Posterior capsular rupture (PCR) is an important complication of cataract surgery whose timely recognition and appropriate management can ensure a good visual outcome. Here are ten pearls for its prevention and management.
Pearl 1: Understand the risk/predisposing factors
- It is imperative to assess the preoperative and intra-operative risk to prevent the development of a PCR.
- Elderly, anxious, and uncooperative patients are at a higher risk of developing posterior capsular rupture. Extraocular factors include a deep socket, narrow palpebral fissures, exaggerated bell’s phenomenon, hazy cornea, and intraocular factors are poor mydriasis, shallow or excessively deep anterior chamber, pseudoexfoliation, and type of cataract (like posterior polar, traumatic, post-intravitreal injections, intumescent or hard cataract), previous vitrectomy or oil-filled eyes.
- Surgical factors like inexperience of the surgeon, leaking incisions, small capsulorhexis, radial tears in rhexis, fluid imbalance in phacoemulsification leading to surge also contribute to the development of a PCR.
Pearl 2: Preventive strategies
- Comprehensive slit-lamp examination to assess the corneal clarity, anterior chamber depth, mydriasis, zonular stability, type, and grade of cataract is essential.
- Any signs of complicated cataract surgery in the fellow eye may be an indicator for intraoperative challenges.
- High-risk cases should be operated on by an experienced surgeon.
- Peribulbar anesthesia with intravenous sedation should be opted over topical anesthesia in anxious, elderly, or uncooperative patients. Gentle and steady ocular massage will reduce intra-operative vitreous upthrust. Preoperative mannitol should be administered in angle-closure glaucoma patients.
- Proper wound construction, staining the anterior capsule, avoiding any radial extension or rhexis margins tears, and a good hydrodissection is essential to prevent PCR.
- Cortical cleaving hydrodissection is contraindicated in posterior polar cataract, post-vitrectomy/intravitreal injection eyes, and traumatic cataracts where there is damage to the posterior capsule.
- The anterior chamber should be adequately filled with OVD during cortex aspiration and IOL implantation.
Pearl 3: Preoperative work-up in PPC/Pre-existing rent
- The common etiologies for pre-existing PCR are posterior polar cataract, blunt or penetrating ocular trauma, previous intravitreal injections, or congenital weakness in the setting of PHPV or posterior lenticonus.
- Careful slit-lamp biomicroscopy of a PPC may demonstrate a central defect or discontinuity of the light beam with refractile lens particles floating in the anterior vitreous, indicating pre-existing dehiscence.
- Imaging techniques like UBM (20-35MHz probes), AS-OCT, or Scheimpflug imaging on Pentacam can help diagnose a pre-existing PCR. Fine vitreous opacities on USG-B scan may indicate leaked-out lens material in vitreous.
Pearl 4: Intraoperative tips and early recognition
- Proper adjustment of the microscope magnification and focus, and checking the coaxial illumination before beginning the surgery is essential.
- The use of a lightweight speculum avoids any pressure on the globe.
- Self-sealing wound architecture prevents chamber collapse during surgery.
- Use trypan blue to stain the anterior capsule whenever necessary (for beginners, white cataracts, advanced sclerotic cataracts, cortical cataracts, and others that block red glow).
- The use of microforceps to grasp and pull the anterior capsule centripetally helps achieve a good rhexis in pediatric elastic capsules, intumescent cataracts, and whenever there is a tendency for rhexis runoff.
- The anterior chamber should be decompressed to burp out the OVD before starting hydrodissection. This step must be performed carefully to prevent a rise in intralenticular pressure and posterior capsular blowout. A gentle tap on the nucleus allows the uniform spread of fluid waves. Hydrodissection is unnecessary in liquefied white cataracts and must be avoided in posterior polar cataracts. A pupil snap is noticed wherein the pupil suddenly and briskly constricts following a PCR at this stage (due to posterior displacement of the lens diaphragm). The surgeon must convert to a non-phaco technique upon its recognition. In case the snap sign is missed, and the surgeon continues with phacoemulsification, there will be difficulty in sculpting, and an asymmetric deepening of the anterior chamber or tilting of the nucleus will be noticed.
