Endophthalmitis is a potentially devastating condition of intraocular procedures. It is an inflammation of internal coats of the eye along with inflammation of intraocular cavities i.e. aqueous and vitreous humour that occur as a result of intraocular colonization of microorganisms. It is one of the most feared complications of cataract extraction and other intraocular surgeries. Rarely, infectious endophthalmitis is the presenting feature of an underlying systemic infection. The incidence of endophthalmitis has sharply declined within two to three decades because of variations in surgical technique, scrupulous asepsis, and use of preoperative and post-operative broad-spectrum antibiotics.
Classification Of Endophthalmitis:
General categories include postoperative endophthalmitis (acute, delayed-onset, conjunctival filtering–bleb associated, glaucoma drainage devices associated), posttraumatic endophthalmitis (blunt or penetrating ocular trauma, foreign bodies), endophthalmitis associated with intravitreal injection, microbial keratitis, endogenous endophthalmitis, and miscellaneous non-infectious causes of endophthalmitis (sterile uveitis, phacoanaphylactic endophthalmitis, sympathetic ophthalmia).
Cataract Surgery:
Post-operative endophthalmitis following cataract surgery can be categorized on the basis of time of onset into fulminant (<4 days), acute (2-11 days), sub-acute (2 weeks- 6 weeks) or chronic (>6 weeks). The reported rate of post-operative endophthalmitis varies between a range of 0.04%–0.2%. (1) Gram-positive organisms account for 90% or more of pathogens isolated in culture-positive cases of postoperative endophthalmitis following cataract surgery, with coagulase-negative Staphylococci (Staphylococcus epidermidis) and Staphylococcus aureus being the leading causes. Other gram-positive organisms are Bacillus cereus andEnterococcus faecalis. However, in India, Gram-negative organisms (Pseudomonas aeruginosa, Haemophilus influenzae, Serratia, Proteus) and fungi (Candida, aspergillus, Histoplasma, Blastomyces) are important in etiology. Gram-negative bacteria, Pseudomonas aeruginosa, Streptococcus pneumonia, Staphylococcus aureus are known to cause fulminant post-operative endophthalmitis. Coagulase-negative staphylococci, Staphylococcus aureus, streptococcus species, and Gram-negative bacteria cause acute post-operative endophthalmitis. In subacute/delayed onset endophthalmitis, Propionibacterium acnes is the most commonly implicated pathogen, accounting for nearly 40% of isolates. Other organisms include Staphylococcus epidermidis, fungi (Candida parapsilosis) and Corynebacterium species. Streptococcus species, Enterococci, Hemophilus influenza, Staphylococcus species, and fungi are known to cause chronic/ late-onset post-operative endophthalmitis. In a recent study by Stevenson et al, microbialkeratitisand Pseudomonas aeruginosa were the most common precipitating event and causative organisms in gram-negative endophthalmitis. (2)
The severity of endophthalmitis has been related generally with the virulence of the offending pathogen.The inflammatory capacity of gram-positive cell walls and their components (peptidoglycan, lipoteichoic acid, and capsular polysaccharide) is well documented. Toxins, intraocular localization, and, to a lesser extent, the intraocular host response to cell walls all can contribute to the pathogenesis ofB. cereus, S. aureus and E. faecalis endophthalmitis in a pathogen-specific manner. These organismspossess similar general biochemical traits (i.e., cell wall composition and toxin production), however, infections caused by them are not in a similar manner.Both B. cereusandS. aureusproduce multiple toxins i.e. pore-forming and cell membrane destabilizing. A study by Callegan et al have suggested that pore-forming toxins are of primary importance in terms of retinal function inS. aureusendophthalmitis, whereas secreted products other than hemolysin BL may be more important inB. cereusendophthalmitis. (3) They reported that thoughS. aureusandE. faecalisinfections were clinically similar to one another andB. cereusintraocular infections were more severe. Another important difference observed was that ofB. cereusmotility and migration into retinal tissues and into the anterior segment, a phenomenon that was not observed for S. aureusorE. faecalis. The pathogenic capability of gram-negative bacteria is often associated with certain components of their cell membrane. The outer leaflet of this membrane comprises a complex lipopolysaccharide (LPS) whoselipidportion acts as an endotoxin that can cause a toxic reaction.
