The present overview highlights major microbiological aspects of mucormycosis and attempts to increase the awareness of this ubiquitous group of fungi that are responsible for the difficult-to-manage infections presently being encountered in the setting of the ongoing COVID-19 pandemic.
1. What is Mucormycosis and why is mucormycosis emerging as a serious threat in the present COVID-19 pandemic?
Mucormycosis(previously called zygomycosis; sometimes calledblack fungus infection) is a serious, but rare fungal infection caused by a group of molds belonging to the order Mucorales(1). Mucormycosis is a fulminant disease with high rates of morbidity and mortality that mainly affects immunocompromised patients. Depending on the location of the infection, the mucormycosis can be rhino-orbito-cerebral (most common), pulmonary, gastrointestinal, cutaneous, or disseminated. Although it has a low incidence rate, varying from 0.005 to 1.7 per million population, a sudden rise in cases of rhino-orbital mucormycosis in patients with past or current COVID-19 infection has been observed during the recent outbreak. A complex interplay of factors that include uncontrolled diabetes mellitus, any previous respiratory pathology, over-zealous use of steroids, nosocomial infection sources, and systemic immune alterations of Covid-19 infection has been suggested as the reason behind susceptibility to mucormycosis in COVID-19 patients (2,3).
2. What are the types of fungi that cause mucormycosis?
Several different types of fungi belonging to the order Mucorales cause mucormycosis. Rhizopus species are by far the predominant clinical isolates, among which Rhizopus oryzae occcurs most frequently. Species of Mucor, Lichtheimia (formerly Absidia), Rhizomucor, Saksenaea, Cunninghamella, and Apophysomyces, are the less common aetiologies (4).
3. Where these fungi are normally found?
Fungi causing mucormycosis are present throughout the environment, particularly in soil and in association with decaying organic matter (5). They are frequently seen growing on bread and fruits. These fungi rarely colonize humans. Since they have been found everywhere it is impossible to completely avoid coming in contact with these fungal spores. Outbreaks of mucormycosis have been linked to ventilation systems, contaminated wound dressings, hospital bed sheets, negative-pressure rooms, contaminated medical equipment, and construction work (6).
4. What are the virulence traits of fungi causing mucormycosis?
Mucormycosis results mostly from inhalation of fungal sporangiospores that have been released in the air or occasionally by ingestion or direct inoculation. These fungi have specific virulence factors that increase their pathogenicity in immunosuppressed individuals. The main predisposition to mucormycosis is a state of metabolic acidosis, usually secondary to diabetes mellitus (7,8). This condition decreases not only phagocytosis but also the migration and tissue accumulation of neutrophils and fibroblasts. Fungal growth increases in this glucose-rich, acidic tissue environment, because these fungi possess a ketone reductase system. Furthermore, the ability to acquire iron from the host is a major virulence trait of the Mucorales. During metabolic acidosis, serum unbound iron is increased because of the decreased iron capacity binding of transferrin (9). Mucorales, especially Rhizopus, thrives in the presence of unbound iron, as iron is an essential element for fungal cell growth and development. In addition, these fungi also have a marked propensity for vascular invasion and rapidly produce thrombosis and necrosis of tissue.
5. How is mucormycosis diagnosed microbiologically?
Mucormycosis may be rapidly fatal and hence timely laboratory diagnosis is crucial to guide clinicians in instituting appropriate treatment without delay. Microscopy and culture of clinical specimens are the cornerstones of diagnosing mucormycosis (10). However, it is important to collect proper clinical specimens (for example nasal discharges, scrapings and aspirates from sinuses, orbital biopsies in patients with rhino-orbital mucormycosis) in order to increase the sensitivity of microbiological testing.
6. What are the methods of direct detection of mucormycosis in the specimen?
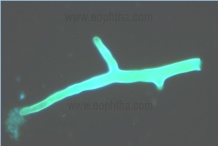
Specimens should be analyzed by microscopy using 10% potassium hydroxide (KOH), if possible with optical brighteners such as Calcofluor white staining solution (11). Hyphae of these fungi are broad, 10-15 µM in diameter, nonseptate or sparsely septate, and have an irregular, ribbon-like appearance. Branching is irregular, often at right angles (Figure 1). Apositive direct microscopy, especially from a sterile site, must be considered significant, even if the laboratory is unable to culture the fungus. A direct microscopy, however, will not be able to identify a fungus to the genus or species level. Histopathology is a very important diagnostic tool since it distinguishes the presence of the fungus as a pathogen in the specimen from a culture contaminant and is indispensable to define whether there is blood vessel invasion. Histopathological examination of fixed tissue sections with haematoxylin and eosin (H&E) or specialized fungal stains, such as Grocott methenamine-silver (GMS) or periodic acid-Schiff (PAS) stains show broad-based, ribbon-like, non-septate hyphae with wide-angle branching (12). However, to detect hyphae, multiple specimens are often necessary.