- During phacoemulsification, care must be taken not to trench the nucleus too deep. Avoid any rhexis margin tears which may extend posteriorly on nuclear manipulation. [Figure 1a] Decreasing the parameters or using additional OVD for emulsification of the last nuclear fragment helps to prevent PCR at this stage. [Figure 1b] Newer machines with active fluidics with surge control allow for chamber maintenance during surgery.
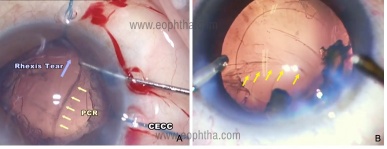
Figure 1:Intraoperative image showing posterior capsular rent A) during the last fragment removal B) following rhexis margin tear
- In the presence of a PC rent the nucleus moves less readily towards the phaco tip (loss of followability) due to fluid outflow through the rent and vitreous prolapse into the anterior chamber.
- Difficulty in rotating a previously mobile nucleus or bizarre behavior of the nucleus due to vitreous entrapment also indicates a PCR.
- During irrigation aspiration (I/A), the aspiration tip must be facing away from the posterior capsule. This step can be performed from the side port to ensure a formed, anterior chamber and keep the posterior capsule away.
- Development of PCR during I/A is evidenced by converging wrinkles (towards the aspiration port) of the PC (spider sign), brighter red reflex in the zone of PCR, pupillary peaking, and OVD going directly into the vitreous cavity rather than inflating the bag.
- Thus, the early indicators of PCR are:
-
- Sudden deepening of AC
- Momentary and abrupt pupil constriction (snap sign) during hydrodissection
- Localised intensification of red reflex
- Loss of nuclear fragment followability or rotation
- Bizzare behaviour or tilting of the nucleus
-
Pearl 5: OVD Strategy and removal of the nucleus
- The first priority in managing PCR is to prevent a collapse of the anterior chamber and the rent extension.
- Phaco foot pedal recedes to foot position 1(only irrigation), bottle height is lowered, the second instrument is withdrawn, and a dispersive OVD like VISCOAT (or any OVD that is available to the surgeon) is injected through the side port in the area of the PCR (viscotamponade). This plugs the posterior capsular tear, prevents its extension and limits the vitreous prolapse. The foot pedal is released gradually, and then the phaco probe can be withdrawn. Anterior chamber depth must be maintained for all subsequent procedures.
- The nuclear fragments can be removed by:
- Phacoemulsification using reduced fluidics settings after bringing the nuclear pieces to a safe position away from the PCR using the Viscoshield Strategy where the OVD creates a barrier between the nuclear fragments and vitreous. But - Do not Phaco in the presence of vitreous which exerts massive vitreoretinal traction!
- Phacoemulsification using the IOL scaffold technique, in which a multi-piece hydrophobic acrylic intraocular lens is placed at a plane anterior to the posterior capsular rent, and the nucleus is emulsified in the anterior chamber in front of the intraocular lens. This should be done with adequate endothelial protection using repeat injection of OVDs. IOL scaffold strategy should not be taken lightly and the surgical team should be familiar with alternate methods of IOL fixation in scenarios when mere sulcus support is not an option (large capsular deficiency situations).
- Manual removal after extending the incision and converting into a Manual SICS [Figure 2] or conventional ECCE.
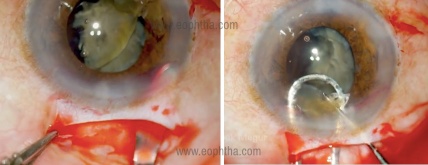
Figure 2: Intraoperative image showing conversion to manual SICS and manual extraction of nuclear fragments (courtesy Dr.Deepak Megur)
Pearl 6: Anterior vitrectomy and cortical removal tips
- Topical anesthesia may need to be augmented with a subtenon or peribulbar block, and the dose of midazolam can be increased for intravenous sedation in patients requiring monitored anesthesia care.
- The use of preservative-free triamcinolone acetonide, which is diluted with BSS (1:10) to obtain a dose of 4mg/ml, helps to identify vitreous strands in the anterior chamber. It has changed the way we manage vitreous loss today. [Figure 3a]
- Bimanual anterior vitrectomy should be performed with separated irrigation and vitrectomy/aspiration through two side ports preventing any chamber collapse.