Preoperative risk factors include advanced age (>85 years old), rural residence, male sex, and immunosuppressive states such as diabetes. (4-6)Performing cataract surgery on the same day rather than one day after admission may be accompanied by a higher risk of endophthalmitis. (7) Intraoperative risk factors include intracapsular and extracapsular cataract surgery, defects in the surgical wound, violation of lens capsule, vitreous loss, and anterior vitrectomy. Silicone and polymethyl methacrylate (PMMA) intraocular lens (IOL) optic material compared to acrylic are associated with higher rates of endophthalmitis. The absence of an injector system for these IOLs and the need for a larger incision for non-foldable PMMA lens can be related for this association. (8) The patient’s own periocular flora is a source of infection in majority of the cases. However, there are many other sources that may play a role in the occurrence of endophthalmitis, i.e. the surgeon’s hands, instruments, airborne pathogens (either from the surgeon’s nose or the air conditioner) and solutions injected into the eye. Post-op risk factors include starting topical antibiotics the day after surgery instead of the same day as surgery, not patching following surgery, and using older generations of fluoroquinolone antibiotics. (9-11) 5% topical povidone-iodine into the conjunctival sac is the most important prophylaxis to prevent post-cataract surgeryendophthalmitis. (12) In a study by Carrim et al, 30% of conjunctival cultures were positive after 5% povidone-iodine was applied for 3 minutes. (13) Hosseini et al reported that theuse of 5%povidone-iodinefor 15 minutes can prevent the growth of most post-cataract surgeryendophthalmitisbacterial isolates. (14) An outbreak of post-cataract surgeryendophthalmitis was reported by Pinna et al in which Pseudomonas aeruginosa was isolated from 20 eyes with postoperativeendophthalmitis, phacoemulsifier's internal tubes, povidone-iodinesolution, and operating theatre air-conditioning system.(15) They suggested the use of small sterile single-use bottles to minimize microbial contamination of the povidone-iodine solution.
Clinical features:
The patient presents with ocular pain, redness, watering, headache, diminution of vision, and lid edema. There is an increased number of cells in the anterior chamber, presence of vitreous exudates and decrease in fundal glow. Hypopyon may or may not be present. TheColour of the hypopyon may give a clue to the causative agent. The presence of a pinkhypopyonin the absence of hyphaema may suggest the diagnosis ofSerratiamarcescensendophthalmitis. (16) Pain is an important symptom, it is not universal. Patients with bacterial endophthalmitis are more symptomatic as compared to fungal endophthalmitis. It is important to differentiate infective endophthalmitis from sterile post-op inflammation. Toxic Anterior Segment Syndrome (TASS) is an acute post-operative inflammatory reaction in which a non-infectious substance enters the anterior segment and induces toxic damage to the intraocular tissues. The following table describes the differences between TASS and endophthalmitis.
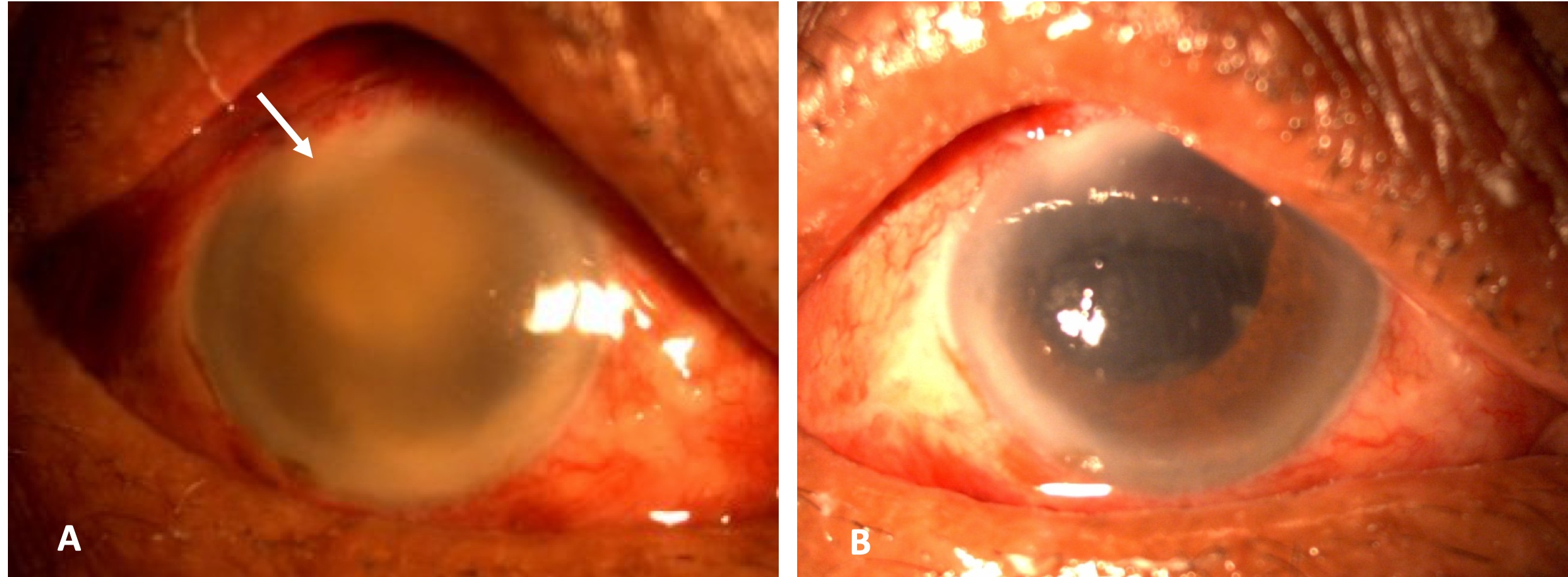
Figure: A. Slit-lamp photograph of post-cataract surgery acute endophthalmitis with severe fibrinous and exudative reaction and corneal infiltration ( white arrow). B. Slit-lamp photograph of the same eye in the postoperative period after undergoing vitrectomy and Intraocular lens removal. The corneal clarity improved after the debulking of the exudates and bacterial load.