7. How is the culture of the specimens for mucormycosis done?
The culture of specimens is extremely essential for the diagnosis of mucormycosis and it allows identification to the genus and species level (13). The major concern about culture, however, is its low sensitivity, as it can be falsely negative in up to 50% of mucormycosis cases (14). This can be attributed to a number of reasons, such as grinding or homogenization of tissue specimens, which may destroy the delicate hyphae of these fungi, recent or ongoing therapy with antifungals effective on Mucorales, or even a lack of expertise. Proper sampling and handling of the specimens before the examination are a prerequisite for an optimal yield. A positive culture from a sterile site confirms the diagnosis, while a positive culture from a non-sterile site could be due to a contaminant and must be combined with clinical and radiological data to establish a probable diagnosis. Mucorales grow well on most fungal culture media, such as Sabouraud’s dextrose agar incubated at 25–30°C. Colonies characteristically produce a fluffy, white to gray, or brown hyphal growth that diffusely covers the entire surface of the agar in 24 to 48 hrs (Figure 2). The fungal growth is so vigorous that they lift the lid of the Petri plate and hence this group has come to be known as ‘lid lifters'.

8. How the fungus is conventionally identified in the lab to genus/species level?
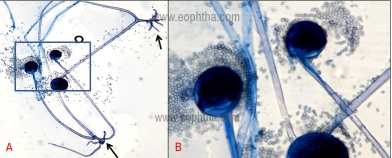
Identification of the agents responsible for mucormycosis is mainly based on macroscopic and microscopic morphological observations. Important macroscopic features are a hyaline appearance, vigorous growth, light coloration on the reverse side of the plate (tan to yellow for most species), and variable degrees of coloration on the sporulating surface of the colonies (from pure white to tan, brown, grey or even black). It is impossible to distinguish between the genera and species of mucormycosis based on colony morphology. Microscopically, the fungi can be observed using a Lactophenol cotton blue staining to achieve better visualization. Mucorales are characterized by large, ribbon-like hyphae that are irregular in diameter and are nonseptate. A globose fruiting structure, sporangium forms at the tip of a specialized branch called sporangiophore. The sporangium develops small spherical yellow or brown sporangiospores (Figure 3). (These spores are released for aerial dispersal when the wall of the mature sporangium ruptures). The identification of the three common genera, Rhizopus, Mucor, and Absidia is based on the absence or presence of rhizoids and the position of the rhizoids in relation to the sporangiophores (15).
9. What are the latest molecular methods used in the diagnosis of mucormycosis?
In recent years, advances in molecular biology have significantly contributed to the development of rapid, accurate, and sensitive methods for the detection of mucormycosis. The most commonly used are conventional polymerase chain reactions (PCRs), quantitative PCRs, RFLP (Restriction fragment length polymorphism), and DNA sequencing of defined gene regions (16-18). The majority of the molecular assays are based on the internal transcribed spacer (ITS) region that has been chosen as the pan-fungal barcode by the International Subcommission on Fungal Barcoding (19). All these techniques can also be used for the identification of mucormycetes from cultures. The efficiency of these in-house assays presently lacks thorough clinical evaluation and therefore cannot be recommended as a stand-alone, single approach in clinical routine diagnostics. However, they can be recommended as valuable add-on tools that complement conventional diagnostic procedures. MALDI-TOF (Matrix-assisted laser desorption ionization-time of flight mass spectrometry), a technique that measures the mass of molecules from a sample that has been embedded in a matrix by using a laser to ablate and desorb the molecules with minimal fragmentation, has been foundpromising for thespecies identification of mucormycetes but is not yet validated for all Mucorales (20).
10. Can we perform in vitro antifungal susceptibility testing of the culture isolate in the lab?
Liposomal amphotericin B is the therapy of choice for this condition. The second-line anti-fungal drugs include extended-spectrum triazoles, posaconazole, and isavuconazole. As the interpretive Minimum Inhibitory Concentration (MIC) breakpoints of various antifungal drugs have not yet been defined for members of this group of fungi and the correlations between clinical response and MIC values for a given strain are uncertain, the use of antifungal susceptibility testing in mucormycosis for routine clinical decisions is not recommended at this time (10).
Summary: Mucormycosis has emerged as a serious long-term manifestation of Covid-19 infection. The diagnosis of mucormycosis remains challenging. Histopathology, direct examination, and culture remain essential tools. Molecular assays have shown promise both for the detection or identification of mucormycetes, and these are now valuable add-on tools that complement conventional diagnostic procedures.