- Irrigation should be kept anterior to the iris and facing upwards to prevent any hydration of the vitreous. The first cut is made slightly behind the rent to sever the stalk of the vitreous connecting the prolapsed vitreous in the anterior chamber and the vitreous in the posterior chamber, followed by clearing of the loose mass of vitreous in the anterior chamber. A limited amount of vitreous behind the PCR is removed.
- The cut rate is kept at the highest possible while bottle height, vacuum, and AFR are kept fairly low.
- Modern machine settings include 4000 cuts/min, vacuum 150 mmHg, AFR of 10 mm Hg using a 23gauge cutter, and irrigation-cut-aspiration (ICA) mode.

Figure 3 :A)Use of Triamcinolone as a guide to anterior vitrectomy B)Pars plana approach for anterior vitrectomy
- The bimanual pars-plana anterior vitrectomy approach using the 23-gauge trocar-cannula system is advocated by many surgeons. It enables safe vitrectomy by reducing vitreoretinal traction and avoiding excessive removal of the vitreous. [Figure 3b]
- The use of intracameral miochol or pilocarpine to constrict the pupil may help to identify any residual vitreous strands which lead to a peaked pupil.
- Repeat injection of intracameral triamcinolone acetonide before closing the case also helps identify residual vitreous strands and is strongly recommended.
- Following vitrectomy, the mode can be changed to irrigation-aspiration-cut (IAC), and the same cutter can then be used for cortex aspiration.
- Cortical removal is accomplished at very low flow, and the irrigation should be directed farthest from the area of PCR. The cortex is engaged from the peripheral area, stripped and brought to the central area, and aspirated. The cortical matter in the area of PCR is removed last. Bimanual I/A is preferred by many over coaxial I/A because of the control it offers. Dry cortex aspiration with the help of OVD and using a Simcoe or hydrodissection cannula can also be employed.
Pearl 7: Posterior continuous curvilinear capsulorhexis (PCCC)
- A PCCC is considered after an accidental PCR without a vitreous loss that is sufficiently small and central to allow its creation. OVD is injected carefully on top of and around the area of the PCR. A 2-3mm posterior rhexis is performed with microforceps by multiple grasping under high magnification and retro illumination. [Figure 4]
- This limits the extension of PCR, and a single piece foldable IOL can still be placed in the capsular bag.
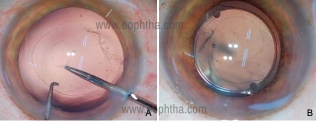
Figure 4: Intraop image showing conversion of PC rent into a PCCC and B) in the bag IOL placement of a foldable IOL (courtesy Dr.Sourabh Patwardhan)
Pearl 8: Posterior assisted levitation (PAL)
- It is considered a controversial technique.
- If the nucleus is still in the patellar fossa or anterior vitreous, one can use a vectis or a posterior assisted levitation to retrieve it into the anterior chamber as long as there is no vitreous in the anterior chamber.
- A disposable 20 G MVR blade is passed through the pars plana at 11’o clock, 3.5mm posterior to the limbus, to elevate the nuclear piece through the capsule.
- The nucleus is removed after enlarging the incision using OVD, Vectis, or Sheet’s glide.
- If the nucleus has dropped into the mid-vitreous or beyond, it is best handled by a vitreoretinal surgeon.
Pearl 9: IOL options
- In cases with small PCR, especially after a primary posterior capsulorhexis, an experienced surgeon can still implant an IOL in the capsular bag.
- If the rent is large or peripheral (when PCCC is not possible), it is better to perform a limited bimanual anterior vitrectomy if needed and place a three-piece IOL (0.5 D less) in the sulcus. The attributes of a sulcus fixated 3 piece IOL include large optic and overall IOL diameter with non-square edge configuration.