|
Toxic Anterior Segment Syndrome |
Endophthalmitis |
|
|
Cause |
Non-infectious reaction to toxic agent present in BSS solution, antibiotic injection, endotoxin residue |
Bacterial, fungal or viral infection |
|
Onset |
12-24 hours |
3-7 days |
|
Pain |
Usually, none but can be mild to moderate |
Usually severe (25% have no pain) |
|
Intraocular pressure (IOP) |
May increase suddenly |
Usually not elevated |
|
Lid swelling |
Usually not evident |
Often present |
|
Corneal edema |
Diffuse, limbus to limbus |
Central |
|
Anterior chamber inflammation |
Mild to severe reaction with cells, flare, fibrin, with or without hypopyon |
Marked inflammatory response with hypopyon (85%). Fibrin is variable. |
|
Vitritis |
Very rare |
Always present |
|
USG B scan |
Anechoeic |
Vitreous echoes |
|
Response to steroids |
Dramatic improvement |
Equivocal |
Rishi et al reported a study on acute postoperative Bacillus cereusendophthalmitismimickingtoxic anterior segment syndrome. (17) They suggested one must closely observe every case ofTASSthat presents with intense pain and extremely high IOP and rule out acute post-operative endophthalmitis owing to Bcereuswith microbiologic testing.
Glaucoma Surgeries:
The incidence of endophthalmitis after glaucoma filtering surgery is estimated to be between 0.12% and 1.2% per year. (18) In a retrospective study of Medicare claims data, the cumulative incidence of endophthalmitis in the first year after placement of glaucoma drainage device was 0.4 percent. (19) Endophthalmitis following glaucoma surgery tends to be delayed and is often associated with prior episodes of blebitis. Diabetes, the use of anti-metabolites as well as inferior bleb location increases risk and hastens the onset of endophthalmitis. The most likely pathogens that can colonize an intact bleb are Streptococcus species and Haemophilus influenzae; however, S. aureus, Pseudomonas, fungi, and other organisms may be causative.
Intravitreal Injections:
A recent study by Mishra et al has reported the incidence of endophthalmitis afterintravitreal injections of antivascular endothelial growth factor agents and triamcinolone acetonide to be 0.131%. (20)The riskmay be higher in diabetics. The majority of the cases are caused by Streptococcus or Staphylococcus species representing commensal flora of ocular adnexa and oropharynx. The risk of infection can be lessened by decreasing oropharyngeal droplet transmission at the time of injection. Bevacizumab is always a source of concern as there is no uniform method; multiple pricks are involved during aliquoting which can lead to cluster endophthalmitis.
Trauma:
The risk of endophthalmitis following open globe injuries ranges from 4.2 to 7%. (21,22) In contrast, following closed globe injuries, endophthalmitis is exceedingly rare. Staphylococcus epidermidis is the most common pathogen, but Bacillus cereus is one of the most aggressive pathogens isolated in these cases. The source of the infection is typically the penetrating material. The rate of infection rises dramatically to 10–15% in the presence of the intraocular foreign body (IOFB) and if the repair is delayed beyond 24 hours of the injury. It is important to rule out any IOFB by ultrasound (in cases of non-metallic and organic IOFBs) and CT scan. MRI is contraindicated since it would result in the migration of a magnetic IOFB. In a study by Rishi et al on post-traumaticendophthalmitisinchildren, they reported that itis more common in boys less than 10 years of age and most often caused by injury with organic matter. (23) Corneal abscess and retinal detachment lead to poor outcomes. They found out that E. fecalis was the most common causative organism. Early vitrectomy resulted in better outcomes.