References
- Kwon-Chung KJ. Taxonomy of fungi causing mucormycosis and entomophthoramycosis (zygomycosis) and nomenclature of the disease: molecular mycologic perspectives. Clinical Infectious Diseases. 2012 Feb 1;54(suppl_1):S8-15.
- Sharma S, Grover M, Bhargava S, Samdani S, Kataria T. Post coronavirus disease mucormycosis: a deadly addition to the pandemic spectrum. The Journal of Laryngology & Otology. 2021 Apr 8:1-6.
- Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Yu T. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. The lancet. 2020 Feb 15;395(10223):507-13.
- Skiada A, Pavleas I, Drogari-Apiranthitou M. Epidemiology and Diagnosis of Mucormycosis: An Update. Journal of Fungi. 2020 Dec;6(4):265.
- Richardson M. The ecology of the Zygomycetes and its impact on environmental exposure. Clinical Microbiology and Infection. 2009 Oct;15:2-9.
- Rammaert B, Lanternier F, Zahar JR, Dannaoui E, Bougnoux ME, Lecuit M, Lortholary O. Healthcare-associated mucormycosis. Clinical Infectious Diseases. 2012 Feb 1;54(suppl_1):S44-54.
- Petrikkos G, Skiada A, Lortholary O, Roilides E, Walsh TJ, Kontoyiannis DP. Epidemiology and clinical manifestations of mucormycosis. Clinical Infectious Diseases. 2012 Feb 1;54(suppl_1):S23-34.
- Chakrabarti A, Das A, Mandal J, Shivaprakash MR, George VK, Tarai B, Rao P, Panda N, Verma SC, Sakhuja V. The rising trend of invasive zygomycosis in patients with uncontrolled diabetes mellitus. Sabouraudia. 2006 Jun;44(4):335-42.
- Ibrahim A, Spellberg B, Edwards Jr J. Iron Acquisition: a novel prospective on mucormycosis pathogenesis and treatment. Current opinion in infectious diseases. 2008 Dec;21(6):620.
- Lass‐Flörl C. Zygomycosis: conventional laboratory diagnosis. Clinical Microbiology and Infection. 2009 Oct;15:60-5.
- Monheit JE, Cowan DF, Moore DG. Rapid detection of fungi in tissues using calcofluor white and fluorescence microscopy. Archives of pathology & laboratory medicine. 1984 Aug 1;108(8):616-8.
- Frater JL, Hall GS, Procop GW. Histologic features of zygomycosis: emphasis on perineural invasion and fungal morphology. Archives of pathology & laboratory medicine. 2001 Mar;125(3):375-8.
- Lackner M, Caramalho R, Lass-Flörl C. Laboratory diagnosis of mucormycosis: current status and future perspectives. Future microbiology. 2014 May;9(5):683-95.
- Walsh TJ, Gamaletsou MN, McGinnis MR, Hayden RT, Kontoyiannis DP. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clinical Infectious Diseases. 2012 Feb 1;54(suppl_1):S55-60.
- Bouza E, Munoz P, Guinea J. Mucormycosis: an emerging disease?. Clinical Microbiology and Infection. 2006 Dec;12:7-23.
- Nagao K, Ota T, Tanikawa A, Takae Y, Mori T, Udagawa SI, Nishikawa T. Genetic identification and detection of human pathogenic Rhizopus species, a major mucormycosis agent, by multiplex PCR based on internal transcribed spacer region of rRNA gene. Journal of dermatological science. 2005 Jul 1;39(1):23-31.
- Larché J, Machouart M, Burton K, Collomb J, Biava MF, Gé; rard A, Fortier B. Diagnosis of cutaneous mucormycosis due to Rhizopus microsporus by an innovative PCR-restriction fragment-length polymorphism method. Clinical infectious diseases. 2005 Nov 1;41(9):1362-5.
- Kasai M, Harrington SM, Francesconi A, Petraitis V, Petraitiene R, Beveridge MG, Knudsen T, Milanovich J, Cotton MP, Hughes J, Schaufele RL. Detection of a molecular biomarker for zygomycetes by quantitative PCR assays of plasma, bronchoalveolar lavage, and lung tissue in a rabbit model of experimental pulmonary zygomycosis. Journal of clinical microbiology. 2008 Nov;46(11):3690.
- Consortium for the Barcode of Life (CBOL). www.barcoding.si.edu.
- De Carolis E, Posteraro B, Lass-Fl C, Vella A, Florio AR, Torelli R, Girmenia C, Colozza C, Tortorano AM, Sanguinetti M, Fadda G. Species identification of Aspergillus, Fusarium and Mucorales with direct surface analysis by matrix-assisted laser desorption ionization time-of-flight mass spectrometry. Clinical microbiology and infection. 2012 May 1;18(5):475-84.