- Posterior optic capture/buttonholing through the CCC should be done if the rhexis size <5-5.5mm since it promotes stabilization of a sulcus fixated IOL locking it in place. It is not needed to adjust IOL power in situations where posterior optic capture has been achieved.For in-the-bag foldable IOL placed in large PC rent situations, reverse optic capture is mandatory. [Figure 5]
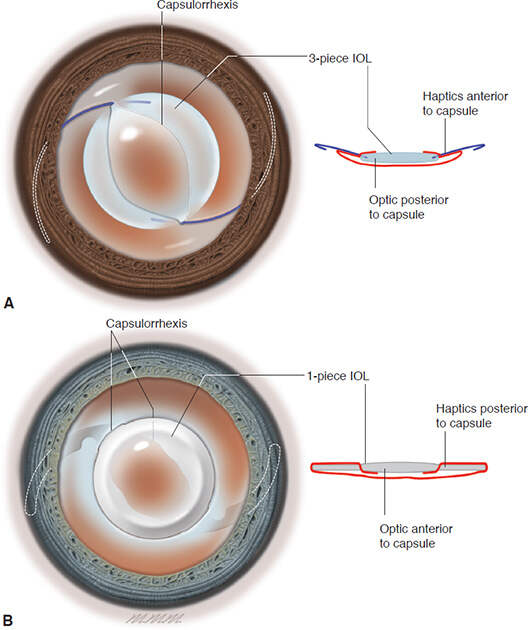
Figure 5: A,IOL optic capture. A 3-piece IOL with haptics in the sulcus. The optic is “captured” within the capsule so that the anterior capsule edge is anterior to the optic.B,IOL reverse optic capture. A 1-piece IOL with the optic in the sulcus. The optic is “captured” by capsulorhexis so that the anterior capsule edge is anterior to the haptics (Figure developed by Natalie Afshari, MD, and illustrated by Mark Miller. Excerpted fromBCSC 2020-2021 series: Section 11 - Lens and Cataract)
- If there is a PCR with inadequate anterior and posterior capsular support, an anterior chamber ACIOL, iris fixated IOL, or scleral fixated (sutured/unsutured) IOL can be used.
- Following complicated surgeries with a poor endothelial count, the patient can be left aphakic, and secondary IOL implantation can be undertaken after 4-8 weeks.
- Advanced IOL Scenario (Toric and Multifocal IOLs)
-
- A competent surgeon can still implant a multifocal or a toric IOL in the desired axis with the slow unfolding of haptics into the capsular bag in the presence of a small PCR.
- The above manouvere becomes easier if the surgeon has performed a PCCC.
- Multifocal or Toric IOLs should be abandoned if the surgeon is not confident or there is extensive posterior capsular damage.The surgeon should always have a 3-piece IOL as a stand-by.
- A single-piece acrylic lens should never be implanted in the ciliary sulcus.
-
Pearl 10: Prevention of post-operative complications
- Proper preoperative counselling of the patient is important. It is a good practice to inform the patient or the bystanders of the complication and have a frank discussion on the consequences.
- A surgical peripheral iridectomy (PI) can be created efficiently with the vitrector using a very low cut-rate (30-50 cuts/min), low aspiration flow rate (12cc/min), and high vacuum (300-400mmHg) to prevent vitreopupillary block.
- Intracameral antibiotic (0.1 ml of 0.5% moxifloxacin) is given at the end of the surgery, and a wound check is done and sutured with 10-0 nylon to prevent endophthalmitis.
- More frequent topical steroids, cycloplegics, and antiglaucoma medications may be required to control inflammation and glaucoma.
- Topical NSAIDs may be used for a longer duration of up to 3 months postoperatively to prevent cystoid macular edema.
- Post-operative vitreoretinal consultations to look for any macular edema or peripheral retinal breaks may be necessary on follow-up
Suggested reading:
- Chakrabarti A, Nazm N. Posterior capsular rent: Prevention and management.Indian J Ophthalmol. 2017;65:1359-1369.
- Arup Chakrabarti. Posterior capsular rent. Genesis and management. Springer India; 2017.
- Hong AR, Sheybani A, Huang AJ. Intraoperative management of posterior capsular rupture.Curr Opin Ophthalmol. 2015;26(1):16-21.
- Harbansh Lal. Management of posterior capsular tear. AIOS CME series. 2003
- Astbury N, Wood M, Gajiwala U, et al. Management of capsular rupture and vitreous loss in cataract surgery.Community Eye Health. 2008;21(65):6-8.