Endogenous Endophthalmitis:
It is less common than exogenous endophthalmitis and is usually seen in debilitated or immunocompromised patients or in patients with a history of intravenous drug abuse, urinary tract infection or diabetes mellitus. It is the result of hematogenous spread of microorganisms from a distant foci or from contaminated catheters or needles. It accounts for approximately 2–8% of all cases of endophthalmitis. (24,25) In the developed countries, gram-positive organisms (StreptococciandStaphylococci) dominate the infection, whereas gram-negative organisms (Escherichia coli,Neisseria meningitidis, Klebsiella species) are more common in the Asian population. (26,27) Fungal organisms account for more than one half of cases of endogenous endophthalmitis. Candida albicans is the most common cause and accounts for 75% to 80% of cases of fungal endophthalmitis. (28) A complete systemic workup should be done to look for the cause. Complete blood count, ESR (to rule out malignancy, chronic infections, rheumatic disease), cultures (blood and urine), throat swab, stool examination, a swab from indwelling catheter’s tip if any, Chest X-ray, HIV testing can be done. In an Indian case series of endogenous endophthalmitis in children and adolescents by Maitray et al, they reported gram-positive bacteria to be the most common organism followed by gram-negative bacteria, toxocara, fungi, and cysticercus. (29) The differences in endophthalmitis among children and adolescents from that in adults include the relative lack of systemic features and lack of underlying systemic disorders as well as potential misdiagnosis as masquerade syndrome. Early vitrectomy could result in good functional outcomes.
Investigations:
1. Ultrasonography:
USG B scan should be done to assess the posterior segment status. The objective of the USG B scan is to evaluate the eye for the extent of inflammation, vitreous membranes and opacities, presence and location of posterior vitreous detachment, presence of retinal detachment, choroidal detachment or T sign, any retained lens matter or IOFB and retinochoroidal thickening. It helps in monitoring response to treatment.
2. Aqueous tap/ Vitreous tap or biopsy
With the patient in primary gaze, a 30-gauge/27-gauge needle attached to a tuberculin syringe is inserted through the limbus into the anterior chamber tangentially in a lamellar fashion, keeping direction oblique over iris surface away from the lens surface sand 0.1-0.2 ml of aqueous humor is aspirated. A vitreous specimen may be obtained either by a needle tap or by vitreous biopsy. In a vitreous tap, a large-bore needle (22G) is passed through pars plana and 0.2 cc of undiluted vitreous is removed. Aspiration may not provide an adequate sample as vitreous is denser and contain inflammatory membranes. There is also a risk of retinal detachment. The vitreous biopsy is a safer option as it causes less traction on the inflamed retina. In a vitreous biopsy, a syringe attached to the aspiration port of the vitreous cutter hand-piece is used to remove 0.3 to 0.4mL of undiluted vitreous, by using the automated cutting mechanism of the probe and slow, manual aspiration into the syringe. The other advantage with a biopsy over tap is increased tissue yield. Samples should be sent for Grams stain, KOH mount, culture for bacteria and fungi and testing for antibiotic sensitivities. Anaerobic cultures should be kept for at least 14 days in chronic postoperative endophthalmitis as P. acnes takes a longer time to grow. Polymerase chain reaction testing gives faster results as compared to culture. (30) It can be done if facilities are available.
3. Intravitreal Antibiotics And Steroid
The mainstay of treatment for postoperative endophthalmitis is the intravitreal injection of antibiotics. (31) Usually, a combination of two antibiotics is chosen with both gram-positive and gram-negative bacteria coverage. The most commonly used and effective combination at present is Vancomycin (1mg/0.1ml) to cover gram-positive organisms and ceftazidime (2.2mg in 0.1ml) to cover gram-negative organisms. While amikacin (400 μg/0.1 ml) could also be used in place of ceftazidime, it possesses a higher risk of macular toxicity. (32) Some people use an intravitreal injection of steroids (Dexamethasone 400μg/0.1ml) to limit the post-inflammatory effect. The use of intravitreal steroids is controversial. However, steroids should be avoided in suspected or proven cases of fungal endophthalmitis. In patients with resistance to vancomycin, linezolid, a synthetic oxazolidinone antibiotic, may prove useful. Intravitreal doses of 400 μg have been reported to be nontoxic in rabbits. (33) A combination of intravitreal tazobactam- piperacillin (225 µg/0.1 ml)has also been reported to be effective in the treatment of multidrug-resistant Pseudomonas aeruginosa endophthalmitis (34) and enterobacter endophthalmitis. (35)
