeutic concentrations. These methods are usually adopted when conventional therapy with topical and oral antifungals is ineffective and the keratitis is deep. Targeted drug delivery can be achieved with the following methods
Intrastromal injections
Drugs commonly used for intrastromal injections are Voriconazole, amphotericin B and natamycin. Among these voriconazole is most commonly used.
Intrastromal voriconazole: It is given in the dose of 50 - 100µg/0.1ml around the ulcer. It is given with a tuberculin syringe having a 30 gauge needle and administered as a depot in five areas around the ulcer like a barrage. The needle is inserted in a bevel down position. The injection should be inserted from a clear area and progressed towards the ulcer. These areas of injections acts as a depot from where drug is released slowly. In case of suboptimal response, 3 injections can be given 72 hours apart.
Complications of intrastromal injections include new focus of infection can form at the needle track, inadvertent anterior chamber entry, intrastromal haemorrhage and hyphaema. Intrastromal voriconazole did show some benefit in refractory deep mycotic keratitis, however, data is still limited.30
In a randomized trial, comparing intrastromal voriconazole and topical 5% natamycin with topical 5% natamycin alone in cases of moderate to severe fungal keratitis, it was found that the group receiving intrastromal voriconazole, in addition, topical natamycin, though not statistically significant, had more culture positivity after 1 week, worse visual outcome, increased scar size and increased rate of perforation.40 Hence, the authors suggests that intrastromal voriconazole has no beneficial effect in cases of moderate to severe filamentous fungal keratitis.
Intracameral injections
Intracameral amphotericin B: Considered in deep keratitis, associated with severe anterior chamber reaction, and recalcitrant mycotic keratitis. It is given in a dose of 5-10µg/0.1ml and can be repeated after 72 hours in case of a suboptimal response. The adverse effects include cataract (anterior subcapsular), iritis, hyphaema, and corneal edema. There are various case reports and short series which demonstrate effectiveness and safety of intracameral amphotericin B in refractory cases of deep mycotic keratitis or with endophthalmitis; however, a randomized trial did not suggest any additional benefit of intracameral amphotericin over routine topical and systemic antifungal therapy.30
Intracameral Voriconazole: It is used in a dose of 50µg/0.1ml. The advantage over amphotericin B is that it has a broad spectrum of activity. Its half-life in the anterior chamber is short, hence it requires more frequent dosing. Side effects are similar to the intracameral amphotericin B but less toxic.30
In general, intracameral injections can have the following complications; breach of natural barriers of infection, the deeper spread of infection, the risk of iatrogenic spread to the site of injection, intraocular inflammation, lenticular damage, glaucoma, hyphema, and potential endothelial damage.
Subconjunctival injections
There are studies which have shown the efficacy of subconjunctival injections however, this method is limited due to its toxicity and corneal melt.30
Nanoparticle-based drug delivery
Most of the current clinical evidence on the nanoparticle-based drug delivery is based on the animal model. Further human studies are needed to determine its importance.
Drug-eluting contact lenses
A layer of the tear film is formed between the contact lens and the cornea in which there are minimal tear mixing and exchange. The drug delivered to this tear film layer through the contact lens will remain in contact with the cornea for a longer period of time and hence can achieve a higher concentration of drug in the stroma as compared to topical medication. The amount of drug delivered depends upon the contact lens material and other properties. There are very few contact lens-based drug delivery system available commercially and the usefulness in mycotic keratitis is uncertain.30
After the MUTT I and MUTT II trial, there was a dilemma, especially in treating severe fungal keratitis cases not responding to natamycin, regarding the use of routinely available topical or systemic antifungals. Therefore, to ease this a little bit Sharma et al.41 proposed a topical, systemic, and targeted therapy (TST) protocol (Flowchart 141) for managing fungal keratitis cases with available modes of treatment. Though there was no comparative arm available in the study, they have found that the success rate with this protocol was about 79.8% which is comparable with other studies in the management of fungal keratitis.41
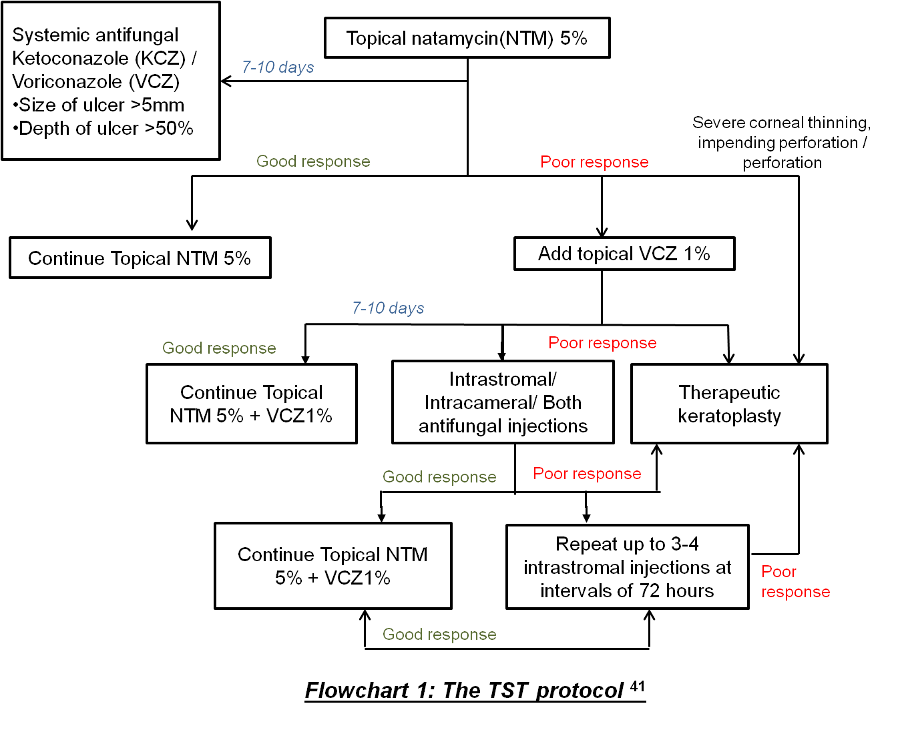
The success of medical therapy depends upon the clinical characteristics of the ulcer like the presence of hypopyon/ endothelial exudates, deep infiltration, prior topical steroid use, vertically oriented hyphae in the corneal stroma seen by histopathology; all these factors are associated with treatment failure leading to corneal perforation and therapeutic penetrating keratoplasty.42
To understand better the predictors of outcome in fungal keratitis cases a prospective study was done by Prajna et al. 43 which took various demographical and clinical features into considerations. They have suggested that the final visual outcome at 3 months is worse if the initial BSCVA at presentation is not good, infiltrate size is large, male gender, old age, and pigmented lesion. Old age was also found to be a significant factor for delayed re-epithelialization. Large ulcers have higher chances of perforation. This study emphasized the importance of initial BSCVA, which can help to guide the treatment and in counseling of the patients regarding the prognosis.