Rampant use of antibiotics is increasing the sensitivitypatterns and there is an emergence of resistant strains. There are various reports regarding increasing drug resistance among gram-negative bacteria to fluoroquinolones, aminoglycosides, piperacillin-tazobactam, and ceftazidime. (36,37) Enterococci are the second commonest cause of nosocomial catheter-associated infections of the bloodstream and urinary tract and of skin and soft tissues. (38) In a study by Rishi et al, surgery and trauma were the most common causes of E. faecalis endophthalmitis. (39) They considered Vancomycin as the first-line drug in the management of these eyes. In children, broomstick and needle injuries have been reported to be most common. (40) In a recent study by Dave et al, they reported 29 cases of Enterococcus endophthalmitis out which E. casseliflavus was the commonest cause was highly susceptible to vancomycin. (38) However, there are few reports on endophthalmitis caused by vancomycin-resistant enterococci (VRE). One type of VRE,Enterococcus casseliflavusis inherently resistant to vancomycin due to the chromosomally encodedvanCgene which has been reported once as a cause of endogenous endophthalmitis. (41,42) There is another report of exogenous endophthalmitis caused byE casseliflavus. (43) For infections caused by VRE, ampicillin, or amoxicillin are reasonable antibiotic choices if the isolate is susceptible. In the absence of high-level aminoglycoside resistance, ampicillin can be combined with an aminoglycoside for a synergistic effect. Other antibiotics found to be effective include linezolid, daptomycin, and tigecycline. (43) Endophthalmitis caused byVRE is challenging to treat, due to reduced antimicrobial options and the poor penetration of topical and systemic antibiotics into the vitreal space.
Dose and preparation of intravitreal antibiotics for endophthalmitis is given in the following table. Intravitreal antibiotics can be repeated after 48 hours if the infection is not responding or if the antibiotic sensitivity testing shows susceptibility profiles of the microorganisms to be resistant to the antibiotics injected. Caution should be taken to avoid mixing drugs which may get precipitated, for eg. vancomycin and ceftazidime in the same syringe.
|
Serial No. |
Name of antibiotic (available dose) |
Intravitreal dose (per 0.1 ml) |
Salient steps for preparation (1 ml tuberculin syringe) |
|
1 |
Vancomycin (500 mg) |
1 mg |
1. Add 5 ml distilled water 2. Mix well (shake)—100 mg/ml 3. Take 0.1 ml of drug solution 4. Dilute with 0.9 ml of sterile water 5. Mix well (moving air bubble up and down) 6. Discard 0.9 ml 7. Use 0.1 ml for injection |
|
2 |
Ceftazidime (1000 mg) |
2.25 mg |
1. Add 4 ml of distilled water (250 mg/ml) 2. Follow Steps 2–7 as in row 2 Use dexamethasone in Step 4 if indicated |
|
3 |
Amikacin (250 mg/2 ml) |
125 µg |
1. Take 0.1 ml solution (12.5 mg/ 0.1 ml) 2. Follow steps 4–6 as in row 2 3. Take 0.1 ml solution 4. Take 0.9 ml of distilled water again and mix well Can be mixed with dexamethasone for final concentration (step 4) |
|
4 |
Gentamycin (80 mg/2 ml) |
80 µg |
1. Take 0.2 ml of Gentamycin (8 mg/ 0.2 ml) 2. Dilute with 0.8 ml of distilled water 3. Mix well 4. Discard 0.9 ml of solution 5. Dilute with 0.9 ml of distilled water and mix well 6. Discard 0.9 ml of solution 7. Use 0.1 ml for injection |
|
5 |
Ciprofloxacin (200 mg/ 100 ml) |
100 µg |
Directly loaded from the sterile vial and injected intravitreally, 0.05 ml |
|
6 |
Moxifloxacin |
200 µg |
Take 0.05 ml of 0.5% moxifloxacin (preservative-free) |
|
7 |
Imipenem (250 mg) |
50–100 µg |
1. Dilute with 100 ml of distilled water 2. Take 0.2 ml (0.5 mg) |
4. Systemic Antibiotics And Steroid
The Endophthalmitis vitrectomy study (EVS) has recommended that systemic antibiotics (ceftazidime or amikacin) have no additional role in patients being treated by intravitreal injections or vitrectomy. (31) However, intravenous ceftazidime has been reported to achieve clinically effective intravitreal levels (i.e., levels greater than MIC90) against gram-negative organisms and streptococci in inflamed, aphakic, vitrectomized rabbit eyes. (44) Experimental studies have also shown that intravenous vancomycin and cefazolin also achieve clinically significant levels in the vitreous cavity of inflamed eyes, and would penetrate better and would provide better gram-positive coverage than amikacin or ceftazidime. (45,46)In view of the above limitations of the EVS design, the decision to use or not to use intravenous antibiotics in the management of postoperative endophthalmitis must be based on the clinical findings in the case and the judgment of the treating physician. Systemic antibiotics can be used in the setting of advanced infection in a one-eyed patient or in an immunocompromised host, post-traumatic endophthalmitis with IOFB, associated scleral buckle infection or corneal abscess. Vancomycin (1g iv 12 hourly) or cefazolin (1-2 g iv 8 hourly) may be an appropriate choice for gram-positive coverage and ceftazidime (1-2 g iv 8 hourly)may be appropriate for gram-negative coverage. Because vancomycin and ceftazidime are primarily excreted by the kidneys, appropriate dose adjustments should be done in patients with renal impairment. Serum urea nitrogen and creatinine levels can be monitored during the treatment. Oral ciprofloxacin can be used, particularly for coagulase-negative Staphylococcus species. It provides a broad spectrum of coverage (including Pseudomonas species) and also has good vitreous penetration. (47) To reduce the destructive effect of the significant inflammation that coexists with infection in endophthalmitis, systemic steroids can be used in combination with antibiotics, provided that no contraindications exist (e.g., diabetes mellitus, tuberculosis, fungal infection, etc.). It reduces the intraocular inflammatory process and secondary complications associated with bacterial endophthalmitis. (48)
5.Vitrectomy
Vitrectomy has the potential advantages of removing the infecting organism and associated toxins, removing vitreous membranes that could lead to retinal detachment, better posterior segment visualization, and improving the intraocular distribution of antibiotics. (48) However, the absence of vitreous might increase drug toxicity as well as rapid drug clearance. The EVS showed that in cases of acute endophthalmitis after cataract extraction or secondary intraocular lens (IOL) placement, initial vitrectomy did not achieve better results than a simple vitreous biopsy and intravitreal antibiotics if the initial visual acuity is hand movements or better. For patients with initial visual acuity of light perception only, substantially better visual outcomes were reported in the immediate three-port pars plana vitrectomy group versus the vitreous tap/biopsy group. (31) However, these results should be used to guide the management only of similar postoperative patients. The management of other patients (e.g. posttraumatic endophthalmitis) must be individualized and should be based on the clinician’s judgment. In patients with visual acuity is hand movements or better, the patient should be monitored for 24-36 hours after intravitreal antibiotics. If there is worsening, the patient has to be taken up for vitrectomy. If there is no worsening, medical treatment can be continued for 48 hours following which decision regarding additional intravitreal antibiotics or vitrectomy is to be taken. Removal of the IOL is usually not required, except in cases of chronic low-grade endophthalmitis associated with P. acnes.
Conclusion:
Management of endophthalmitis remains a challenge in spite of newer drugs, advances in diagnostic methods, and better surgical techniques. Early detection and timely treatment are of utmost importance to prevent devastating intraocular complications.
References:
- Packer M., Chang D.F., Dewey S.H. Prevention, diagnosis, and management of acute postoperative bacterial endophthalmitis.JCataract Refract Surg.2011;37:1699–1714.
- Stevenson LJ,Dawkins RCH,Sheorey H,McGuinness MB,Hurley AH,Allen PJ. Gram-negativeendophthalmitis: A prospective study examining the microbiology, clinical associations and visual outcomes following infection. Clin Exp Ophthalmol.2020 Apr 29. doi: 10.1111/ceo.13768. [Epub ahead of print]
- Callegan MC, Booth MC, Jett BD, Gilmore MS. Pathogenesis of gram-positive bacterial endophthalmitis.Infect Immun. 1999;67(7):3348‐3356.
- Jabbarvand M., Hashemian H., Khodaparast M., Jouhari M., Tabatabaei A., Rezaei S. Endophthalmitis occurring after cataract surgery: outcomes of more than 480 000 cataract surgeries, epidemiologic features, and risk factors.Ophthalmology.2016;123(2):295–301.
- Lundstrom M., Wejde G., Stenevi U., Thorburn W., Montan P. Endophthalmitis after cataract surgery: a nationwide prospective study evaluating incidence in relation to incision type and location.Ophthalmology.2007;114:866–870.
- Lundström M., Friling E., Montan P. Risk factors for endophthalmitis after cataract surgery: predictors for causative organisms and visual outcomes.JCataract Refract Surg.2015;41:2410–2416.
- Ng J.Q., Morlet N., Bulsara M.K., Semmens J.B. Reducing the risk for endophthalmitis after cataract surgery: population-based nested case-control study: endophthalmitis population study of Western Australia sixth report.JCataract Refract Surg.2007;33:269–280.
- ESCRS Endophthalmitis Study Group Prophylaxis of postoperative endophthalmitis following cataract surgery: results of the ESCRS multicenter study and identification of risk factors.JCataract Refract Surg.2007;33(6):978–988.