Collagen cross-linking (CXL) in fungal keratitis
Application of CXL for keratitis along with the photoactivated chromophore (i.e. riboflavin) is known as Photoactivated Chromophore for keratitis (PACK-CXL).44 Three main mechanisms of action have been postulated for its effect, (i) Chromosomal damage inhibiting organisms replication, (ii) release of reactive oxygen species which damages the pathogen cell wall and (iii) increase of covalent bond between collagen fibers causing reduced enzymatic degradation and melt.44 Usually it is done using the Dresden protocol (365nm UV-A; 3mW/cm2; 30 minutes); however, the accelerated protocol has also been tried with unfavorable results.44 However, the effect of PACK-CXL in fungal keratitis is still inconclusive and has been shown to be effective in early cases.44 Based on the available literature, its use in the treatment of fungal keratitis, as an adjuvant, is still controversial
In a recent randomized trial done by Prajna et al.31 on CXL assisted infection reduction (CLAIR), it was found that CXL was not beneficial as an adjuvant to topical medication, natamycin or amphotericin, in treatment of moderate cases of fungal keratitis. They found no benefit in the rate of epithelial healing, scar/infiltrate size, and no difference in the rate of adverse events like corneal perforation/ Therapeutic keratoplasty. However, on the contrary, their result suggests a negative effect of CXL on the final visual outcome.
Surgical Treatment
Medical therapy is considered as the mainstay in the management of fungal keratitis cases, however, cases that do not respond to medical therapy or have impending perforation requires surgical management. The range of surgical options varies from lamellar keratoplasty or penetrating keratoplasty to evisceration or enucleation or exenteration.19 Even after surgical therapy with lamellar or penetrating keratoplasty recurrence is common and is reported to be in the range of 6.34% - 20%.19,45 However, there was no significant difference in the recurrence rates between the two procedures(DALK and TPK).19
DALK performed with big bubble technique has good visual and therapeutic outcomes which are comparable to penetrating keratoplasty.45 DALK can be performed in the cases having deep mycotic keratitis not responding to conventional treatment and not involving the endothelial layer of the cornea. The most common complication associated with DALK surgery in intraoperative perforation of DM. The decision to convert into a full-thickness penetrating keratoplasty or continue with the DALK depends on the size of perforation and surgeon's expertise. Double anterior chamber, recurrence, and rejection are other complications, although chances of rejection are less than the full thickness penetrating keratoplasty. It is important to remember that if anterior lamellar keratoplasty is done without using a big bubble or not completely removing the stroma then it should be ensured that the host tissue remaining is free of any infective foci.45,46 Intraoperatively, the trephination of the host tissue should be 0.5mm larger than the area of fungal infection to reduce the chance of recurrence.
Fungal filaments in the corneal stroma tend to grow either perpendicular to the corneal stromal lamellae or parallel to them.45–47 When the filaments grow perpendicularly there are high chances of DM and endothelial penetration and perforation and this could be one of the limiting causes in surgeons preferring to do therapeutic penetrating keratoplasty rather than lamellar keratoplasty.47
Recurrence after LK usually occurs in the first two weeks of surgery.47 Risk of treatment failure increases or is considered to be more if the fungal hyphae have growth pattern perpendicular to corneal lamellae if the patient has used preoperative topical steroids if there was the presence of hypopyon or endothelial plaque preoperatively if the host tissue was not properly trephined or dissected to clear the infected tissue.47 Hence, the first two weeks are important after surgical therapy for fungal keratitis and topical and oral antifungals should be continued and topical steroids should be avoided during this period.47
Therapeutic penetrating keratoplasty is performed in 12.1% to 38% of cases with fungal keratitis.48A cure rate of 69%-90% has been found and the recurrence rate ranges from 6.8% to 50%.48 The recurrence rate is more in cases with preoperative hypopyon, corneal perforation, and infection spreading to the limbus and lenticular abscess needing lens extraction.48 In some cases, with non-severe superficial fungal keratitis, superficial keratectomy can be done with or without amniotic membrane grafting as an alternative to keratoplasty.19
In summary, fungal keratitis is a difficult entity to diagnose and treat and early diagnosis with appropriate topical and systemic antifungals is associated with better outcomes.

.jpg)