- Wallin T., Parker J., Jin Y., Kefalopoulos G., Olson R.J. Cohort study of 27 cases of endophthalmitis at a single institution.JCataract Refract Surg.2005;31:735–741.
- Jensen M.K., Fiscella R.G., Crandall A.S. Aretrospective study of endophtalmitis rates comparing quinolone antibiotics.Am J Ophthalmol.2005;139:141–148.
- Jensen M.K., Fiscella R.G., Moshirfar M., Mooney B. Third- and fourth-generation fluoroquinolones: retrospective comparison of endophthalmitis after cataract surgery performed over 10 years.JCataract Refract Surg.2008;34:1460–1467.
- Speaker MG, Menikoff JA: Prophylaxis of endophthalmitis with topical povidone-iodine. Ophthalmology 98:1769–1774, 1991
- Carrim ZI, Mackie G, Gallacher G, Wykes WN. The efficacy of 5% povidone-iodine for 3 minutes prior to cataract surgery. Eur J Ophthalmol 2009; 19:560–564.
- Hosseini H,Ashraf MJ,Saleh M,Nowroozzadeh MH,Nowroozizadeh B,Abtahi MB,Nowroozizadeh S. Effect ofpovidone-iodineconcentration and exposure time on bacteria isolated fromendophthalmitiscases. J Cataract Refract Surg.2012 Jan;38(1):92-6.
- Pinna A,Usai D,Sechi LA,Zanetti S,Jesudasan NC,Thomas PA,Kaliamurthy J. An outbreak of post-cataract surgeryendophthalmitiscaused byPseudomonasaeruginosa. Ophthalmology.2009 Dec;116(12):2321-6.e1-4.
- al Hazzaa SA, Tabbara KF, Gammon JA. Pink hypopyon: a sign of Serratia marcescens endophthalmitis.Br J Ophthalmol. 1992;76(12):764‐765.
- Rishi E,Rishi P,Sengupta S,Jambulingam M,Madhavan HN,Gopal L,Therese KL. Acute postoperative Bacillus cereusendophthalmitismimickingtoxic anterior segment syndrome. Ophthalmology.2013;120(1):181-5.
- Ang GS1,Varga Z,Shaarawy T. Postoperative infection in penetrating versus non-penetrating glaucoma surgery. Br J Ophthalmol.2010 Dec;94(12):1571-6.
- Stein JD, Ruiz D Jr, Belsky D, Lee PP, Sloan FA. Longitudinal rates of postoperative adverse outcomes after glaucoma surgery among medicare beneficiaries 1994 to 2005.Ophthalmology.2008;115(7):1116.e7.
- Mishra C,Lalitha P,Rameshkumar G,Agrawal R,Balne PK,Iswarya M,Kannan NB,Ramasamy K.Incidence of Endophthalmitis after Intravitreal Injections: Risk Factors, Microbiology Profile, and Clinical Outcomes. Ocul Immunol Inflamm.2018;26(4):559-568.
- Brinton GS, Topping TM, Hyndiuk RA, et al: Posttraumatic endophthalmitis. Arch Ophthalmol. 1984;102:547–550.
- Thompson WS, Rubsamen PE, Flynn HW Jr, et al: Endopthalmitis after penetrating trauma. Risk factors and visual acuity outcomes. Ophthalmology. 1995; 102:1696–1701.
- Rishi E, Rishi P, Koundanya VV, Sahu C, Roy R, Bhende PS. Post-traumatic endophthalmitis in 143 eyes of children and adolescents from India. Eye (Lond).2016;30(4):615-20.
- Novosad BD, Callegan MC. Severe bacterial endophthalmitis: towards improving clinical outcomes.Expert Rev Ophthalmol.2010;5(5):689–698.
- Jackson TL, Eykyn SJ, Graham EM, Stanford MR. Endogenous bacterial endophthalmitis: a 17-year prospective series and review of 267 reported cases.Surv Ophthalmol.2003;48(4):403–423.
- Schiedler V, Scott IU, Flynn HW, Jr, Davis JL, Benz MS, Miller D. Culture-proven endogenous endophthalmitis: clinical features and visual acuity outcomes.Am J Ophthalmol.2004;137(4):725–731.
- Sharma S, Padhi TR, Basu S, Kar S, Roy A, Das T. Endophthalmitis patients seen in a tertiary eye care centre in Odisha: a clinico-microbiological analysis.Indian J Med Res.2014;139(1):91–98.
- Brod RD, Flynn HW Jr, Clarkson JG, et al: Endogenous Candida endophthalmitis: management without intravenous amphotericin B. Ophthalmology. 1990;97:666–671.
- Maitray A,Rishi E,Rishi P,Gopal L,Bhende P,Ray R,Therese KL. Endogenous endophthalmitis in children and adolescents: Case series and literature review. Indian J Ophthalmol.2019;67(6):795-800.
- Kosacki J,Boisset S,Maurin M,Cornut PL,Thuret G,Hubanova R,Vandenesch F,Carricajo A,Aptel F,Chiquet C;Friends Group. Specific PCR and Quantitative Real-Time PCR in Ocular Samples from Acute and Delayed-Onset PostoperativeEndophthalmitis. Am J Ophthalmol.2020 Apr;212:34-42.
- The Endophthalmitis Vitrectomy Study Group: Results of the Endophthalmitis Vitrectomy Study: microbiologic factors and visual outcome in the Endophthalmitis Vitrectomy Study. Am J Ophthalmol 1996; 122: 830-846.
- Campochiaro PA, Lin JI, and Aminoglycoside Study Group: Aminoglycoside toxicity in the treatment of endophthalmitis. Arch Ophthalmol 1994; 112: 48-53.
- Buerk M, Fiscella R, Johnson S, Edward D. Intravitreal toxicity of linezolid in rabbits. Invest Ophthalmol Vis Sci 2004; 45: E-Abstract 4902.
- Pathengay A,Mathai A,Shah GY,Ambatipudi S. Intravitrealpiperacillin/tazobactam in the management of multidrug-resistant Pseudomonas aeruginosaendophthalmitis. J Cataract Refract Surg.2010 Dec;36(12):2210-1.
- Singh TH, Pathengay A, Das T, Sharma S.Enterobacterendophthalmitis: Treatment with intravitreal tazobactam- piperacillin. Indian J Ophthalmol 2007;55:482-3.
- Jindal A, Pathengay A, Khera M, et al. Combined ceftazidime and amikacin resistance among Gram-negative isolates in acute-onset postoperative endophthalmitis: prevalence, antimicrobial susceptibilities, and visual acuity outcome.J Ophthalmic Inflamm Infect.2013;3(1):62.
- Altan T, Acar N, Kapran Z, et al. Acute-onset endophthalmitis after cataract surgery: success of initial therapy, visual outcomes, and related factors.Retina.2009;29(5):606–612.
- Dave VP,Pathengay A,Braimah IZ,Panchal B,Sharma S,Pappuru RR,Mathai A,Tyagi M,Narayanan R,Jalali S,Das T. ENTEROCOCCUSENDOPHTHALMITIS: Clinical Settings, Antimicrobial Susceptibility, and Management Outcomes. Retina.2020 May;40(5):898-902.
- Rishi E, Rishi P, Nandi K, et al. Endophthalmitis caused by Enterococcus faecalis: a case series. Retina 2009;29:214–221.
- RishiE,Rishi P,Bhende P,Raman R,Sen P,Susvar P,Rao C,Therese L,Hirawat R. Enterococcus faecalis EndophthalmitisinChildren- A 21 Year Study. Ocul Immunol Inflamm.2018;26(4):543-549.
- Cetinkaya Y, Falk P, Mayhall CG. Vancomycin-resistantEnterococci.Clin Microbiol Rev.2000;13:686–707.
- Sambhav K, Mathai A, Reddy AK, Reddy BV, Bhatia K, Balne PK. Endogenous endophthalmitis caused byEnterococcus casseliflavus.J Med Microbiol.2011;60:670–2.
- Berenger BM, Kulkarni S, Hinz BJ, Forgie SE. Exogenous endophthalmitis caused by Enterococcus casseliflavus: A case report and discussion regarding treatment of intraocular infection with vancomycin-resistant enterococci.Can J Infect Dis Med Microbiol. 2015;26(6):330‐332.
- Aguilar HE, Meredith TA, Shaarawy A, et al: Vitreous cavity penetration of ceftazidime after intravenous administration. Retina. 1995;15:154–159.
- Alfaro DV, Liggett PE: Intravenous cefazolin in penetrating eye injuries. I. Effects of trauma and multiple doses on intraocular delivery. Graefes Arch Clin Exp Ophthalmol. 1994;232: 238–241.
- Meredith TA, Aguilar HE, Shaarawy A, et al: Vancomycin levels in the vitreous cavity after intravenous administration. Am J Ophthalmol. 1995;119:774–778.
- Lesk MR, Ammann H, Marcil G, et al: The penetration of oral ciprofloxacin into the aqueous humor, vitreous, and subretinal fluid of humans. Am J Ophthalmol. 1993;115:623–628.
- Kresloff MS, Castellarin AA, Zarbin MA. Endophthalmitis.Surv Ophthalmol1998;43: 193-224.

